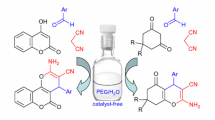
Abstract
A wide variety of carbonyl compounds can be transformed to their corresponding cyanohydrins in a single step using a dimethyl sulfoxide (DMSO)–water system in excellent yields (75–94%). The major advantages of this system are that the reaction conditions are mild and neutral; the reaction proceeds without catalyst and gives the corresponding cyanohydrins in short time (15–120 min).
Graphical Abstract



Similar content being viewed by others
References
Gregory R (1999) Chem Rev 99:3649
North M (2003) Tetrahedron Asymmetr 14:147
Groutas WC, Felker D (1980) Synthesis 861
North M, Usanov DL, Young C (2008) Chem Rev 108:5146
Surya KD, Richard AG (2005) J Mol Catal A: Chem 232:123
Chen F, Feng X, Jiang Y (2003) Arkivoc (ii):21
Cordoba R, Csaky AG, Plumet J (2004) Arkivoc (iv):94
Lundgren S, Ihre H, Moberg C (2008) Arkivoc (vi):73
Saravanan P, Anand RV, Singh VR (1998) Tetrahedron Lett 39:38
Gassman PG, Talley JJ (1978) Tetrahedron Lett 40:3773
Jenner G (1999) Tetrahedron Lett 40:491
Whitesell JK, Apodaca R (1996) Tetrahedron Lett 37:2525
Curini M, Epifanio F, Marcotullio MC, Rosati O, Rossi M (1999) Synlett 315
Chen F, Feng X, Qin B, Zhang G, Jiang Y (2003) Synlett 558
Wang X, Tian S (2007) Tetrahedron Lett 48:6010
Surya Prakash GK, Vaghoo H, Panja C, Surampudi V, Kultyshev R, Mathew T, Olah GA (2007) Proc Natl Acad Sci 104:3026
Sharma YO, Degani MS (2009) Green Chem 11:526
Sharma YO, Degani MS (2007) J Mol Cat A: Chem 277:215
Bag S, Vaze VV, Degani MS (2006) J Chem Res 267
Chavan SS, Sharma YO, Degani MS (2009) Green Chem Lett Rev 175
Terashima S, Jew S (1977) Tetrahedron Lett 18:1005
Berezin GH (1972) US Patent 3 697 593; (1972) Chem Abstr 77:61845
Gray G, Toyne K, Lacey D, Holmes D (1992) Eur Pat Appl 333 760 B1
Kirsten CN, Herm M, Schrader TH (1997) J Org Chem 62:6882
Marco JL, Ingate ST, Chinchon PM (1999) Tetrahedron 55:7625
Schmidt M, Herve S (1996) Tetrahedron 5:7833
Nanda S, Kato Y (2006) Tetrahedron Asymmetr 17:735
Ohno H, Mori A, Inoue S (1993) Chem Lett 22:375
Gassman PG, Talley JJ (1990) Organic syntheses collect Vol VII. Wiley, New York, p 20
Kiyoshi K, Hiroki S, Chikara U (2006) WO/2006/090279 A1; (2006) Chem Abstr 154:292877
Acknowledgments
The authors N.H.P.D. and R.B. thank UGC, and M.D.K. thanks DBT, New Delhi for their financial support. Authors are grateful to Centaur Pharmaceuticals Pvt. Ltd. for recording NMR spectra.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Degani, M.S., Kakwani, M.D., Palsule Desai, N.H. et al. A single-step, mild, neutral, catalyst-free method for cyanohydrin synthesis. Monatsh Chem 143, 461–465 (2012). https://doi.org/10.1007/s00706-011-0613-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-011-0613-4