Abstract
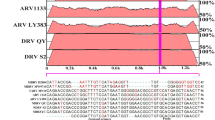
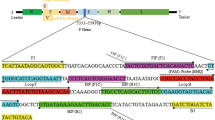
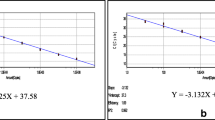
A new duck Tembusu virus (TMUV), also known as BYD virus, has been identified as the causative agent for the emerging duck egg-drop syndrome in mainland China. The rapid spread and wide distribution of the new TMUV in mainland China result in heavy loss to the poultry industry and pose great threats to public health. Rapid and sensitive detection methods are critical for prevention and control of TMUV infections. In this study, a reverse-transcription loop-mediated isothermal amplification assay (RT-LAMP) and an SYBR Green-I–based real-time RT-PCR assay specific for the duck TMUV were developed and validated with laboratory and field samples, respectively. The detection limits were 1 × 10−4 and 1 × 10−3 PFU per reaction for the RT-LAMP assay and real-time RT-PCR assay, respectively. The specificities were analyzed with other related members of the genus Flavivirus, and no cross-reaction was observed. Furthermore, both assays were evaluated with field samples, and they exhibited high sensitivity and specificity. In addition, the real-time RT-PCR assay worked well in viral load analysis, which revealed that the spleen may be the primary target for the replication of new duck TMUV in ducks. The TMUV-specific RT-LAMP and real-time RT-PCR assays will provide useful tools for the diagnosis and epidemiological surveillance of TMUV infection.




Similar content being viewed by others
References
Barban V, Girerd Y, Aguirre M, Gulia S, Petiard F, Riou P, Barrere B, Lang J (2007) High stability of yellow fever 17D–204 vaccine: a 12-year restrospective analysis of large-scale production. Vaccine 25:2941–2950
Cao Z, Zhang C, Liu Y, Ye W, Han J, Ma G, Zhang D, Xu F, Gao X, Tang Y, Shi S, Wan C, He B, Yang M, Lu X, Huang Y, Diao Y, Ma X (2011) Tembusu virus in ducks, china. Emerg Infect Dis 17:1873–1875
Chvala S, Bakonyi T, Hackl R, Hess M, Nowotny N, Weissenbock H (2006) Limited pathogenicity of usutu virus for the domestic goose (Anser anser f. domestica) following experimental inoculation. J Vet Med B Infect Dis Vet Public Health 53:171–175
Gao X, Nasci R, Liang G (2010) The neglected arboviral infections in mainland China. PLoS Negl Trop Dis 4:e624
Hubalek Z (2004) An annotated checklist of pathogenic microorganisms associated with migratory birds. J Wildl Dis 40:639–659
Ji J, Du LQ, Xie QM, Cao YC, Zuo KJ, Xue CY, Ma JY, Chen F, Bee YZ (2009) Rapid diagnosis of duck plagues virus infection by loop-mediated isothermal amplification. Res Vet Sci 87:53–58
Ji J, Xie QM, Chen CY, Bai SW, Zou LS, Zuo KJ, Cao YC, Xue CY, Ma JY, Bi YZ (2010) Molecular detection of Muscovy duck parvovirus by loop-mediated isothermal amplification assay. Poult Sci 89:477–483
Jiang T, Liu J, Deng YQ, Xu LJ, Li XF, Han JF, Cao RY, Qin ED, Qin CF (2011) Development and evaluation of a reverse transcription-loop-mediated isothermal amplification assay for rapid detection of enterovirus 71. J Clin Microbiol 49:870–874
Kono Y, Tsukamoto K, Abd Hamid M, Darus A, Lian TC, Sam LS, Yok CN, Di KB, Lim KT, Yamaguchi S, Narita M (2000) Encephalitis and retarded growth of chicks caused by Sitiawan virus, a new isolate belonging to the genus Flavivirus. Am J Trop Med Hyg 63:94–101
Notomi T, Okayama H, Masubuchi H, Yonekawa T, Watanabe K, Amino N, Hase T (2000) Loop-mediated isothermal amplification of DNA. Nucleic Acids Res 28:e63
Parida M, Horioke K, Ishida H, Dash PK, Saxena P, Jana AM, Islam MA, Inoue S, Hosaka N, Morita K (2005) Rapid detection and differentiation of dengue virus serotypes by a real-time reverse transcription-loop-mediated isothermal amplification assay. J Clin Microbiol 43:2895–2903
Parida M, Sannarangaiah S, Dash PK, Rao PV, Morita K (2008) Loop mediated isothermal amplification (LAMP): a new generation of innovative gene amplification technique; perspectives in clinical diagnosis of infectious diseases. Rev Med Virol 18:407–421
Richardson J, Molina-Cruz A, Salazar MI, Wt Black (2006) Quantitative analysis of dengue-2 virus RNA during the extrinsic incubation period in individual Aedes aegypti. Am J Trop Med Hyg 74:132–141
Su J, Li S, Hu X, Yu X, Wang Y, Liu P, Lu X, Zhang G, Liu D, Li X, Su W, Lu H, Mok NS, Wang P, Wang M, Tian K, Gao GF (2011) Duck egg-drop syndrome caused by BYD virus, a new Tembusu-related flavivirus. PLoS One 6:e18106
Tang Y, Diao Y, Yu C, Gao X, Chen L, Zhang D (2011) Rapid detection of Tembusu virus by reverse-transcription, loop-mediated isothermal amplification (RT-LAMP). Transbound Emerg Dis 59:208–213
Wang Y, Yuan X, Li Y, Yu K, Yang J, Xu H, Zhang Y, Liao M, Qin Z (2011) Rapid detection of newly isolated Tembusu-related Flavivirus by reverse-transcription loop-mediated isothermal amplification assay. Virol J 8:553
Yan L, Yan P, Zhou J, Teng Q, Li Z (2011) Establishing a TaqMan-based real-time PCR assay for the rapid detection and quantification of the newly emerged duck Tembusu virus. Virol J 8:464
Yan P, Zhao Y, Zhang X, Xu D, Dai X, Teng Q, Yan L, Zhou J, Ji X, Zhang S, Liu G, Zhou Y, Kawaoka Y, Tong G, Li Z (2011) An infectious disease of ducks caused by a newly emerged Tembusu virus strain in mainland China. Virology 417:1–8
Acknowledgments
This work was supported, in part, by the Major Special Program of National Science and Technology of China (No.2009ZX10004-204), and the National Natural Science Foundation of China (No. 30972613, No. 81000722). CFQ was supported by Beijing Nova Program of Science and Technology (No.2010B041).
Conflict of interests
The authors have declared that no competing interests exist.
Author information
Authors and Affiliations
Corresponding author
Additional information
T. Jiang and J. Liu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Jiang, T., Liu, J., Deng, YQ. et al. Development of RT-LAMP and real-time RT-PCR assays for the rapid detection of the new duck Tembusu-like BYD virus. Arch Virol 157, 2273–2280 (2012). https://doi.org/10.1007/s00705-012-1431-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-012-1431-7