Abstract
Background
Women are over-represented in aSAH cohorts, but whether their outcomes differ to men remains unclear. We examined if sex differences in neurological complications and aneurysm characteristics contributed to aSAH outcomes.
Methods
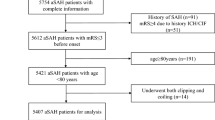
In a retrospective cohort (2010–2016) of all aSAH cases across two hospital networks in Australia, information on severity, aneurysm characteristics and neurological complications (rebleed before/after treatment, postoperative stroke < 48 h, neurological infections, hydrocephalus, seizures, delayed cerebral ischemia [DCI], cerebral infarction) were extracted. We estimated sex differences in (1) complications and aneurysm characteristics using chi square/t-tests and (2) outcome at discharge (home, rehabilitation or death) using multinomial regression with and without propensity score matching on prestroke confounders.
Results
Among 577 cases (69% women, 84% treated) aneurysm size was greater in men than women and DCI more common in women than men. In unadjusted log multinomial regression, women had marginally greater discharge to rehabilitation (RRR 1.15 95% CI 0.90–1.48) and similar likelihood of in-hospital death (RRR 1.02 95% CI 0.76–1.36) versus discharge home. Prestroke confounders (age, hypertension, smoking status) explained greater risk of death in women (rehabilitation RRR 1.13 95% CI 0.87–1.48; death RRR 0.75 95% CI 0.51–1.10). Neurological complications (DCI and hydrocephalus) were covariates explaining some of the greater risk for poor outcomes in women (rehabilitation RRR 0.87 95% CI 0.69–1.11; death RRR 0.80 95% CI 0.52–1.23). Results were consistent in propensity score matched models.
Conclusion
The marginally poorer outcome in women at discharge was partially attributable to prestroke confounders and complications. Improvements in managing complications could improve outcomes.

Similar content being viewed by others
References
Aarhus M, Helland CA, Wester K (2009) Differences in anatomical distribution, gender, and sidedness between ruptured and unruptured intracranial aneurysms in a defined patient population. Acta Neurochir 151:1569. https://doi.org/10.1007/s00701-009-0316-3
Adams H, Ban VS, Leinonen V, Aoun SG, Huttunen J, Saavalainen T, Lindgren A, Frosen J, Fraunberg M, Koivisto T, Hernesniemi J, Welch BG, Jaaskelainen JE, Huttunen TJ (2016) Risk of shunting after aneurysmal subarachnoid hemorrhage. Stroke 47:2488–2496. https://doi.org/10.1161/STROKEAHA.116.013739
Awe OO, Gonzalez LF, Hasan D, Maltenfort M, Rossenwasser R, Jabbour P (2011) Treatment outcome of aneurysmal subarachnoid hemorrhage in patients aged 70 years and older. Neurosurg 68:753–758. https://doi.org/10.1227/NEU.0b013e318207a9fb
Bijlenga P, Gondar R, Schilling S, Morel S, Hirsch S, Cuony J, Corniola M-V, Perren F, Rüfenacht D, Schaller K (2017) PHASES score for the management of intracranial aneurysm: a cross-sectional population-based retrospective study. Stroke 48:2105–2112
Buczacki S, Kirkpatrick P, Seeley H, Hutchinson P (2004) Late epilepsy following open surgery for aneurysmal subarachnoid haemorrhage. J Neurol Neurosurg Psychiatry 75:1620–1622
Budohoski KP, Guilfoyle M, Helmy A, Huuskonen T, Czosnyka M, Kirollos R, Menon DK, Pickard JD, Kirkpatrick PJ (2014) The pathophysiology and treatment of delayed cerebral ischaemia following subarachnoid haemorrhage. J Neurol Neurosurg Psychiatry 85:1343–1353
Cha KC, Kim JH, Kang HI, Moon BG, Lee SJ, Kim JS (2010) Aneurysmal rebleeding: factors associated with clinical outcome in the rebleeding patients. J Korean Neurosurg Soc 47:119
Chotai S, Ahn SY, Moon HJ, Kim JH, Chung HS, Chung YG, Kwon TH (2013) Prediction of outcomes in young adults with aneurysmal subarachnoid hemorrhage. Neurol Med Chir 53:157–162
D’Souza S (2015) Aneurysmal subarachnoid hemorrhage. J Neurosurg Anesthesiol 27:222
De Marchis GM, Schaad C, Fung C, Beck J, Gralla J, Takala J, Jakob SM (2017) Gender-related differences in aneurysmal subarachnoid hemorrhage: a hospital based study. Clin Neurol Neurosurg Clinical Neurology and Neurosurgery 157:82–87. https://doi.org/10.1016/j.clineuro.2017.04.009
Duan W, Pan Y, Wang C, Wang Y, Zhao X, Wang Y, Liu L (2018) Risk factors and clinical impact of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage: analysis from the China National Stroke Registry. Neuroepidemiology 50:128–136. https://doi.org/10.1159/000487325
Duijghuisen JJ, Greebe P, Nieuwkamp DJ, Algra A, Rinkel GJ (2016) Sex-related differences in outcome in patients with aneurysmal subarachnoid hemorrhage. J Stroke and Cerebrovasc Dis 25:2067–2070
Ferguson S, Macdonald RL (2007) Predictors of cerebral infarction in patients with aneurysmal subarachnoid hemorrhage. Neurosurg 60:658–667
Francoeur CL, Mayer SA (2016) Management of delayed cerebral ischemia after subarachnoid hemorrhage. Crit Care 20:277. https://doi.org/10.1186/s13054-016-1447-6
Galea JP, Dulhanty L, Patel HC (2017) Predictors of outcome in aneurysmal subarachnoid hemorrhage patients: observations from a multicenter data set. Stroke 48:2958–2963
Gall S, Donnan G, Dewey H, Macdonell R, Sturm J, Gilligan A, Srikanth V, Thrift A (2010) Sex differences in presentation, severity, and management of stroke in a population-based study. Neurology 74:975–981
Germans MR, Jaja BN, de Oliviera Manoel AL, Cohen AH, Macdonald RL (2018) Sex differences in delayed cerebral ischemia after subarachnoid hemorrhage. J Neurosurg 129:458–464
Ghods AJ, Lopes D, Chen M (2012) Gender differences in cerebral aneurysm location. Front Neurol 3:78
Greenland S (1989) Modeling and variable selection in epidemiologic analysis. Am J Public Health 79:340–349
Hamdan A, Barnes J, Mitchell P (2014) Subarachnoid hemorrhage and the female sex: analysis of risk factors, aneurysm characteristics, and outcomes. J Neurosurg 121:1367–1373
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG (2009) Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 42:377–381. https://doi.org/10.1016/j.jbi.2008.08.010
He Z, Wan Y (2018) Is fetal-type posterior cerebral artery a risk factor for intracranial aneurysm as analyzed by multislice CT angiography? Exp Ther Med 15:838–846
Horikoshi T, Akiyama I, Yamagata Z, Sugita M, Nukui H (2002) Magnetic resonance angiographic evidence of sex-linked variations in the circle of Willis and the occurrence of cerebral aneurysms. J Neurosurg 96:697. https://doi.org/10.3171/jns.2002.96.4.0697
Horiuchi T, Tanaka Y, Hongo K (2006) Sex-related differences in patients treated surgically for aneurysmal subarachnoid hemorrhage. Neurol Med Chir 46:328–332
Kongable GL, Lanzino G, Germanson TP, Truskowski LL, Alves WM, Torner JC, Kassell NF (1996) Gender-related differences in aneurysmal subarachnoid hemorrhage. J Neurosurg 84:43–48
Lin B, Chen W, Ruan L, Chen Y, Zhong M, Zhuge Q, Fan LH, Zhao B, Yang Y (2016) Sex differences in aneurysm morphologies and clinical outcomes in ruptured anterior communicating artery aneurysms: a retrospective study. BMJ Open 6:e009920. https://doi.org/10.1136/bmjopen-2015-009920
Lindbohm JV, Kaprio J, Jousilahti P, Salomaa V, Korja M (2017) Risk factors of sudden death from subarachnoid hemorrhage. Stroke 48:2399–2404. https://doi.org/10.1161/STROKEAHA.117.018118
Lindbohm JV, Kaprio J, Korja M (2019) Survival bias explains improved survival in smokers and hypertensive individuals after aSAH. Neurology 93:e2105–e2109
Nichols L, Stirling C, Otahal P, Stankovich J, Gall S (2018) Socioeconomic disadvantage is associated with a higher incidence of aneurysmal subarachnoid hemorrhage. J Stroke Cerebrovasc Dis 27:660–668. https://doi.org/10.1016/j.jstrokecerebrovasdis.2017.09.055
Nieuwkamp DJ, Vaartjes I, Algra A, Bots ML, Rinkel GJ (2013) Age-and gender-specific time trend in risk of death of patients admitted with aneurysmal subarachnoid hemorrhage in the Netherlands. Int J Stroke 8:90–94
Phan HT, Blizzard CL, Reeves MJ, Thrift AG, Cadilhac D, Sturm J, Heeley E, Otahal P, Konstantinos V, Anderson C (2017) Sex differences in long-term mortality after stroke in the INSTRUCT (INternational STRoke oUtComes sTudy) a meta-analysis of individual participant data. Circ Cardiovasc Qual Outcomes 10:e003436
Rodríguez DR, Matamoros CS, Cúe LF, Hernández JM, Sánchez YP, Nellar JP (2017) Factors associated with poor outcome for aneurysmal subarachnoid haemorrhage in a series of 334 patients. Neurología (English Edition) 32:15–21
Rooij NK, Rinkel GJE, Dankbaar JW, Frijns CJM (2013) Delayed cerebral ischemia after subarachnoid hemorrhage. Stroke 44:43–54. https://doi.org/10.1161/STROKEAHA.112.674291
Rosengart AJ, Schultheiss KE, Tolentino J, Macdonald RL (2007) Prognostic factors for outcome in patients with aneurysmal subarachnoid hemorrhage. Stroke 38:2315–2321
Rosenørn J, Eskesen V, Schmidt K (1993) Clinical features and outcome in females and males with ruptured intracranial saccular aneurysms. Br J Neurosurg 7:287–290
Rowland M, Hadjipavlou G, Kelly M, Westbrook J, Pattinson K (2012) Delayed cerebral ischaemia after subarachnoid haemorrhage: looking beyond vasospasm. Br J Anaesth 109:315–329
Sanelli PC, Anumula N, Gold R, Elias E, Johnson C, Comunale J, Tsiouris AJ, Segal AZ (2012) Outcomes-based assessment of a new reference standard for delayed cerebral ischemia related to vasospasm in aneurysmal subarachnoid hemorrhage. Acad Radiol 19:1066–1074
Sheehan JP, Polin RS, Sheehan JM, Baskaya MK, Kassell NF (1999) Factors associated with hydrocephalus after aneurysmal subarachnoid hemorrhage. Neurosurg 45:1120–1128
Shishido H, Zhang H, Okubo S, Hua Y, Keep RF, Xi G (2016) The effect of gender on acute hydrocephalus after experimental subarachnoid hemorrhage. In: Brain Edema XVI. Springer, pp 335–339
Suarez JI, Sheikh MK, Macdonald RL, Amin-Hanjani S, Brown RD, de Oliveira Manoel AL, Derdeyn CP, Etminan N, Keller E, Leroux PD, Mayer SA, Morita A, Rinkel G, Rufennacht D, Stienen MN, Torner J, Vergouwen MDI, Wong GKC, Suarez JI, Macdonald RL, Amin-Hanjani S, Brown RD, de Oliveira Manoel AL, Derdeyn CP, Etminan N, Keller E, LeRoux PD, Mayer S, Morita A, Rinkel G, Rufennacht D, Stienen MN, Torner J, Vergouwen MDI, Wong GKC, Bijlenga P, Ko N, McDougall CG, Mocco J, Murayama Y, Werner MJH, Broderick J, Dhar R, Jauch EC, Kirkpatrick PJ, Martin RH, Mocco J, Muehlschlegel S, Mutoh T, Nyquist P, Olson D, Mejia-Mantilla JH, van der Jagt M, Bambakidis NC, Brophy G, Bulsara K, Claassen J, Connolly ES, Hoffer SA, Hoh BL, Holloway RG, Kelly A, Nakaji P, Rabinstein A, Vajkoczy P, Woo H, Zipfel GJ, Chou S, Doré S, Dumont AS, Gunel M, Kasuya H, Roederer A, Ruigrok Y, Vespa PM, Sarrafzadeh-Khorrasani AS, Hackenberg K, Huston J, Krings T, Lanzino G, Meyers PM, Wintermark M, Daly J, Ogilvy C, Rhoney DH, Roos YB, Siddiqui A, Algra A, Frösen J, Hasan D, Juvela S, Langer DJ, Salman RA-S, Hanggi D, Schweizer T, Visser-Meily J, Amos L, Ludet C, Moy C, Odenkirchen J, Ala’i S, Esterlitz J, Joseph K, Sheikh M, Aneurysms tUI, Investigators SCP (2019) Common data elements for unruptured intracranial aneurysms and subarachnoid hemorrhage clinical research: a National Institute for Neurological Disorders and Stroke and National Library of Medicine Project. Neurocrit Care 30:4–19. https://doi.org/10.1007/s12028-019-00723-6
Vale FL, Bradley EL, Fisher WS 3rd (1997) The relationship of subarachnoid hemorrhage and the need for postoperative shunting. J Neurosurg 86:462–466. https://doi.org/10.3171/jns.1997.86.3.0462
Vergouwen MD, Ilodigwe D, Macdonald RL (2011) Cerebral infarction after subarachnoid hemorrhage contributes to poor outcome by vasospasm-dependent and-independent effects. Stroke 42:924–929
Wáng YXJ, He J, Zhang L, Li Y, Zhao L, Liu H, Yang L, Zeng XJ, Yang J, Peng GM, Ahuja A, Yang ZH (2016) A higher aneurysmal subarachnoid hemorrhage incidence in women prior to menopause: a retrospective analysis of 4,895 cases from eight hospitals in China. Quant Imaging Med Surg 6:151–156. https://doi.org/10.21037/qims.2016.01.06
Wouter IS, Eelco FMW, David GP, Chu-Pin C, O'Fallon WM, Jack PW (1995) The poor prognosis of ruptured intracranial aneurysms of the posterior circulation. J Neurosurg 82:791–795. https://doi.org/10.3171/jns.1995.82.5.0791
Zhao X, Zhao M, Amin-Hanjani S, Du X, Ruland S, Charbel FT (2015) Wall shear stress in major cerebral arteries as a function of age and gender—a study of 301 healthy volunteers. J Neuroimaging 25:403–407. https://doi.org/10.1111/jon.12133
Zheng K, Zhong M, Zhao B, Chen S-Y, Tan X-X, Li Z-Q, Xiong Y, Duan C-Z (2019) Poor-grade aneurysmal subarachnoid hemorrhage: risk factors affecting clinical outcomes in intracranial aneurysm patients in a multi-center study. Front Neurol 10:123
Acknowledgements
We gratefully thank Gemma Kitsos for creating the database and managing the project, Sue Mosley for data collection in Victoria, and Chao Zhu for statistical help (PhD candidate).
Funding
REDDISH (REducing Delays In aneurysmal Subarachnoid Hemorrhage) was funded by the National Health and Medical Research Council of Australia (NHMRC APP1143155).
Author information
Authors and Affiliations
Contributions
S. Rehman acquired, analysed and interpreted data, drafted the manuscript; R.V. Chandra, K. Smith, N. Thani designed the study, acquired and interpreted data, provided critical intellectual content to draft; K. Zhou and D. Tan acquired and interpreted data, provided critical intellectual content to draft; L. Lai, H. Asadi, M. Reeves, M. Breslin, M. Callisaya interpreted data and provided critical intellectual content to draft; A. Thrift, J. Froelich, L. Nichols, S Gall, L Blizzard and C Stirling designed the study, interpreted data, provided critical intellectual content to draft. All authors approve the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the Human Research Ethics Committee in Victoria (RES-18-0000-036A) and Tasmania (H0014563). For this retrospective study formal consent is not required.
Disclaimer
The sponsor had no role in the design or conduct of this research.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on [Sex Differences in Aneurysmal Subarachnoid Hemorrhage (aSAH): Aneurysm characteristics, Neurological complications, and Outcome]
Electronic supplementary material
ESM 1
(DOCX 46 kb)
Rights and permissions
About this article
Cite this article
Rehman, S., Chandra, R.V., Zhou, K. et al. Sex differences in aneurysmal subarachnoid haemorrhage (aSAH): aneurysm characteristics, neurological complications, and outcome. Acta Neurochir 162, 2271–2282 (2020). https://doi.org/10.1007/s00701-020-04469-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-020-04469-5