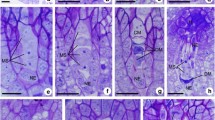
Abstract
Apomixis, the asexual formation of seeds, seems to be a reproductive alternative for many angiosperms, involving various pathways with different genetic and ecological consequences. It is common in some megadiverse tropical groups such as Melastomataceae, of which approximately 70% of the species studied so far in the tribe Miconieae are autonomous apomictics. Hence, Miconia appears to be a good model for the study of the embryological pathways associated with apomixis. In the present study, we analyzed the polyploid and autonomous apomictic M. fallax and compared its embryology to that of the diploid and sexual M. pepericarpa, both treelets species common in the Cerrado, the Neotropical savanna areas of Central Brazil. Ovule structure and basic megasporogenesis and megagametogenesis events were similar in both species. However, M. fallax showed exclusive features associated with apomixis: aposporous embryo sac development, with the parthenogenetic development of unreduced egg cells, autonomous endosperm formation, nucellar embryony and polyembryony. Moreover, both gametophytic and sporophytic apomixis occurred in parallel to the development of a sexual embryo sac, a rarely described condition, which probably confers a great reproductive flexibility to the species.







Similar content being viewed by others
References
Albertini E, Porceddu A, Ferranti F, Reale L, Barcaccia G, Romano B, Falcinelli M (2001) Apospory and parthenogenesis may be uncoupled in Poa pratensis: a cytological investigation. Sexual Pl Reprod 14:213–217. https://doi.org/10.1007/s00497-001-0116-2
Aliyu OM, Schranz ME, Sharbel TF (2010) Quantitative variation for apomictic reproduction in the genus Boechera (Brassicaceae). Amer J Bot 97:1719–1731. https://doi.org/10.3732/ajb.1000188
Alves ER, Carneiro VTC, Araújo ACG (2001) Direct evidence of pseudogamy in apomictic Brachiaria brizantha (Poaceae). Sexual Pl Reprod 14:207–212. https://doi.org/10.1007/s00497-001-0120-6
Asker SE, Jerling L (1992) Apomixis in plants. CRC Press, Boca Raton
Batygina TB, Vinogradova GY (2007) Phenomenon of polyembryony. Genetic heterogeneity of seeds. Russ J Developm Biol 38:126–151. https://doi.org/10.1134/S1062360407030022
Bicknell RA, Koltunow AM (2004) Understanding apomixis: recent advances and remaining conundrums. Pl Cell 16:228–245. https://doi.org/10.1105/tpc.017921
Bicknell RA, Lambie SC, Butler RC (2003) Quantification of progeny classes in two facultatively apomictic accessions of Hieracium. Hereditas 138:11–20. https://doi.org/10.1034/j.1601-5223.2003.01624.x
Bittencourt NS Jr, Semir J (2005) Late-acting self-incompatibility and other breeding systems in Tabebuia (Bignoniaceae). Int J Pl Sci 166:493–506. https://doi.org/10.1086/428758
Borges HBN (1991) Biologia reprodutiva de quatro espécies de Melastomataceae. MSc Thesis, Universidade Estadual de Campinas, Campinas
Caetano APS, Simão DG, Carmo-Oliveira R, Oliveira PE (2013a) Diplospory and obligate apomixis in Miconia albicans (Miconieae, Melastomataceae) and an embryological comparison with its sexual congener M. chamissois. Pl Syst Evol 299:1253–1262. https://doi.org/10.1007/s00606-013-0793-y
Caetano APS, Teixeira SP, Forni-Martins ER, Carmello-Guerreiro SM (2013b) Pollen insights into apomictic and sexual Miconia (Miconieae, Melastomataceae). Int J Pl Sci 174:760–768. https://doi.org/10.1086/669927
Caetano APS, Basso-Alves J, Cortez PA, Brito VLG, Michelangeli FA, Reginato M, Goldenberg R, Carmello-Guerreiro SM, Teixeira SP (2017) Evolution of the outer ovule integument and its systematic significance in Melastomataceae. Bot J Linn Soc. https://doi.org/10.1093/botlinnean/box093
Cappuccino N (2004) Allee effect in an invasive alien plant, pale swallow-wort Vincetoxicum rossicum (Asclepiadaceae). Oikos 106:3–8. https://doi.org/10.1111/j.0030-1299.2004.12863.x
Carman JG (1997) Asynchronous expression of duplicate genes in angiosperms may cause apomixis, bispory, tetraspory, and polyembryony. Biol J Linn Soc 61:51–94. https://doi.org/10.1111/j.1095-8312.1997.tb01778.x
Cooper DC, Brink RA (1949) The endosperm-embryo relationship in an autonomous apomict, Taraxacum officinale. Bot Gaz 111:139–153. https://doi.org/10.1086/335582
Cortez PA, Carmello-Guerreiro SM (2008) Ontogeny and structure of the pericarp and the seed coat of Miconia albicans (Sw.) Triana (Melastomataceae) from “cerrado”Brazil. Revista Brasil Bot 31:71–79. https://doi.org/10.1590/S0100-84042008000100008
Cortez PA, Carmello-Guerreiro SM, Teixeira SP (2012) Understanding male sterility in Miconia species (Melastomataceae): a morphological approach. Austral J Bot 60:506–516. https://doi.org/10.1071/BT12076
Costa ME, Sampaio DS, Paoli AAS, Leite SCAL (2004) Poliembrionia e aspectos da embriogênese em Tabebuia ochraceae (Chamisso) Standley (Bignoniaceae). Revista Brasil Bot 27:395–406. https://doi.org/10.1590/S0100-84042004000200017
Crane CF (2001) Classification of apomictic mechanisms. In: Savidan Y, Carman JG, Dresselhaus T (eds) The flowering of apomixis: from mechanisms to genetic engineering. CIMMYT, IRD, European Commission DG VI, Mexico, pp 24–43
Davis GL (1966) Systematic embryology of the angiosperms. Wiley, New York
Galla G, Barcaccia G, Schallau A, Puente Molins M, Bäumlein H, Sharbel TF (2011) The cytohistological basis of apospory in Hypericum perforatum L. Sexual Pl Reprod 24:47–61. https://doi.org/10.1007/s00497-010-0147-7
Goldenberg R (2009) Miconia Ruiz and Pav. In: Wanderley MGL, Shepherd GJ, Melhem TS, Giulietti AM, Martins SE (eds) Flora fanerogâmica do estado de São Paulo, vol. 6. Fapesp, São Paulo, pp 73–103
Goldenberg R, Shepherd GJ (1998) Studies on the reproductive biology of Melastomataceae in “cerrado” vegetation. Pl Syst Evol 211:13–29. https://doi.org/10.1007/BF00984909
Goldenberg R, Varassin IG (2001) Sistemas reprodutivos de espécies de Melastomataceae da Serra do Japi, Jundiaí, São Paulo, Brasil. Revista Brasil Bot 24:283–288. https://doi.org/10.1590/S0100-84042001000300006
Grimanelli D, Hernández M, Perotti E, Savidan Y (1997) Dosage effects in the endosperm of diplosporous apomictic Tripsacum (Poaceae). Sexual Pl Reprod 10:279–282. https://doi.org/10.1007/s004970050098
Guan L, Chen L, Terao H (2006) Ultrastructural studies of gametophytic apomicts in Guinea grass (Panicum maximum) I. Differentiation of aposporous initial cell. Cytologia 71:379–389. https://doi.org/10.1508/cytologia.71.379
Hand ML, Koltunow AM (2014) The genetic control of apomixis: asexual seed formation. Genetics 197:441–450. https://doi.org/10.1534/genetics.114.163105
Hojsgaard D, Hörandl E (2015) A little bit of sex matters for genome evolution in asexual plants. Frontiers Pl Sci 6:82. https://doi.org/10.3389/fpls.2015.00082
Hojsgaard D, Klatt S, Baier R, Carman JG, Hörandl E (2014) Taxonomy and biogeography of apomixis in angiosperms and associated biodiversity characteristics. Crit Rev Pl Sci 33:414–427. https://doi.org/10.1080/07352689.2014.898488
Hörandl E, Cosendai A, Temsch EM (2008) Understanding the geographic distributions of apomictic plants: a case for a pluralistic approach. Pl Ecol Diversity 1:309–320. https://doi.org/10.1080/17550870802351175
Hotchkiss EE, DiTommaso A, Brainard DC, Mohler CL (2008) Survival and performance of the invasive vine Vincetoxicum rossicum (Apocynaceae) from seeds of different embryo number under two light environments. Amer J Bot 95:447–453. https://doi.org/10.3732/ajb.95.4.447
Johansen DA (1940) Plant microtechnique. McGraw-Hill Book Company, New York
Johri BM, Ambegaokar KB, Srivastava PS (1992) Comparative embryology of angiosperms, vol. 2. Springer, Berlin
Koltunow AM (1993) Apomixis: embryo sacs and embryo formed without meiosis or fertilization in ovules. Pl Cell 5:1425–1437. https://doi.org/10.1105/tpc.5.10.1425
Koltunow AM, Grossniklaus U (2003) Apomixis: a developmental perspective. Annual Rev Pl Biol 54:547–574. https://doi.org/10.1146/annurev.arplant.54.110901.160842
Koltunow AM, Johnson SD, Bicknell RA (1998) Sexual and apomictic development in Hieracium. Sexual Pl Reprod 11:213–230. https://doi.org/10.1007/s004970050144
Koltunow AM, Johnson SD, Bicknell RA (2000) Apomixis is not developmentally conserved in related, genetically characterized Hieracium plants of varying ploidy. Sexual Pl Reprod 12:253–266. https://doi.org/10.1007/s004970050193
Ladd D, Cappuccino N (2005) A field study of seed dispersal and seedling performance in the invasive exotic vine Vincetoxicum rossicum. Canad J Bot 83:1181–1188. https://doi.org/10.1139/b05-093
Maia FR, Varassin IG, Goldenberg R (2016) Apomixis does not affect visitation to flowers of Melastomataceae, but pollen sterility does. Pl Biol 18:132–138. https://doi.org/10.1111/plb.12364
Martin FN (1959) Staining and observing pollen tubes in the style by means of fluorescence. Stain Technol 34:125–128. https://doi.org/10.3109/10520295909114663
Martins RL, Oliveira PE (2003) RAPD evidence for apomixis and clonal populations in Eriotheca (Bombacaceae). Pl Biol 5:338–340. https://doi.org/10.1055/s-2003-40792
Mazzucato A, Falcinelli M, Veronesi F (1996) Evolution and adaptedness in a facultatively apomictic grass, Poa pratensis L. Euphytica 92:13–19. https://doi.org/10.1007/BF00022823
McDowell EM, Trump B (1976) Histological fixatives for diagnostic light and electron microscopy. Arch Pathol Lab Med 100:405–414
Medeiros JD, Morretes BL (1996) The embryology of Miconia cabucu (Melastomataceae). Cytologia 61:83–91. https://doi.org/10.1508/cytologia.61.83
Mendes-da-Glória FJ, Mourão Filho FAA, Appezzato-da-Glória B (2001) Morfologia de embriões nucelares de laranja ‘Valência’ (Citrus sinensis (L.) Osbeck). Acta Bot Brasil 15:17–25. https://doi.org/10.1590/S0102-33062001000100003
Mendes-Rodrigues C, Oliveira PE (2012) Polyembryony in Melastomataceae from Brazilian Cerrado: multiple embryos in a small world. Pl Biol 14:845–853. https://doi.org/10.1111/j.1438-8677.2011.00551.x
Mendes-Rodrigues C, Carmo-Oliveira R, Tavalera S, Arista M, Ortiz PL, Oliveira PE (2005) Polyembryony and apomixis in Eriotheca pubescens (Malvaceae-Bombacoideae). Pl Biol 7:533–550. https://doi.org/10.1055/s-2005-865852
Mendes-Rodrigues C, Ranal MA, Oliveira PE (2011) Does polyembryony reduce seed germination and seedling development in Eriotheca pubescens (Malvaceae: Bombacoideae)? Amer J Bot 98:1–10. https://doi.org/10.3732/ajb.1100022
Mendes-Rodrigues C, Sampaio DS, Costa ME, Caetano APS, Ranal MA, Bittencourt NS Jr, Oliveira PE (2012) Polyembryony increases embryo and seedling mortality but also enhances seed individual survival in Handroanthus species (Bignoniaceae). Flora 207:264–274. https://doi.org/10.1016/j.flora.2011.10.008
Naumova TN (1993) Apomixis in angiosperms: nucellar and integumentar embryony. CRC Press, Boca Raton
Naumova TN (1997) Apomixis in tropical fodder crops: cytological and functional aspects. Euphytica 96:93–99. https://doi.org/10.1023/A:1002909110354
Naumova TN (2008) Apomixis and amphimixis in flowering plants. Cytol Genet 42:179–188. https://doi.org/10.3103/S0095452708030055
Naumova TN, Vielle-Calzada JP (2001) Ultrastructural analysis of apomictic development. In: Savidan Y, Carman JG, Dresselhaus T (eds) The flowering of apomixis: from mechanisms to genetic engineering. CIMMYT, IRD, European Commission DG VI, Mexico, pp 44–63
Naumova TN, Willemse MTM (1995) Ultrastructural characterization of apospory in Panicum maximum. Sexual Pl Reprod 8:197–204. https://doi.org/10.1007/BF00228937
Nogler GA (1984) Gametophytic apomixis. In: Johri BM (ed) Embryology of Angiosperms. Springer, Berlin, pp 475–518
Noyes RD (2007) Apomixis in the asteraceae: diamonds in the rough. Funct Pl Sci Biotechnol 1:207–222
Noyes RD, Givens AD (2013) Quantitative assessment of megasporogenesis for the facultative apomicts Erigeron annuus and Erigeron strigosus (Asteraceae). Int J Pl Sci 174:1239–1250. https://doi.org/10.1086/673243
O’Brien TP, Feder N, McCully ME (1964) Polychromatic staining of plant cell walls by toluidine Blue O. Protoplasma 59:367–373. https://doi.org/10.1007/BF01248568
Quarin CL (1999) Effect of pollen source and pollen ploidy on endosperm formation and seed set in pseudogamous apomictic Paspalum notatum. Sexual Pl Reprod 11:331–335. https://doi.org/10.1007/s004970050160
Renner SS (1989) A survey of reproductive biology in Neotropical Melastomataceae and Memecylaceae. Ann Missouri Bot Gard 76:496–518. https://doi.org/10.2307/2399497
Ribeiro RC, Oliveira DMT, Silveira FAO (2015) A new seed coat water-impermeability mechanism in Chaetostoma armatum (Melastomataceae): evolutionary and biogeographical implications of physiophysical dormancy. Seed Sci Res 25:194–202. https://doi.org/10.1017/S0960258515000070
Richards AJ (1997) Plant breeding systems. Chapman and Hall, London
Rodrigo JM, Zappacosta DC, Selva JP, Garbus I, Albertini E, Echenique V (2017) Apomixis frequency under stress conditions in weeping lovegrass (Eragrostis curvula). PLoS ONE 12:e0175852. https://doi.org/10.1371/journal.pone.0175852
Rutishauser A (1982) Introducción a la embriología y biología de la reproducción de las angiospermas. Hemisferio Sur, Buenos Aires
Santos APM, Fracasso CM, Santos ML, Romero R, Sazima M, Oliveira PE (2012) Reproductive biology and species geographical distribution in the Melastomataceae: a survey based on New World taxa. Ann Bot (Oxford) 110:667–679. https://doi.org/10.1093/aob/mcs125
Sartor ME, Quarin CL, Urbani MH, Espinoza F (2011) Ploidy levels and reproductive behaviour in natural populations of five Paspalum species. Pl Syst Evol 293:31–41. https://doi.org/10.1007/s00606-011-0416-4
Subramanyan K (1944) A contribution on the life-history of Sonerila wallichii Benn. Proc Indian Acad Sci B 19:115–120. https://doi.org/10.1007/BF03049776
Tobe H (1989) The embryology of angiosperms: its broad application to the systematic and evolutionary study. Bot Mag 102:351–367. https://doi.org/10.1007/BF02488572
Tobe H, Raven PH (1983) An embryological analysis of Myrtales, its definition and characteristics. Ann Missouri Bot Gard 70:71–94. https://doi.org/10.2307/2399008
Tucker MR, Araujo AG, Paech NA, Hecht V, Schmidt EDL, Rossell J, de Vries SC, Koltunow AMG (2003) Sexual and apomictic reproduction in Hieracium subgenus Pilosella are closely interrelated developmental pathways. Pl Cell 15:1524–1537. https://doi.org/10.1105/tpc.011742
Van Dijk PJ (2003) Ecological and evolutionary opportunities of apomixis: insights from Taraxacum and Chondrilla. Phil Trans Roy Soc London B 358:1113–1121. https://doi.org/10.1098/rstb.2003.1302
Wen XS, Ye XL, Li YQ, Chen ZL, Xu SX (1998) Embryological studies on apomixis in Pennisetum squamulatum. Acta Bot Sin 40:598–604
Whitton J, Sears CJ, Baack EJ, Otto SP (2008) The dynamic nature of apomixis in the angiosperms. Int J Pl Sci 169:169–182. https://doi.org/10.1086/523369
Yao J, Zhou Y, Hu C (2007) Apomixis in Eulaliopsis binata: characterization of reproductive mode and endosperm development. Sexual Pl Reprod 20:151–158. https://doi.org/10.1007/s00497-007-0051-y
Acknowledgements
This work is part of the Master’s dissertation of the first author. We thank the São Paulo Research Foundation (FAPESP-Fundação de Amparo à Pesquisa do Estado de São Paulo) for the financial support (Grant Numbers 2008/10793-0 and 2010/15077-0) and J.Y. Tamashiro for help with the fieldwork. We also thank Fabian Michelangeli and two anonymous reviewers for their helpful comments. P.E. Oliveira and S.P. Teixeira are funded by CNPq fellowships (Grant Numbers 306551/2014-4 and 303493/2015-1, respectively). The final organization of the manuscript was carried out under a CAPES-PNPD Postdoctoral grant for the first author.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Hans de Jong.
Rights and permissions
About this article
Cite this article
Caetano, A.P.S., Cortez, P.A., Teixeira, S.P. et al. Unusual diversity of apomictic mechanisms in a species of Miconia, Melastomataceae. Plant Syst Evol 304, 343–355 (2018). https://doi.org/10.1007/s00606-017-1480-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00606-017-1480-1