Abstract
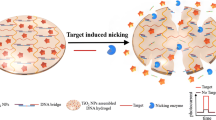
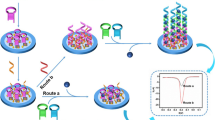
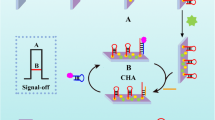
A novel photoelectrochemical (PEC) biosensor based on an enzyme-free nucleic acid dual-amplification strategy combined with a mimic enzyme to catalyze the deposition of a quencher is reported for the ultrasensitive detection of miRNA-21. A limited amount of target miRNA-21 can trigger the formation of long DNA duplexes on the electrode, owing to the synergistic effect of the enzyme-free nucleic acid dual-amplification strategy of entropy-driven strand displacement reaction (ESDR) amplification and hybridization chain reaction (HCR) amplification. The embedded manganese porphyrin (MnPP) in the long DNA duplexes acts as a horseradish peroxidase (HRP)–mimicking enzyme to catalyze the transformation of benzo-4-chlorohexadienone on the electrode surface, resulting in a significant reduction in photocurrent intensity. As a photosensitive material, BiOCl-BiOI is used as a tag to provide strong initial PEC signals. Based on the cascade integration of the enzyme-free nucleic acid dual-amplification strategy and the mimic enzyme-catalyzed precipitation reaction, the current PEC biosensor exhibits outstanding performance for miRNA-21 detection with an ultralow detection limit (33 aM) and a wide quantification range (from 100 aM to 1 nM). This work provides a new avenue toward the ultrasensitive detection of miRNAs, and is expected to be used for clinical and biochemical samples.
Graphical abstract
A unique PEC biosensor with the BiOCl-BiOI composite, as the photosensitive material, has been developed for ultrasensitive miRNA-21 determination based on the combination of an enzyme-free nucleic acid dual-amplification strategy and mimic enzyme catalytic precipitation reaction.



taken from BiOCl-BiOI composites





Similar content being viewed by others
References
Ma W, Fu P, Sun M, Xu L, Kuang H, Xu C (2017) Dual quantification of microRNAs and telomerase in living cells. J Am Chem Soc 139:11752–11759
Wang H, Tang H, Yang C, Li Y (2019) Selective single molecule nanopore sensing of microRNA using PNA functionalized magnetic core–shell Fe3O4–Au nanoparticles. Anal Chem 91:7965–7970
Calin GA, Croce CM (2006) MicroRNA signatures in human cancers. Nat Rev Cancer 6:857–866
Dong H, Lei J, Ding L, Wen Y, Ju H, Zhang X (2013) microRNA: function, detection, and bioanalysis. Chem Rev 113:6207–6233
Yi J-T, Chen T-T, Huo J, Chu X (2017) Nanoscale zeolitic imidazolate framework-8 for ratiometric fluorescence imaging of microRNA in living cells. Anal Chem 89:12351–12359
Greco F, Inferrera A, La Rocca R, Navarra M, Casciaro M, Grosso G, Gangemi S, Ficarra V, Mirone V (2019) The potential role of microRNAs as biomarkers in benign prostatic hyperplasia: a systematic review and meta-analysis. Eur Urol Focus 5:497–507
Zhao T, Zhang N, Zhang Y, Ren J, Xu P, Liu Z, Cheng L, Hu Y (2017) A novel method to identify pre-microRNA in various species knowledge base on various species. J Biomedical Semantics 8:30
Bejerano T, Etzion S, Elyagon S, Etzion Y, Cohen S (2018) Nanoparticle delivery of miRNA-21 mimic to cardiac macrophages improves myocardial remodeling after myocardial infarction. Nano Lett 18:5885–5891
Li M-X, Zhao W, Wang H, Li X-L, Xu C-H, Chen H-Y, Xu J-J (2018) Dynamic single molecular rulers: toward quantitative detection of microRNA-21 in living cells. Anal Chem 90:14255–14259
Zhang P, Lin Z, Zhuo Y, Yuan R, Chai Y (2017) Dual microRNAs-fueled DNA nanogears: a case of regenerated strategy for multiple electrochemiluminescence detection of microRNAs with single luminophore. Anal Chem 89:1338–1345
Xia Y, Zhang R, Wang Z, Tian J, Chen X (2017) Recent advances in high-performance fluorescent and bioluminescent RNA imaging probes. Chem Soc Rev 46:2824–2843
van Rooij E, Sutherland Lillian B, Qi X, Richardson James A, Hill J, Olson Eric N (2007) Control of stress-dependent cardiac growth and gene expression by a microRNA. Science 316:575–579
Zhao W-W, Xu J-J, Chen H-Y (2014) Photoelectrochemical DNA biosensors. Chem Rev 114:7421–7441
Haddour N, Chauvin J, Gondran C, Cosnier S (2006) Photoelectrochemical immunosensor for label-free detection and quantification of anti-cholera toxin antibody. J Am Chem Soc 128:9693–9698
Wen G, Ju H (2016) Enhanced photoelectrochemical proximity assay for highly selective protein detection in biological matrixes. Anal Chem 88:8339–8345
Hu Q, Han D, Gan S, Bao Y, Niu L (2018) Surface-initiated-reversible-addition–fragmentation-chain-transfer polymerization for electrochemical DNA biosensing. Anal Chem 90:12207–12213
Ge L, Wang W, Li F (2017) Electro-grafted electrode with graphene-oxide-like DNA affinity for ratiometric homogeneous electrochemical biosensing of microRNA. Anal Chem 89:11560–11567
Zhang Y, Chai Y, Wang H, Yuan R (2019) Target-induced 3d DNA network structure as a novel signal amplifier for ultrasensitive electrochemiluminescence detection of microRNAs. Anal Chem 91:14368–14374
Chen P, Wu P, Zhang Y, Chen J, Jiang X, Zheng C, Hou X (2016) Strand displacement-induced enzyme-free amplification for label-free and separation-free ultrasensitive atomic fluorescence spectrometric detection of nucleic acids and proteins. Anal Chem 88:12386–12392
Zhang N, Shi X-M, Guo H-Q, Zhao X-Z, Zhao W-W, Xu J-J, Chen H-Y (2018) Gold nanoparticle couples with entropy-driven toehold-mediated DNA strand displacement reaction on magnetic beads: toward ultrasensitive energy-transfer-based photoelectrochemical detection of miRNA-141 in Real Blood Sample. Anal Chem 90:11892–11898
Xia L-Y, Li M-J, Wang H-J, Yuan R, Chai Y-Q (2020) Novel single-enzyme-assisted dual recycle amplification strategy for sensitive photoelectrochemical microRNA assay. Anal Chem 92:14550–14557
Huang L, Zhang L, Yang L, Yuan R, Yuan Y (2018) Manganese porphyrin decorated on DNA networks as quencher and mimicking enzyme for construction of ultrasensitive photoelectrochemistry Aptasensor. Biosens Bioelectron 104:21–26
Kong W, Xiang M-H, Xia L, Zhang M, Kong R-M, Qu F (2020) In-situ synthesis of 3D Cu2O@Cu-based Mof nanobelt arrays with improved conductivity for sensitive photoelectrochemical detection of vascular endothelial growth factor 165. Biosens Bioelectron 167:112481
Li L, Zhang Y, Yan Z, Chen M, Zhang L, Zhao P, Yu J (2020) Ultrasensitive photoelectrochemical detection of microRNA on paper by combining a cascade nanozyme-engineered biocatalytic precipitation reaction and target-triggerable DNA motor. ACS Sensors 5:1482–1490
Sun L, Xiang L, Zhao X, Jia C-J, Yang J, Jin Z, Cheng X, Fan W (2015) Enhanced visible-light photocatalytic activity of BIOI/BIOCl heterojunctions: key role of crystal facet combination. ACS Catal 5:3540–3551
Su X, Yang J, Yu X, Zhu Y, Zhang Y (2018) In situ grown hierarchical 50%Biocl/Bioi hollow flowerlike microspheres on reduced graphene oxide nanosheets for enhanced visible-light photocatalytic degradation of rhodamine B. Appl Surf Sci 433:502–512
Yi W, Cai R, Xiang D, Wang Y, Zhang M, Ma Q, Cui Y, Bian X (2019) A novel photoelectrochemical strategy based on an integrative photoactive heterojunction nanomaterial and a redox cycling amplification system for ultrasensitive determination of microRNA in cells. Biosens Bioelectron 143:111614
Gao X, Niu S, Ge J, Luan Q, Jie G (2020) 3D DNA nanosphere-based photoelectrochemical biosensor combined with multiple enzyme-free amplification for ultrasensitive detection of cancer biomarkers. Biosens Bioelectron 147:111778
Chu Y, Deng A-P, Wang W, Zhu J-J (2019) Concatenated catalytic hairpin assembly/hyperbranched hybridization chain reaction based enzyme-free signal amplification for the sensitive photoelectrochemical detection of human telomerase RNA. Anal Chem 91:3619–3627
Li H, Wang J, Wang X, Lin H, Li F (2019) Perylene-based photoactive material as a double-stranded DNA intercalating probe for ultrasensitive photoelectrochemical biosensing. ACS Appl Mater Interfaces 11:16958–16964
Yuan Y, Hu T, Zhong X, Zhu M, Chai Y, Yuan R (2020) Highly sensitive photoelectrochemical biosensor based on quantum dots sensitizing Bi2te3 nanosheets and DNA-amplifying strategies. ACS Appl Mater Interfaces 12:22624–22629
Liu P, Yang X, Sun S, Wang Q, Wang K, Huang J, Liu J, He L (2013) Enzyme-free colorimetric detection of DNA by using gold nanoparticles and hybridization chain reaction amplification. Anal Chem 85:7689–7695
Chu Y, Wu R, Fan G-C, Deng A-P, Zhu J-J (2018) Enzyme-free photoelectrochemical biosensor based on the co-sensitization effect coupled with dual cascade toehold-mediated strand displacement amplification for the sensitive detection of microRNA-21. ACS Sustain Chem Eng 6:11633–11641
Li S-K, Liu Z-T, Li J-Y, Chen A-Y, Chai Y-Q, Yuan R, Zhuo Y (2018) Enzyme-free target recycling and double-output amplification system for electrochemiluminescent assay of mucin 1 with MoS2 nanoflowers as co-reaction accelerator. ACS Appl Mater Interfaces 10:14483–14490
Li J, Weng X, Mo F, Han M, Li H (2020) Superparamagnetic nanostructures coupled with an entropy-driven DNA circuit for elegant and robust photoelectrochemical biosensing. Anal Chem 92:15145–15151
Feng Q-M, Guo Y-H, Xu J-J, Chen H-Y (2017) Self-assembled DNA tetrahedral scaffolds for the construction of electrochemiluminescence biosensor with programmable DNA cyclic amplification. ACS Appl Mater Interfaces 9:17637–17644
Long D, Li M, Wang H, Wang H, Chai Y, Li Z, Yuan R (2020) Ultrasensitive photoelectrochemical assay for DNA detection based on a novel SnS2/Co3O4 sensitized structure. Anal Chem 92:14769–14774
Li M, Tian X, Liang W, Yuan R, Chai Y (2018) Ultrasensitive photoelectrochemical assay with PtB7-Th/CdTe quantum dots sensitized structure as signal tag and benzo-4-chlorohexadienone precipitate as efficient quencher. Anal Chem 90:14521–14526
Xie S, Dong Y, Yuan Y, Chai Y, Yuan R (2016) Ultrasensitive lipopolysaccharides detection based on doxorubicin conjugated N-(aminobutyl)-N-(ethylisoluminol) as electrochemiluminescence indicator and self-assembled tetrahedron DNA dendrimers as nanocarriers. Anal Chem 88:5218–5224
Li M, Xiong C, Zheng Y, Liang W, Yuan R, Chai Y (2018) Ultrasensitive photoelectrochemical biosensor based on DNA tetrahedron as nanocarrier for efficient immobilization of CdTe Qds-methylene blue as signal probe with near-zero background noise. Anal Chem 90:8211–8216
Ye S, Li X, Wang M, Tang B (2017) Fluorescence and SERS imaging for the simultaneous absolute quantification of multiple miRNAs in living cells. Anal Chem 89:5124–5130
Si ML, Zhu S, Wu H, Lu Z, Wu F, Mo YY (2007) MIR-21-mediated tumor growth. Oncogene 26:2799–2803
Funding
This work was financially supported by the National Natural Science Foundation of China (22064010, 51862014, 21775089, 22074080), Changjiang Scholar Program of the Ministry of Education of China (Q2019258), and the Taishan Scholar Program of Shandong Province (tsqn201909106).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
604_2022_5345_MOESM1_ESM.docx
Supplementary file1 Materials and reagents, DNA and RNA sequence, optimization of experimental conditions, mechanism of the electron transformation, and additional data. (DOCX 286 KB)
Rights and permissions
About this article
Cite this article
Zhao, Y., Li, X., Xiang, MH. et al. Enzyme-free nucleic acid dual-amplification strategy combined with mimic enzyme catalytic precipitation reaction for the photoelectrochemical detection of microRNA-21. Microchim Acta 189, 249 (2022). https://doi.org/10.1007/s00604-022-05345-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-022-05345-y