Abstract
A nonenzymatic electrochemical sensor is described for the prostate cancer biomarker sarcosine (Sar). Riboflavin was employed to mimic the active center of the enzyme sarcosine oxidase for constructing the biomimetic sensor. The use of riboflavon (Rf) avoids the disadvantages of an enzymatic sensor, such as high cost and poor stability. A glassy carbon electrode (GCE) was modified with a graphene-chitosan (GR) composite and further modified with gold-platinum bimetallic nanoparticles in a polypyrrole (PPy) matrix in order to enhance the catalytic activity of the enzyme mimic. Finally, Rf was electrodeposited on the surface of the AuPt-PPy/GR-modified GCE. Under optimized conditions, the GCE provided high sensitivity and selectivity for Sar at around 0.61 V. Response covers the 2.5–600 μM concentration range, and the detection limit is 0.68 μM. The method was successfully applied to the determination of Sar in spiked urine with 98.0%–103.2% recovery.

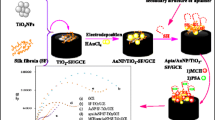
Schematic presentation of the fabrication of the Rf/AuPt-PPy/GR/GCE surface and the measurement principle by differential pulse voltammetry (DPV).






Similar content being viewed by others
References
Reis STD, Jose PJ, Antunes AA, Sousa-Canavez JM, Abe DK, Cruz JAS, Dall’ogllio MF, Crippa A, Passerotti CC, Ribeiro-Filho LA, Viana NI, Srouqi M, Leite KRM (2011) Tgf-β1 expression as a biomarker of poor prognosis in prostate cancer. Clinics 66:1143–1147
Bostrom PJ, Soloway MS (2007) Secondary cancer after radiotherapy for prostate cancer: should we be more aware of the risk? Eur Urol 52:973–982
Oesterling JE (1991) Prostate specific antigen: a critical assessment of the most useful tumor marker for adenocarcinoma of the prostate. J Urol 145:907–923
Leman ES, Getzenberg RH (2009) Biomarkers for prostate cancer. J Cell Biochem 108:3–9
Draisma G, Boer R, Otto SJ, van der Cruijsen IW, Damhuis RAM, Schröder FH, de Koning HJ (2003) Lead times and overdetection due to prostate-specific antigen screening: estimates from the European randomized study of screening for prostate cancer. J Natl Cancer Inst 95:868–878
Etzioni R, Falcon S, Gann PH, Kooperberg CL, Penson DF, Stampfer MJ (2004) Prostate-specific antigen and free prostate-specific antigen in the early detection of prostate cancer: do combination tests improve detection? Cancer Epidemiol Biomark Prev 13:1640–1645
Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J, Laxman B, Mehra R, Lonigro RJ, Li Y, Nyati MK, Ahsan A, Kalyana-Sundaram S, Han B, Cao X, Byun J, Omenn GS, Ghosh D, Pennathur S, Alexander DC, Berger A, Shuster JR, Wei JT, Varambally S, Beecher C, Chinnaiyan AM (2009) Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature 457:910–914
Xue Z, Yin B, Wang H, Li M, Rao H, Liu X, Zhou X, Lu X (2016) An organic indicator functionalized graphene oxide nanocomposite-based colorimetric assay for the detection of sarcosine. Nanoscale 8:5488–5496
Lan J, Xu W, Wan Q, Zhang X, Lin J, Chen J, Chen J (2014) Colorimetric determination of sarcosine in urine samples of prostatic carcinoma by mimic enzyme palladium nanoparticles. Anal Chim Acta 825:63–68
Hamid HM, Mohammad H (2015) Nonderivatized sarcosine analysis by gas chromatography after solid-phase microextraction by newly synthesized monolithic molecularly imprinted polymer. Chromatographia 78:1263–1270
Chen J, Zhang J, Zhang W, Chen Z (2014) Sensitive determination of the potential biomarker sarcosine for prostate cancer by LC-MS with N,N′- dicyclohexylcarbodiimide derivatization. J Sep Sci 37:14–19
Meyer TE, Fox SD, Issaq HJ, Xu X, Chu LW, Veenstra TD, Hsing AW (2011) A reproducible and high-throughput HPLC/MS method to separate sarcosine from α- and β-alanine and to quantify sarcosine in human serum and urine. Anal Chem 83:5735–5740
Cernei N, Zitka O, Ryvolova M, Adam V, Masarik M, Hubalek J, Kizek R (2012) Spectrometric and electrochemical analysis of sarcosine as a potential prostate carcinoma marker. Int J Electrochem Sci 7:4286–4301
Yang P, Wang J, Yang C, Zhao X, Xie S, Ge Z (2018) Nano Pt@ZIF8 modified electrode and its application to detect sarcosine. J Electrochem Soc 165:H247–H250
Zhou Y, Yin H, Meng X, Xu Z, Fu Y, Ai S (2012) Direct electrochemistry of sarcosine oxidase on graphene, chitosan and silver nanoparticles modified glassy carbon electrode and its biosensing for hydrogen peroxide. Electrochim Acta 71:294–301
Rebelo TS, Pereira CM, Sales MG, Noronha JP, Costa-Rodrigues J, Silva F, Fernandes MH (2014) Sarcosine oxidase composite screen-printed electrode for sarcosine determination in biological samples. Anal Chim Acta 850:26–32
Xue Z, Wang H, Rao H, He N, Wang X, Liu X, Lu X (2017) Amperometric indicator displacement assay for biomarker monitoring: indirectly sensing strategy for electrochemically inactive sarcosine. Talanta 167:666–671
Nguy TP, Phi TV, Tram DTN, Eersels K, Wagner P, Lien TTN (2017) Development of an impedimetric sensor for the label-free detection of the amino acid sarcosine with molecularly imprinted polymer receptors. Sensors Actuators B Chem 246:461–470
Scognamiglio V, Antonacci A, Lambreva MD, Litescu SC, Rea G (2015) Synthetic biology and biomimetic chemistry as converging technologies fostering a new generation of smart biosensors. Biosens Bioelectron 74:1076–1086
Kuah E, Toh S, Yee J, Ma Q, Gao Z (2016) Enzyme mimics: advances and applications. Chemistry 22:8404–8430
Hassan-Abdallah A, Zhao G, Jorns MS (2008) Covalent flavinylation of monomeric sarcosine oxidase: identification of a residue essential for holoenzyme biosynthesis. Biochemistry 47:1136–1143
Trickey P, Wagner MA, Jorns MS, Mathews FS (1999) Monomeric sarcosine oxidase: structure of a covalently flavinylated amine oxidizing enzyme. Structure 7:331–345
Zhao G, Jorns MS (2005) Ionization of Zwitterionic amine substrates bound to monomeric sarcosine oxidase. Biochemistry 44:16866–16874
Cao X, Wang N, Jia S, Guo L, Li K (2013) Bimetallic AuPt nanochains: synthesis and their application in electrochemical immunosensor for the detection of carcinoembryonic antigen. Biosens Bioelectron 39:226–230
Ahuja T, Mir IA, Kumar D (2007) Biomolecular immobilization on conducting polymers for biosensing applications. Biomaterials 28:791–805
Lee JW, Serna F, Schmidt CE (2006) Carboxy-endcapped conductive polypyrrole: biomimetic conducting polymer for cell scaffolds and electrodes. Langmuir 22:9816–9819
Lin M, Hu X, Ma Z, Chen L (2012) Functionalized polypyrrole nanotube arrays as electrochemical biosensor for the determination of copper ions. Anal Chim Acta 746:63–69
Hu W, Li CM, Dong H (2008) Poly(pyrrole-co-pyrrole propylic acid) film and its application in label-free surface plasmon resonance immunosensors. Anal Chim Acta 630:67–74
Yeo BS, Bell AT (2011) Enhanced activity of gold-supported cobalt oxide for the electrochemical evolution of oxygen. J Am Chem Soc 133:5587–5593
Wang W, Gao Y, Jia X, Xi K (2013) A novel Au-Pt@PPy(polypyrrole) coral-like structure: facile synthesis, high SERS effect, and good electro catalytic activity. J Colloid Interface Sci 396:23–28
Daniel MC, Astruc D (2004) Gold nanoparticles: assembly, supramolecular chemistry, quantum-size-related properties, and applications toward biology, catalysis, and nanotechnology. Chem Rev 104:293–346
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant No. 81573678) and Natural Science Foundation of Guangdong Province, China (Grant No. 2015A030313584).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
We declare that we have no competing interests. And informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOC 7571 kb)
Rights and permissions
About this article
Cite this article
Liu, T., Fu, B., Chen, J. et al. An electrochemical sarcosine sensor based on biomimetic recognition. Microchim Acta 186, 136 (2019). https://doi.org/10.1007/s00604-019-3240-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-019-3240-0