Abstract
Purpose
Earlier studies have investigated the suitability of various materials and autologous grafts for the repair of diaphragmatic defects. Our group investigated the feasibility of using an artificial diaphragm (AD) to repair wide diaphragmatic defects.
Methods
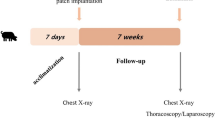
Twelve pigs were laparotomized and, in each pig, a defect was fashioned by resecting a round 8-cm diameter hole in the left diaphragm. Next, the defect was repaired by implanting an AD. The animals were relaparotomized 8 or 24 weeks after implantation for gross, histological and radiological observation of the implanted sites.
Results
All recipient animals survived until killing for evaluation. Chest X-ray examinations showed no differences between the preoperative diaphragms and the grafted diaphragms at 8 and 24 weeks after implantation. At 8 weeks after implantation, the implanted sites exhibited fibrous adhesions to the liver and lungs without deformities or penetrations. Parts of the surface tissue at the graft sites had a varnished appearance similar to those of the native diaphragm. Histology performed at 8 weeks detected no trace of the ADs in the graft sites; however, numerous inflammatory cells and profuse fibrous connective tissue were observed. At 24 weeks after implantation, no differences were found in the thorax between the areas with the grafts and the unaffected areas. Histology of the graft sites in the thorax confirmed growth of mesothelial cells similar to that observed in the native diaphragm.
Conclusions
Artificial diaphragms can be a novel substitute for diaphragmatic repair.








Similar content being viewed by others
References
Chao H, Liu C, Chen C, et al. Congenital diaphragmatic hernia in the neonatal period: review of 21 years’ experience. Pediatr Neonatol. 2010;51:97–102.
Lally P, Paranka MS, Roden J, Georgeson KE, Wilson JM, Lillehei CW, et al. Congenital diaphragmatic hernia. Stabilization and repair on ECMO. Ann Surg. 1992;216:569–73.
Crandall M, Popowich D, Shapiro M, West M. Posttraumatic hernias: historical overview and review of the literature. Am Surg. 2007;73:845–50.
Rocco G, Rendina EA, Meroni A, Venuta F, Della Pona C, De Giacomo T, et al. Prognostic factors after surgical treatment of lung cancer invading the diaphragm. Ann Thorac Surg. 1999;68:2065–8.
Sugarbaker DJ, Mentzer SJ, Strauss G. Extrapleural pneumonectomy in the treatment of malignant pleural mesothelioma. Ann Thorac Surg. 1992;54:941–6.
Geisler F, Gotlieb A, Fried D. Agenesis of the right diaphragm: repaired with marlex. J Pediatr Surg. 1977;12:587–8.
Kuhn R, Schubert D, St, Marusch F, Lippert H, Pross M. Repair of diaphragmatic rupture by laparoscopic implantation of a polytetrafluoroethylene patch. Surg Endosc. 2002;16:1495.
Menezes, Chagas, Macedo-Neto, Santos, Rocco, Zin. Suture or prosthetic reconstruction of experimental diaphragmatic defects. Chest. 2000;117:1443–8.
Rocco G, Rendina EA. Prosthetic reconstruction of the resected diaphragm: the way to go. Chest. 2001;119:671.
Lantis JC, Gallivan EK, Hekier R, Connolly R, Schwaitzberg SD, Crombleholme T. A comparison of collagen and PTFE patch repair in a rabbit model of congenital diaphragmatic hernia. J Invest Surg. 2000;13:319–25.
Tatum RP, Shalhub S, Oelschlager BK, Pellegrini CA. Complications of PTFE mesh at the diaphragmatic hiatus. J Gastrointest Surg. 2008;12:953–7.
Schneider R, Herrington JL, Granda AM. Marlex mesh in repair of a diaphragmatic defect later eroding into the distal esophagus and stomach. Am Surg. 1979;45:337–9.
Barbosa RF, Rodrigues J, Correia-Pinto J, Costa-Ferreira A, Cardoso A, Reis JC, et al. Repair of a large congenital diaphragmatic defect with a reverse latissimus dorsi muscle flap. Microsurgery. 2008;28:85–8.
Bedini AV, Andreani SM, Muscolino G. Latissimus dorsi reverse flap to substitute the diaphragm after extrapleural pneumonectomy. Ann Thorac Surg. 2000;69:986–8.
Kageyama Y, Suzuki K, Matsushita K, Takahashi T, Kazui T. Diaphragm reconstruction with autologous fascia lata: report of a case. Surg Today. 1999;29:1285–6.
Suzuki K, Takahashi T, Itou Y, Asai K, Shimota H, Kazui T. Reconstruction of diaphragm using autologous fascia lata: an experimental study in dogs. Ann Thorac Surg. 2002;74:209–12.
Sydorak RM, Hoffman W, Lee H, Yingling CD, Longaker M, Chang J, et al. Reversed latissimus dorsi muscle flap for repair of recurrent congenital diaphragmatic hernia. J Pediatr Surg. 2003;38:296–300. (Discussion 296–300).
Dalla Vecchia L, Engum S, Kogon B, Jensen E, Davis M, Grosfeld J. Evaluation of small intestine submucosa and acellular dermis as diaphragmatic prostheses. J Pediatr Surg. 1999;34:167–71.
Sandoval JA, Lou D, Engum SA, Fisher LM, Bouchard CM, Davis MM, et al. The whole truth: comparative analysis of diaphragmatic hernia repair using 4-ply vs 8-ply small intestinal submucosa in a growing animal model. J Pediatr Surg. 2006;41:518–23.
Koot VC, Bergmeijer JH, Molenaar JC. Lyophylized dura patch repair of congenital diaphragmatic hernia: occurrence of relapses. J Pediatr Surg. 1993;28:667–8.
Fauza DO, Marler JJ, Koka R, Forse RA, Mayer JE, Vacanti JP. Fetal tissue engineering: diaphragmatic replacement. J Pediatr Surg. 2001;36:146–51.
Kunisaki SM, Fuchs JR, Kaviani A, Oh JT, LaVan DA, Vacanti JP, et al. Diaphragmatic repair through fetal tissue engineering: a comparison between mesenchymal amniocyte- and myoblast-based constructs. J Pediatr Surg. 2006;41:34–9. Discussion 9.
Toshimitsu Miyazawa. Ikada. Tissue-engineered patch for the reconstruction of inferior vena cava during living-donor liver transplantation. J Gastrointest Surg. 2005;9:789–93.
Toshimitsu Y, Miyazawa M, Torii T, Koyama I, Ikada Y. Tissue-engineered patch for the reconstruction of inferior vena cava during living-donor liver transplantation. J Gastrointest Surg. 2005;9:789–93.
Beck DE, Cohen Z, Fleshman JW, Kaufman HS, van Goor H, Wolff BG. A prospective, randomized, multicenter, controlled study of the safety of Seprafilm adhesion barrier in abdominopelvic surgery of the intestine. Dis Colon Rectum. 2003;46:1310–9.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aikawa, M., Miyazawa, M., Okamoto, K. et al. A newly designed bioabsorbable substitute for the treatment of diaphragmatic defects. Surg Today 43, 1298–1304 (2013). https://doi.org/10.1007/s00595-012-0414-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-012-0414-3