Abstract
Aims
To determine the fasting plasma glucose (FPG) level at which an oral glucose tolerance test (OGTT) could be avoided to screen for gestational diabetes (GDM) and to evaluate the characteristics of women across this FPG threshold.
Methods
A multi-centric prospective cohort study with 1843 women receiving universal screening for GDM with a 75 g OGTT.
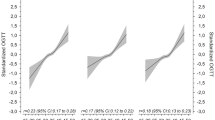
Results
In the total population, GDM prevalence was 12.5% (231). A FPG < 78 mg/dL was the cut-off with best trade-off to limit the number of missed GDM cases [44 (19.0%)] with a negative predictive value of 97.3% (95% CI 96.5–98.0) for GDM, while avoiding 52.2% OGTTs. Compared to GDM with FPG ≥ 78 mg/dL [187 (81.0%)], GDM women with FPG < 78 mg/dL had a significantly lower BMI (27.1 ± 4.5 vs. 29.6 ± 5.2 kg/m2, p = 0.003), less insulin resistance [Matsuda: 0.4 (0.4–0.7) vs. 0.3 (0.2–0.5), p < 0.001] and better β-cell function [ISSI-2: 0.13 (0.08–0.25) vs. 0.09 (0.04–0.15), p = 0.004]. Compared to NGT women (1612) with FPG ≥ 78 mg/dL [846 (52.5%)], NGT with FPG < 78 mg/dL [766 (47.5%)] had a significantly lower BMI (26.0 ± 3.9 vs. 27.8 ± 4.7 kg/m2, p < 0.001), less insulin resistance [Matsuda: 0.7 (0.5–0.9) vs. 0.5 (0.4–0.7), p < 0.001], better β-cell function [ISSI-2: 0.17 (0.10–0.30) vs. 0.12 (0.07–0.21), p < 0.001], and less often large-for-gestational age infants [9.2 (70) vs. 16.2% (136), p < 0.001].
Conclusions
FPG < 78 mg/dL can be used to limit the number of OGTTs when screening for GDM. Women with FPG < 78 mg/dL had a better metabolic profile and in NGT women also less fetal overgrowth.

Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
American Diabetes Association (2021) Addendum. 2. Classification and diagnosis of diabetes: standards of medical care in diabetes. Diabetes Care 44(Suppl. 1):S15–S33. https://doi.org/10.2337/dc21-ad09
Crowther CA, Hiller JE, Moss JR et al (2005) Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. N Engl J Med 352(24):2477–2486. https://doi.org/10.1056/NEJMoa042973
Landon MB, Spong CY, Thom E et al (2009) A multicenter randomized trial of treatment for mild gestational diabetes. N Engl J Med 361(14):1339–48. https://doi.org/10.1056/NEJMoa0902430
Benhalima K, Mathieu C, Damm P et al (2015) A proposal for the use of uniform diagnostic criteria for gestational diabetes in Europe: an opinion paper by the European Board & College of Obstetrics and Gynaecology (EBCOG). Diabetologia 58(7):1422–9. https://doi.org/10.1007/s00125-015-3615-7
O’Sullivan JB, Mahan CM (1964) Criteria for the oral glucose tolerance test in pregnancy. Diabetes 13:278–285
Carpenter MW, Coustan DR (1982) Criteria for screening tests for gestational diabetes. Am J Obstet Gynecol 144(7):768–773. https://doi.org/10.1016/0002-9378(82)90349-0
Metzger BE, Buchanan TA, Coustan DR et al (2007) Summary and recommendations of the fifth international workshop-conference on gestational diabetes mellitus. Diabetes Care 30(Suppl 2):S251-60. https://doi.org/10.2337/dc07-s225
Metzger BE et al (2010) International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 33(3):676–682. https://doi.org/10.2337/dc09-1848
Metzger BE, Contreras M, Sacks DA et al (2008) Hyperglycemia and adverse pregnancy outcomes. N Engl J Med 358(19):1991–2002. https://doi.org/10.1056/NEJMoa0707943
Agarwal MM, Punnose J, Dhatt GS (2004) Gestational diabetes: problems associated with the oral glucose tolerance test. Diabetes Res Clin Pract 63(1):73–74. https://doi.org/10.1016/j.diabres.2003.08.005
Benhalima K, Van Crombrugge P, Moyson C et al (2018) A Modified two-step screening strategy for gestational diabetes mellitus based on the 2013 WHO criteria by combining the glucose challenge test and clinical risk factors. J Clin Med 7(10):351. https://doi.org/10.3390/jcm7100351
Agarwal MM (2016) Gestational diabetes mellitus: Screening with fasting plasma glucose. World J Diabetes 7(14):279–289. https://doi.org/10.4239/wjd.v7.i14.279
Benhalima K, Mathieu C, Van Assche A et al (2016) Survey by the European Board and College of Obstetrics and Gynaecology on screening for gestational diabetes in Europe. Eur J Obstet Gynecol Reprod Biol 201:197–202. https://doi.org/10.1016/j.ejogrb.2016.04.003
Li M, Lan JR, Liang JL, Xiong XL (2020) Diagnostic accuracy of fasting plasma glucose as a screening test for gestational diabetes mellitus: a systematic review and meta-analysis. Eur Rev Med Pharmacol Sci 24(21):11172–11186. https://doi.org/10.26355/eurrev_202011_23605
Benhalima K, Van Crombrugge P, Verhaeghe J et al (2014) The Belgian diabetes in pregnancy study (BEDIP-N), a multi-centric prospective cohort study on screening for diabetes in pregnancy and gestational diabetes: methodology and design. BMC Pregnancy Childbirth 14:226. https://doi.org/10.1186/1471-2393-14-226
Benhalima K, Van Crombrugge P, Moyson C et al (2018) The sensitivity and specificity of the glucose challenge test in a universal two-step screening strategy for gestational diabetes mellitus using the 2013 world health organization criteria. Diabetes Care 41(7):e111–e112. https://doi.org/10.2337/dc18-0556
Benhalima K, Van Crombrugge P, Moyson C et al (2019) Characteristics and pregnancy outcomes across gestational diabetes mellitus subtypes based on insulin resistance. Diabetologia 62(11):2118–2128. https://doi.org/10.1007/s00125-019-4961-7
American Diabetes Association (2013) Standards of medical care in diabetes-2013. Diabetes Care 36(Suppl 1):S11-66. https://doi.org/10.2337/dc13-S011
Durán A, Martín P, Runkle I et al (2010) Benefits of self-monitoring blood glucose in the management of new-onset Type 2 diabetes mellitus: the St Carlos Study, a prospective randomized clinic-based interventional study with parallel groups. J Diabetes 2(3):203–211. https://doi.org/10.1111/j.1753-0407.2010.00081.x
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC (1985) Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28(7):412–419. https://doi.org/10.1007/BF00280883
Matsuda M, DeFronzo RA (1999) Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care 22(9):1462–1470. https://doi.org/10.2337/diacare.22.9.1462
Kahn SE (2003) The relative contributions of insulin resistance and beta-cell dysfunction to the pathophysiology of Type 2 diabetes. Diabetologia 46(1):3–19. https://doi.org/10.1007/s00125-002-1009-0
Kirwan JP, Huston-Presley L, Kalhan SC, Catalano PM (2001) Clinically useful estimates of insulin sensitivity during pregnancy: validation studies in women with normal glucose tolerance and gestational diabetes mellitus. Diabetes Care 24(9):1602–1607. https://doi.org/10.2337/diacare.24.9.1602
Retnakaran R, Qi Y, Goran MI, Hamilton JK (2009) Evaluation of proposed oral disposition index measures in relation to the actual disposition index. Diabet Med 26(12):1198–1203. https://doi.org/10.1111/j.1464-5491.2009.02841.x
Devlieger H, Martens G, Bekaert A, Eeckels R (2000) Standaarden van geboortegewicht-voor-zwangerschapsduur voor de vlaamse boreling. Tijdschrift voor geneeskunde 56(1):1–14
IOM (Institute of Medicine) and NRC (National Research Council) (2009) Weight Gain During Pregnancy: Reexamining the Guidelines. Washington, DC.
Saeedi M, Hanson U, Simmons D, Fadl H (2018) Characteristics of different risk factors and fasting plasma glucose for identifying GDM when using IADPSG criteria: a cross-sectional study. BMC Pregnancy Childbirth 18(1):225. https://doi.org/10.1186/s12884-018-1875-1
d’Emden M, McLeod D, Ungerer J, Appleton C, Kanowski D (2020) Development of a fasting blood glucose-based strategy to diagnose women with gestational diabetes mellitus at increased risk of adverse outcomes in a COVID-19 environment. PLoS ONE 15(12):e0243192. https://doi.org/10.1371/journal.pone.0243192
Perucchini D, Fischer U, Spinas GA, Huch R, Huch A, Lehmann R (1999) Using fasting plasma glucose concentrations to screen for gestational diabetes mellitus: prospective population based study. BMJ 319(7213):812–815. https://doi.org/10.1136/bmj.319.7213.812
Agarwal MM, Dhatt GS, Shah SM (2010) Gestational diabetes mellitus: simplifying the international association of diabetes and pregnancy diagnostic algorithm using fasting plasma glucose. Diabetes Care 33(9):2018–2020. https://doi.org/10.2337/dc10-0572
Agarwal MM, Hughes PF, Punnose J, Ezimokhai M (2000) Fasting plasma glucose as a screening test for gestational diabetes in a multi-ethnic, high-risk population. Diabet Med 17(10):720–726. https://doi.org/10.1046/j.1464-5491.2000.00371.x
Agarwal MM, Dhatt GS, Punnose J (2006) Gestational diabetes: utility of fasting plasma glucose as a screening test depends on the diagnostic criteria. Diabet Med 23(12):1319–1326. https://doi.org/10.1111/j.1464-5491.2006.01987.x
Agarwal MM, Dhatt GS, Punnose J, Koster G (2005) Gestational diabetes in a high-risk population: using the fasting plasma glucose to simplify the diagnostic algorithm. Eur J Obstet Gynecol Reprod Biol 120(1):39–44. https://doi.org/10.1016/j.ejogrb.2004.07.034
Pintaudi B, Di Vieste G, Corrado F et al (2014) Improvement of selective screening strategy for gestational diabetes through a more accurate definition of high-risk groups. Eur J Endocrinol 170(1):87–93. https://doi.org/10.1530/EJE-13-0759
Balaji V, Balaji M, Anjalakshi C, Cynthia A, Arthi T, Seshiah V (2011) Inadequacy of fasting plasma glucose to diagnose gestational diabetes mellitus in Asian Indian women. Diabetes Res Clin Pract 94(1):e21–e23. https://doi.org/10.1016/j.diabres.2011.07.008
Juutinen J, Hartikainen AL, Bloigu R, Tapanainen JS (2000) A retrospective study on 435 women with gestational diabetes: fasting plasma glucose is not sensitive enough for screening but predicts a need for insulin treatment. Diabetes Care 23(12):1858–1859. https://doi.org/10.2337/diacare.23.12.1858
Maegawa Y, Sugiyama T, Kusaka H, Mitao M, Toyoda N (2003) Screening tests for gestational diabetes in Japan in the 1st and 2nd trimester of pregnancy. Diabetes Res Clin Pract 62(1):47–53. https://doi.org/10.1016/s0168-8227(03)00146-3
McIntyre HD, Gibbons KS, Ma RCW et al (2020) Testing for gestational diabetes during the COVID-19 pandemic. An evaluation of proposed protocols for the United Kingdom, Canada and Australia. Diabetes Res Clin Pract 167:108353. https://doi.org/10.1016/j.diabres.2020.108353
McIntyre HD, Moses RG (2020) The diagnosis and management of gestational diabetes mellitus in the context of the COVID-19 pandemic. Diabetes Care 43(7):1433–1434. https://doi.org/10.2337/dci20-0026
Pintaudi B, Fresa R, Dalfrà M et al (2018) The risk stratification of adverse neonatal outcomes in women with gestational diabetes (STRONG) study. Acta Diabetologia 55(12):1261–1273. https://doi.org/10.1007/s00592-018-1208-x
Acknowledgments
KB (Katrien Benhalima) and RD are the recipient of a ‘Fundamenteel Klinisch Navorserschap FWO Vlaanderen’.
Funding
This investigator-initiated study was funded by the Belgian National Lottery, the Fund of the Academic studies of UZ Leuven, and the Fund Yvonne and Jacques François-de Meurs of the King Baudouin Foundation. KB (Katrien Benhalima) and RD are the recipient of a ‘Fundamenteel Klinisch Navorserschap FWO Vlaanderen’.
Author information
Authors and Affiliations
Contributions
KB (Katrien Benhalima), PVC, and CM (Chantal Mathieu) conceived the project. CM (Carolien Moyson), KDW, and AE prepared the data and AL did the statistical analysis. KB (Kaat Beunen) did the literature review. KB (Kaat Beunen) and KB (Katrien Benhalima) wrote the first draft of the manuscript. All authors contributed to the study design, including data collection, data interpretation, and manuscript revision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study protocol was approved by the Institutional Review Boards of all participating centers. All investigations have been carried out in accordance with the principles of the Declaration of Helsinki as revised in 2008.
Consent to participate
All participants provided informed consent prior to inclusion in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article belongs to the topical collection Pregnancy and Diabetes, managed by Antonio Secchi and Marina Scavini.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Beunen, K., Neys, A., Van Crombrugge, P. et al. Fasting plasma glucose level to guide the need for an OGTT to screen for gestational diabetes mellitus. Acta Diabetol 59, 381–394 (2022). https://doi.org/10.1007/s00592-021-01812-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-021-01812-9