Abstract
Purpose
Osteochondral defects have a limited capacity to heal and can evolve to an early osteoarthritis. A surgical possibility is the replacement of the affected cartilaginous area with a resurfacing device BioPoly™ RS Partial Resurfacing Knee Implant. The aim of this study was to report the clinical and survival outcomes of the BioPoly™ after a minimum follow-up of 4 years.
Methods
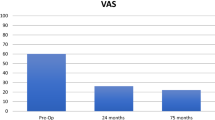
This study included all patients who had a BioPoly™ for femoral osteochondral defects greater than 1 cm2 and at least ICRS grade 2. The main outcome was to observe the KOOS and the Tegner activity score were used to assess outcomes preoperatively and at the last follow-up. The secondary outcomes were the VAS for pain, the complications rate post-surgery and survival rate of BioPoly™ at the last FU.
Results
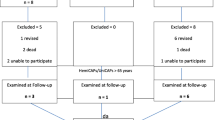
Eighteen patients with 44.4% (8/18) of women were included with a mean age of 46.6 years (11.4), a mean body mass index (BMI) of 21.5 (kg/m2) (2.3). The mean follow-up was 6.3 years (1.3). We found a significant difference comparing pre-operative KOOS score and at last follow-up [respectively, 66.56(14.37) vs 84.17(7.656), p < 0.01]. At last follow-up, the Tegner score was different [respectively, 3.05(1.3) vs 3.6(1.3), p < 0.01]. At 5 years, the survival rate was of 94.7%.
Conclusions
BioPoly™ is a real alternative for femoral osteochondral defects greater than 1 cm2 and at least ICRS grade 2. It will be interesting to compare this implant to mosaicplasty technic and/or microfracture at 5 years postoperatively regarding clinical outcomes and survival rate.
Level of evidence
Therapeutic level III. Prospective cohort study.




Similar content being viewed by others
References
Curl WW, Krome J, Gordon ES et al (1997) Cartilage injuries: a review of 31,516 knee arthroscopies. Arthroscopy 13:456–460. https://doi.org/10.1016/s0749-8063(97)90124-9
Årøen A, Løken S, Heir S et al (2004) Articular cartilage lesions in 993 consecutive knee arthroscopies. Am J Sports Med 32:211–215. https://doi.org/10.1177/0363546503259345
Widuchowski W, Widuchowski J, Trzaska T (2007) Articular cartilage defects: study of 25,124 knee arthroscopies. Knee 14:177–182. https://doi.org/10.1016/j.knee.2007.02.001
Buckwalter JA (1998) Articular cartilage: injuries and potential for healing. J Orthop Sports Phys Ther 28:192–202. https://doi.org/10.2519/jospt.1998.28.4.192
Mankin HJ (1982) The response of articular cartilage to mechanical injury. J Bone Joint Surg Am 64:460–466
Buckwalter JA, Mankin HJ, Grodzinsky AJ (2005) Articular cartilage and osteoarthritis. Instr Course Lect 54:465–480
Shelbourne KD, Jari S, Gray T (2003) Outcome of untreated traumatic articular cartilage defects of the knee: a natural history study. J Bone Joint Surg Am 85-A(Suppl 2):8–16. https://doi.org/10.2106/00004623-200300002-00002
Guccione AA, Felson DT, Anderson JJ et al (1994) The effects of specific medical conditions on the functional limitations of elders in the Framingham Study. Am J Public Health 84:351–358. https://doi.org/10.2105/ajph.84.3.351
Hunter DJ, Schofield D, Callander E (2014) The individual and socioeconomic impact of osteoarthritis. Nat Rev Rheumatol 10:437–441. https://doi.org/10.1038/nrrheum.2014.44
Steadman J, Rodkey W, Rodrigo J (2001) Microfracture: surgical technique and rehabilitation to treat chondral defects. Clin Orthop Relat Res 391:S362–S369
Julin J, Jämsen E, Puolakka T et al (2010) Younger age increases the risk of early prosthesis failure following primary total knee replacement for osteoarthritis. A follow-up study of 32,019 total knee replacements in the Finnish Arthroplasty Register. Acta Orthop 81:413–419. https://doi.org/10.3109/17453674.2010.501747
Rand JA, Trousdale RT, Ilstrup DM, Harmsen WS (2003) Factors affecting the durability of primary total knee prostheses. J Bone Joint Surg Am 85:259–265. https://doi.org/10.2106/00004623-200302000-00012
Campbell J, Lescun T, Conner N et al An UHMWPE-hyaluronan microcomposite material for partial joint resurfacing in a goat model
Nathwani D, McNicholas M, Hart A et al (2017) Partial resurfacing of the knee with the BioPoly implant: interim report at 2 years. JBJS Open Access 2:10011. https://doi.org/10.2106/JBJS.OA.16.00011
Mainil-Varlet P, Aigner T, Brittberg M et al (2003) Histological assessment of cartilage repair: a report by the Histology Endpoint Committee of the International Cartilage Repair Society (ICRS). J Bone Joint Surg Am 85-A(Suppl 2):45–57
Roos EM, Toksvig-Larsen S (2003) Knee injury and Osteoarthritis Outcome Score (KOOS)—validation and comparison to the WOMAC in total knee replacement. Health Qual Life Outcomes 1:17. https://doi.org/10.1186/1477-7525-1-17
Tegner Y, Lysholm J (1985) Rating systems in the evaluation of knee ligament injuries. Clin Orthopaed Relat Res 198:42–49
Hangody L, Füles P (2003) Autologous osteochondral mosaicplasty for the treatment of full-thickness defects of weight-bearing joints: ten years of experimental and clinical experience. JBJS 85:25–32
Lynch TS, Patel RM, Benedick A et al (2015) Systematic review of autogenous osteochondral transplant outcomes. Arthroscopy 31:746–754. https://doi.org/10.1016/j.arthro.2014.11.018
Ollat D, Lebel B, Thaunat M et al (2011) Mosaic osteochondral transplantations in the knee joint, midterm results of the SFA multicenter study. Orthop Traumatol Surg Res 97:S160–S166. https://doi.org/10.1016/j.otsr.2011.08.005
Solheim E, Hegna J, Inderhaug E (2017) Long-term clinical follow-up of microfracture versus mosaicplasty in articular cartilage defects of medial femoral condyle. Knee 24:1402–1407. https://doi.org/10.1016/j.knee.2017.08.061
Andrade R, Vasta S, Pereira R et al (2016) Knee donor-site morbidity after mosaicplasty—a systematic review. J Exp Orthop 3:31. https://doi.org/10.1186/s40634-016-0066-0
Solheim E, Hegna J, Inderhaug E (2020) Long-term survival after microfracture and mosaicplasty for knee articular cartilage repair: a comparative study between two treatments cohorts. Cartilage 11:71–76. https://doi.org/10.1177/1947603518783482
Riboh JC, Cvetanovich GL, Cole BJ, Yanke AB (2017) Comparative efficacy of cartilage repair procedures in the knee: a network meta-analysis. Knee Surg Sports Traumatol Arthrosc 25:3786–3799. https://doi.org/10.1007/s00167-016-4300-1
Armoiry X, Cummins E, Connock M et al (2019) Autologous chondrocyte implantation with Chondrosphere for treating articular cartilage defects in the knee: an evidence review group perspective of a NICE single technology appraisal. Pharmacoeconomics 37:879–886
Zamborsky R, Danisovic L (2020) Surgical techniques for knee cartilage repair: an updated large-scale systematic review and network meta-analysis of randomized controlled trials. Arthrosc J Arthrosc Relat Surg 36:845–858. https://doi.org/10.1016/j.arthro.2019.11.096
Laursen JO, Backer Mogensen C, Skjøt-Arkil H (2021) HemiCAP knee implants: mid- to long-term results. Cartilage 13:1718S-1725S. https://doi.org/10.1177/1947603519894732
Nahas S, Monem M, Li L et al (2020) Ten-year average full follow-up and evaluation of a contoured focal resurface prosthesis (HemiCAP) in patients in the United Kingdom. J Knee Surg 33:966–970. https://doi.org/10.1055/s-0039-1688921
Acknowledgements
None.
Funding
There is no funding source.
Author information
Authors and Affiliations
Contributions
P-AB contributed to article design, data analysis and article writing. VF contributed to data collection and article writing. EK contributed to data collection and article writing. FPE contributed to surgeon, article design and article writing.
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest.
Ethical approval
This study was approved by the research ethics committee of Maussins Clinics under the reference N° Maus-2020-11-01.
Informed consent
All patients gave their consent to participate to the study. All authors gave their consent to publish the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
A video of the surgical technique. After performing a medial parapatellar approach, a pilot pin is inserted at the centre of the osteochondral defect. The second stage is to determine the size of the implant. This is done by a size trial over the defect, ensuring complete coverage. The defect must be shouldered on all sides by normal cartilage. A reamer is then used to prepare the site of the implant. The shoulder is drilled until the reamer makes contact with the top of the cannula. The ideal placement is for the top of the trial to be 0.5 mm recessed below the articulating cartilage. The implant is then inserted carefully by aligning the stem with the previously created pilot hole (MP4 193050 kb)
Appendix 1
Appendix 1
N° | Side | IMC | Gender | Age | Sport level | Biopoly size (mm) | Defect localization | Bony allograft | Meniscal lesion | Prior surgery | Diagnosis |
|---|---|---|---|---|---|---|---|---|---|---|---|
1 | Right | 18.0 | F | 65 | Sedentary | 15 | Lateral condyle | No | No | No | Osteonecrosis |
2 | Right | 19.0 | F | 52 | Occasional | 15 | Lateral condyle | No | No | No | Post-traumatic lesion |
3 | Right | 18.7 | F | 36 | Occasional | 15 | Medial condyle | No | No | No | Post-traumatic lesion |
4 | Left | 22.1 | M | 36 | Occasional | 15 × 25 | Medial condyle | Yes | No | No | Sequel of osteochondritis |
5 | Left | 20.5 | F | 34 | Occasional | 15 × 25 | Medial condyle | No | No | No | Sequel of osteochondritis |
6 | Left | 19.8 | M | 40 | Sedentary | 15 | Medial condyle | No | No | No | Osteonecrosis |
7 | Right | 23.0 | M | 61 | Sedentary | 15 | Medial condyle | Yes | No | No | Osteonecrosis |
8 | Right | 24.8 | M | 55 | Occasional | 15 | Medial condyle | No | No | No | Post-traumatic lesion |
9 | Left | 21.0 | M | 46 | Occasional | 15 | Medial condyle | No | No | No | Post-traumatic lesion |
10 | Left | 24.7 | M | 30 | Occasional | 15 × 25 | Medial condyle | No | No | No | Sequel of osteochondritis |
11 | Right | 24.7 | F | 56 | Sedentary | 15 | Lateral condyle | No | No | No | Post-traumatic lesion |
12 | Right | 19.6 | M | 34 | Occasional | 15 | Lateral condyle | No | No | No | Post-traumatic lesion |
13 | Right | 22.8 | F | 56 | Competition | 15 | Lateral condyle | No | No | No | Post-traumatic lesion |
14 | Right | 19.8 | F | 47 | Sedentary | 15 | Lateral condyle | No | No | No | Osteonecrosis |
15 | Left | 22.9 | M | 29 | Occasional | 15 | Lateral condyle | No | No | No | Osteonecrosis |
16 | Right | 25.0 | M | 56 | Occasional | 15 | Medial condyle | No | No | No | Osteonecrosis |
17 | Left | 19.0 | F | 58 | Sedentary | 15 | Lateral condyle | No | No | No | Osteonecrosis |
18 | Right | 20.7 | M | 47 | Sedentary | 15 | Medial condyle | No | No | No | Post-traumatic lesion |
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bouché, PA., Fiodière, V., Kierszbaum, E. et al. Clinical evaluation of a resurfacing device implant for femoral osteochondral defects greater than 1 cm2 with a minimal follow-up of 4 years: a prospective cohort study. Eur J Orthop Surg Traumatol 33, 3693–3701 (2023). https://doi.org/10.1007/s00590-023-03613-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00590-023-03613-y