Abstract
Purpose
The ovine model is often used to evaluate new spine fusion technologies prior to clinical testing. An important aspect of designing sheep surgery protocols is to select the appropriate postoperative time period for comparing fusion outcomes. Unfortunately, determining the ideal study endpoint is complicated by the fact that prior published studies have not used consistent timeframes. Thus, the primary aim of this study was to provide a reference for investigators as to the expected fusion outcomes of control groups at varying timepoints in sheep spine surgery models.
Methods
We identified published sheep fusion studies using autograft, interbody cages, and/or instrumentation. Fusion data were extracted, converted to a common scale, and analyzed across studied timepoints.
Results
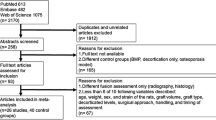
Overall, 29 studies of 360 fusion levels were identified: 11 ALIF (158 levels), 3 PLIF/TLIF (28 levels), 8 PLF (90 levels), and 7 ACDF (84 levels). Studied timepoints ranged from 4 to 48 weeks postoperative. In general, fusion rates varied across techniques and instrumentation. The time to reach solid fusion differed by as many as 20 weeks between control groups.
Conclusions
Recommended timeframes for future studies designed to show either superiority over controls or equivalent outcomes with controls were developed based on aggregate results. Designating ideal study endpoints for sheep fusion models has both ethical implications associated with responsible use of animals in research, and economic implications given the cost of animal research. The current results can guide the development of future research methods and help investigators choose appropriate study timelines for various control groups.








Similar content being viewed by others
References
Sheng SR, Wang XY, Xu HZ, Zhu GQ, Zhou YF (2010) Anatomy of large animal spines and its comparison to the human spine: a systematic review. Eur Spine J 19:46–56
Patel VV (unpublished data)
Gu Y, Chen L, Yang HL, Luo ZP, Tang TS (2011) Evaluation of an injectable silk fibroin enhanced calcium phosphate cement loaded with human recombinant bone morphogenetic protein-2 in ovine lumbar interbody fusion. J Biomed Mater Res A 97:177–185
Qian Y, Lin Z, Chen J, Fan Y, Davey T, Cake M, Day R, Dai K, Xu J, Zheng M (2009) Natural bone collagen scaffold combined with autologous enriched bone marrow cells for induction of osteogenesis in an ovine spinal fusion model. Tissue Eng Part A 15:3547–3558
Steffen T, Stoll T, Arvinte T, Schenk RK (2001) Porous tricalcium phosphate and transforming growth factor used for anterior spine surgery. Eur Spine J 10(Suppl 2):S132–S140
Sandhu HS, Toth JM, Diwan AD, Seim HB, 3rd, Kanim LE, Kabo JM, Turner AS (2002) Histologic evaluation of the efficacy of rhBMP-2 compared with autograft bone in sheep spinal anterior interbody fusion. Spine (Phila Pa 1976) 27:567–575
Toth JM, Seim HB, 3rd, Schwardt JD, Humphrey WB, Wallskog JA, Turner AS (2000) Direct current electrical stimulation increases the fusion rate of spinal fusion cages. Spine (Phila Pa 1976) 25:2580–2587
Assad M, Jarzem P, Leroux MA, Coillard C, Chernyshov AV, Charette S, Rivard CH (2003) Porous titanium-nickel for intervertebral fusion in a sheep model: part 1. Histomorphometric and radiological analysis. J Biomed Mater Res B Appl Biomater 64:107–120
Solchaga LA, et. al. (2012) Augment bone graft products compare favorably with autologous bone graft in an ovine model of lumbar interbody spine fusion. Spine (Phila Pa 1976) 37:461–467
Cunningham BW, Atkinson BL, Hu N, Kikkawa J, Jenis L, Bryant J, Zamora PO, McAfee PC (2009) Ceramic granules enhanced with B2A peptide for lumbar interbody spine fusion: an experimental study using an instrumented model in sheep. J Neurosurg Spine 10:300–307
Sherman BP, Lindley EM, Turner AS, Seim HB 3rd, Benedict J, Burger EL, Patel VV (2010) Evaluation of ABM/P-15 versus autogenous bone in an ovine lumbar interbody fusion model. Eur Spine J 19:2156–2163
Toth JM, Wang M, Estes BT, Scifert JL, Seim HB 3rd, Turner AS (2006) Polyetheretherketone as a biomaterial for spinal applications. Biomaterials 27:324–334
Blattert TR, Delling G, Dalal PS, Toth CA, Balling H, Weckbach A (2002) Successful transpedicular lumbar interbody fusion by means of a composite of osteogenic protein-1 (rhBMP-7) and hydroxyapatite carrier: a comparison with autograft and hydroxyapatite in the sheep spine. Spine (Phila Pa 1976) 27:2697–2705
Blattert TR, Delling G, Weckbach A (2003) Evaluation of an injectable calcium phosphate cement as an autograft substitute for transpedicular lumbar interbody fusion: a controlled, prospective study in the sheep model. Eur Spine J 12:216–223
Magin MN, Delling G (2001) Improved lumbar vertebral interbody fusion using rhOP-1: a comparison of autogenous bone graft, bovine hydroxylapatite (Bio-Oss), and BMP-7 (rhOP-1) in sheep. Spine (Phila Pa 1976) 26:469–478
Baramki HG, Steffen T, Lander P, Chang M, Marchesi D (2000) The efficacy of interconnected porous hydroxyapatite in achieving posterolateral lumbar fusion in sheep. Spine (Phila Pa 1976) 25:1053–1060
Kanayama M, Cunningham BW, Sefter JC, Goldstein JA, Stewart G, Kaneda K, McAfee PC (1999) Does spinal instrumentation influence the healing process of posterolateral spinal fusion? An in vivo animal model. Spine (Phila Pa 1976) 24:1058–1065
Kaya RA, Cavusoglu H, Tanik C, Kaya AA, Duygulu O, Mutlu Z, Zengin E, Aydin Y (2007) The effects of magnesium particles in posterolateral spinal fusion: an experimental in vivo study in a sheep model. J Neurosurg Spine 6:141–149
Kim DH, Jahng TA, Fu TS, Zhang HY, Novak SA (2004) Evaluation of HealosMP52 osteoinductive bone graft for instrumented lumbar intertransverse process fusion in sheep. Spine (Phila Pa 1976) 29:2800–2808
Wheeler DL, Jenis LG, Kovach ME, Marini J, Turner AS (2007) Efficacy of silicated calcium phosphate graft in posterolateral lumbar fusion in sheep. Spine J 7:308–317
Gupta MC, Theerajunyaporn T, Maitra S, Schmidt MB, Holy CE, Kadiyala S, Bruder SP (2007) Efficacy of mesenchymal stem cell enriched grafts in an ovine posterolateral lumbar spine model. Spine (Phila Pa 1976) 32:720–726 (discussion 727)
Mermer MJ, Gupta MC, Wheeler DL, Helgerson J, Reddi AH, Hazelwood S, Benson DR (2004) Efficacy of osteogenic protein-1 in a challenging multilevel fusion model. Spine (Phila Pa 1976) 29:249–256
Kandziora F, Pflugmacher R, Scholz M, Eindorf T, Schnake KJ, Haas NP (2004) Bioabsorbable interbody cages in a sheep cervical spine fusion model. Spine (Phila Pa 1976) 29:1845–1855 (discussion 1856)
Kandziora F, Schmidmaier G, Schollmeier G, Bail H, Pflugmacher R, Gorke T, Wagner M, Raschke M, Mittlmeier T, Haas NP (2002) IGF-I and TGF-beta1 application by a poly-(D,L-lactide)-coated cage promotes intervertebral bone matrix formation in the sheep cervical spine. Spine (Phila Pa 1976) 27:1710–1723
Kandziora F, Schollmeier G, Scholz M, Schaefer J, Scholz A, Schmidmaier G, Schroder R, Bail H, Duda G, Mittlmeier T, Haas NP (2002) Influence of cage design on interbody fusion in a sheep cervical spine model. J Neurosurg 96:321–332
Kandziora F, Pflugmacher R, Scholz M, Knispel C, Hiller T, Schollmeier G, Bail H, Schmidmaier G, Duda G, Raschke M, Haas NP (2002) Comparison of BMP-2 and combined IGF-I/TGF-ss1 application in a sheep cervical spine fusion model. Eur Spine J 11:482–493
Scholz M, Schleicher P, Eindorf T, Friedersdorff F, Gelinsky M, Konig U, Sewing A, Haas NP, Kandziora F (2010) Cages augmented with mineralized collagen and platelet-rich plasma as an osteoconductive/inductive combination for interbody fusion. Spine (Phila Pa 1976) 35:740–746
Goldschlager T, Rosenfeld JV, Ghosh P, Itescu S, Blecher C, McLean C, Jenkin G (2011) Cervical interbody fusion is enhanced by allogeneic mesenchymal precursor cells in an ovine model. Spine (Phila Pa 1976) 36:615–623
Goldschlager T, Ghosh P, Zannettino A, Williamson M, Rosenfeld JV, Itescu S, Jenkin G (2011) A comparison of mesenchymal precursor cells and amnion epithelial cells for enhancing cervical interbody fusion in an ovine model. Neurosurgery 68:1025-1034; discussion 1034–1025
Sugiyama S, Wullschleger M, Wilson K, Williams R, Goss B (2012) Reliability of clinical measurement for assessing spinal fusion: an experimental sheep study. Spine (Phila Pa 1976) 37:763–768
Li Y, Wu ZG, Li XK, Guo Z, Wu SH, Zhang YQ, Shi L, Teoh SH, Liu YC, Zhang ZY (2014) A polycaprolactone-tricalcium phosphate composite scaffold as an autograft-free spinal fusion cage in a sheep model. Biomaterials 35:5647–5659
Wu SH, Li Y, Zhang YQ, Li XK, Yuan CF, Hao YL, Zhang ZY, Guo Z (2013) Porous titanium-6 aluminum-4 vanadium cage has better osseointegration and less micromotion than a poly-ether-ether-ketone cage in sheep vertebral fusion. Artif Organs 37:E191–E201
Yamada K, Ito M, Akazawa T, Murata M, Yamamoto T, Iwasaki N (2015) A preclinical large animal study on a novel intervertebral fusion cage covered with high porosity titanium sheets with a triple pore structure used for spinal fusion. Eur Spine J 24:2530–2537
Yong MR, Saifzadeh S, Woodruff M, Askin GN, Labrom RD, Hutmacher DW, Adam CJ (2014) Biological performance of a polycaprolactone-based scaffold plus recombinant human morphogenetic protein-2 (rhBMP-2) in an ovine thoracic interbody fusion model. Eur Spine J 23:650–657
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts to declare for this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lindley, E.M., Barton, C., Blount, T. et al. An analysis of spine fusion outcomes in sheep pre-clinical models. Eur Spine J 26, 228–239 (2017). https://doi.org/10.1007/s00586-016-4544-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-016-4544-y