Abstract
Purpose
Intravenous dexamethasone is recommended in elective caesarean delivery to decrease postoperative pain. However, the efficacy of spinal anaesthesia with an intrathecal long-acting opioid such as morphine or diamorphine for caesarean delivery has not been systematically investigated.
Methods
We searched all randomized controlled trials (RCTs) of pregnant women undergoing caesarean delivery under spinal anaesthesia with an intrathecal morphine or diamorphine via MEDLINE, CENTRAL, EMBASE, ICTRP, and ClinicalTrials.gov on May 18, 2022. Primary outcomes were time to first rescue analgesia, consumption of oral morphine equivalents, and incidence of drug-related adverse reactions. We evaluated the risk of bias for each outcome using the Risk of Bias 2. We conducted a meta-analysis using a random effects model. We evaluated the certainty of evidence with the GRADE approach.
Results
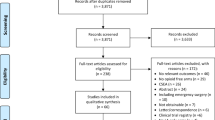
Five RCTs (455 patients) were included. The results of intravenous dexamethasone were as follows: time to first rescue analgesia (mean difference [MD] 0.99 h, 95% confidence interval [CI] − 0.86 to 2.84; very low certainty) and consumption of oral morphine equivalents (MD − 6.55 mg, 95% CI − 17.13 to 4.02; moderate certainty). No incidence of drug-related adverse reactions was reported (very low certainty).
Conclusion
The evidence was very uncertain about the efficacy of intravenous dexamethasone on time to first rescue analgesia and the incidence of drug-related adverse reactions. Intravenous dexamethasone probably reduces the consumption of oral morphine equivalents. Anaesthesiologists might want to consider intravenous dexamethasone for postoperative pain after caesarean delivery under spinal anaesthesia with an intrathecal long-acting opioid.



Similar content being viewed by others
Data availability
The data generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Gamez BH, Habib AS. Predicting severity of acute pain after cesarean delivery: a narrative review. Anesth Analg. 2018;126:1606–14.
Bornstein E, Husk G, Lenchner E, Grunebaum A, Gadomski T, Zottola C, Werner S, Hirsch JS, Chervenak FA. Implementation of a standardized post-cesarean delivery order set with multimodal combination analgesia reduces inpatient opioid usage. J Clin Med. 2020;10:7.
Roofthooft E, Joshi GP, Rawal N, Van de Velde M; PROSPECT working group of the european society of regional anaesthesia and pain therapy and supported by the obstetric anaesthetists’ association. PROSPECT guideline for elective caesarean section: updated systematic review and procedure-specific postoperative pain management recommendations. Anaesthesia. 2021; 76:665–80.
Committee Opinion No. 711: Opioid use and opioid use disorder in pregnancy. Obstet Gynecol. 2017;130:e81-94.
Patrick SW, Schiff DM, Committee on Substance use and Prevention. A Public Health Response to Opioid Use in Pregnancy. Pediatrics. 2017; 139:e20164070.
Landau R. Post-cesarean delivery pain. Management of the opioid-dependent patient before, during and after cesarean delivery. Int J Obstet Anesth. 2019; 39:105–16.
Heesen M, Rijs K, Hilber N, Eid K, Al-Oweidi A, Rossaint R, Klimek M. Effect of intravenous dexamethasone on postoperative pain after spinal anaesthesia—a systematic review with meta-analysis and trial sequential analysis. Anaesthesia. 2019;74:1047–56.
Singh NP, Makkar JK, Yadav N, Goudra BG, Singh PM. The analgesic efficacy of intravenous dexamethasone for postcaesarean pain: a systematic review with meta-analysis and trial sequential analysis. Eur J Anaesthesiol. 2021;39:498–510.
Abdelmonem M, Sayed FM, Mohammed OM, Abdeltawab AK, Abdelmonem H, Hosny MM, Masoud AT, Samy A, Abbas AM. Effect of dexamethasone on reducing pain and gastrointestinal symptoms associated with cesarean section: a systematic review and meta-analysis. Proc Obstet Gynecol. 2021;10:2.
Yonekura H, Mazda Y, Noguchi S, Tsunobuchi H, Shimaoka M. Trend in neuraxial morphine use and postoperative analgesia after cesarean delivery in Japan from 2005 to 2020. Sci Rep. 2022;12:17234.
Reed SE, Tan HS, Fuller ME, Krishnamoorthy V, Ohnuma T, Raghunathan K, Habib AS. Analgesia after cesarean delivery in the United States 2008–2018: a retrospective cohort study. Anesth Analg. 2021;133:1550–8.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372: n71.
Cumpston M, Li T, Page MJ, Chandler J, Welch VA, Higgins JP, Thomas J. Cochrane handbook for systematic reviews of interventions version 6.0 (updated July 2019). Cochrane; 2019.
Patanwala AE, Duby J, Waters D, Erstad BL. Opioid conversions in acute care. Ann Pharmacother. 2007;41:255–66.
Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan-a web and mobile app for systematic reviews. Syst Rev. 2016;5:210.
Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, Cheng HY, Corbett MS, Eldridge SM, Emberson JR, Hernán MA, Hopewell S, Hróbjartsson A, Junqueira DR, Jüni P, Kirkham JJ, Lasserson T, Li T, McAleenan A, Reeves BC, Shepperd S, Shrier I, Stewart LA, Tilling K, White IR, Whiting PF, Higgins JPT. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366: l4898.
Furukawa TA, Barbui C, Cipriani A, Brambilla P, Watanabe N. Imputing missing standard deviations in meta-analyses can provide accurate results. J Clin Epidemiol. 2006;59:7–10.
Higgins JPT, White IR, Wood AM. Imputation methods for missing outcome data in meta-analysis of clinical trials. Clin Trials. 2008;5:225–39.
Chaimani A, Mavridis D, Higgins JPT, Salanti G, White IR. Allowing for informative missingness in aggregate data meta-analysis with continuous or binary outcomes: Extensions to metamiss. Stata J. 2018;18:716–40.
Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, Norris S, Falck-Ytter Y, Glasziou P, DeBeer H, Jaeschke R, Rind D, Meerpohl J, Dahm P, Schünemann HJ. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J Clin Epidemiol. 2011; 64:383–94.
Ankouni T, Kanawati S, El Khatib R, El Hassan J, Itani SE, Rajab O, Naja Z. Ondansetron versus ondansetron with dexamethasone to prevent intrathecal-morphine pruritus for caesarean patients: randomised double-blind trial. J Obstet Gynaecol. 2021;41:1080–6.
Ituk U, Thenuwara K. The effect of a single intraoperative dose of intravenous dexamethasone 8 mg on post-cesarean delivery analgesia: a randomized controlled trial. Int J Obstet Anesth. 2018;35:57–63.
Jadon A, Sinha N, Agrawal A, Jain P. Effect of intravenous dexamethasone on postoperative nausea-vomiting (PONV) after intrathecal morphine during caesarean section. SOJ Anesthesiol Pain Manag. 2016;3:1–5.
Mehdiratta JE, Dominguez JE, Li YJ, Saab R, Habib AS, Allen TK. Dexamethasone as an analgesic adjunct for postcesarean delivery pain: a randomized controlled trial. Anesthesiol Res Pract. 2021;2021:4750149.
Wu JI, Lo Y, Chia YY, Liu K, Fong WP, Yang LC, Tan PH. Prevention of postoperative nausea and vomiting after intrathecal morphine for cesarean section: a randomized comparison of dexamethasone, droperidol, and a combination. Int J Obstet Anesth. 2007;16:122–7.
Polderman JA, Farhang-Razi V, Van Dieren S, Kranke P, DeVries JH, Hollmann MW, Preckel B, Hermanides J. Adverse side effects of dexamethasone in surgical patients. Cochrane Database Syst Rev. 2018; 11:CD011940.
De Oliveira GS, Almeida MD, Benzon HT, McCarthy RJ. Perioperative single dose systemic dexamethasone for postoperative pain: a meta-analysis of randomized controlled trials. Anesthesiology. 2011;115:575–88.
Acknowledgements
We are grateful to Dr. Unyime S. Ituk (Department of Anesthesia, University of Iowa Carver College of Medicine, Iowa, USA), and Dr. Terrence K. Allen (Department of Anesthesiology, Duke University Medical Center, Durham, USA), the authors of the included studies for providing the detailed information. We are also grateful to Dr. Yuki Kataoka (Department of Internal Medicine, Kyoto Min-Iren Asukai Hospital, Kyoto, Japan) for helping to conduct the sensitivity analyses to verify the robustness of the results by seeking informative missingness odds ratios. We would like to thank Editage (http://www.editage.jp) for English language editing.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Yuji Kamimura, Kyosuke Kamijo, Masahiro Banno, Tatsuya Tsuji, Yusuke Aoki, Hidekazu Ito, Motoshi Tanaka and Kazuya Sobue have no conflict of interest.
Registration
Open Science Framework (https://osf.io/42rsu/).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Kamimura, Y., Kamijo, K., Banno, M. et al. Efficacy of intravenous dexamethasone on postoperative pain after caesarean delivery under spinal anaesthesia with an intrathecal long-acting opioid: a systematic review and meta-analysis. J Anesth 37, 416–425 (2023). https://doi.org/10.1007/s00540-023-03183-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-023-03183-7