Abstract
Purpose
Several reports indicate that a neutrophil elastase inhibitor, sivelestat, may have prophylactic efficacy against a systemic inflammatory response after cardiovascular surgery with cardiopulmonary bypass (CPB). We evaluated the clinical pulmonary and cardiac effects of sivelestat.
Methods
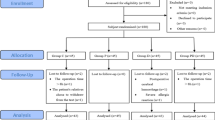
We performed a retrospective study of 25 pediatric patients who underwent elective cardiovascular surgery with CPB for ventricular septal defect with pulmonary hypertension. Ten patients received 0.2 mg·kg−1·h−1 sivelestat; the other is patients were the control group. There were no significant differences in demographic characteristics between the two groups. The \( P_{a_{O_2 } } \)/fractional inspired oxygen (\( F_{I_{O_2 } } \); P/F) ratio, the respiratory index (RI), and the fractional area change (FAC) of the left ventricle (LV) in the postoperative course were measured.
Results
The P/F ratio was higher in the sivelestat group compared with the control group and there were significant differences between the two groups immediately after weaning form CPB, and at 12 h after weaning from CPB (P < 0.05). The RI was lower in the sivelestat group compared with the control group and there were significant differences between the two groups at immediately after weaning from CPB, and at 6 h and 12 h after CPB (P < 0.05). The FAC of the LV was significantly better in the sivelestat group and there was a significant difference between the two groups on postoperative day (POD) 3 (P < 0.05).
Conclusion
We have shown that pediatric patients who underwent cardiovascular surgery with CPB who received sivelestat had a higher P/F ratio, a lower RI, and better FAC of the LV in the postoperative course.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Paparella D, Yau TM, Young E. Cardiopulmonary bypassinduced inflammation: pathophysiology and treatment. An update. Eur J Cardiothorac Surg. 2002;21:232–234.
Hill GE. Cardiopulmonary bypass-induced inflammation: is it important? J Cardiothorac Vasc Anesth. 1998;12(Suppl 1):21–25.
Journois D, Israel-Biet D, Pouard P, Rolland B, Silvester W, Vouhe P, Safran D. High-volume, zero-balanced hemofiltration to reduce delayed inflammatory response to cardiopulmonary bypass in children. Anesthesiology. 1996;85:965–976.
Milot J, Perron J, Lacasse Y, Letourneau L, Cartier PC, Maltais F. Incidence and predictors of ARDS after cardiac surgery. Chest. 2001;119:884–888.
Asimakopoulos G, Smith PL, Ratnatunqa CP, Taylor KM. Lung injury and acute respiratory distress syndrome after cardiopulmonary bypass. Ann Thorac Surg. 1999;68:1107–1115.
Tate RM, Repine JE. Neutrophils and the adult respiratory distress syndrome. Am Rev Respir Dis. 1983;128:552–559.
Lee WL, Downey GP. Leukocyte elastase. Physiological functions and role in acute lung injury. Am J Respir Crit Care Med. 2001;164:896–904.
Kawabata K, Suzuki M, Sugitani M, Imaki K, Toda M, Miyamoto T. ONO-5046, a novel inhibitor of human neutrophil elastase. Biochem Biophys Res Commun. 1991;177:814–820.
Yamazaki T, Ooshima H, Usui A, Watanabe T, Yasuura K. Protective effects of ONO-5046-Na, a specific neutrophil elastase inhibitor, on postperfusion lung injury. Ann Thorac Surg. 1999;68:2141–2146.
Hagio T, Nakao S, Matsuoka H, Matsumoto S, Kawabata K, Ohno H. Inhibition of neutrophil elastase activity attenuates complement-mediated lung injury in the hamster. Eur J Pharmacol. 2001;426:131–138.
Ishikawa N, Oda M, Kawaguchi M, Tsunezuka Y, Watanabe G. The effects of a specific neutrophil elastase inhibitor (ONO-5046) in pulmonary ischemia-reperfusion injury. Transpl Int. 2003;16:341–346.
Tamakuma S, Ogawa M, Aikawa N, Kubota T, Hirasawa H, Ishizuka A, Taenaka N, Hamada C, Matsuoka S, Abiru T. Relationship between neutrophil elastase and acute lung injury in humans. Pulm Pharmacol Ther. 2004;17:271–279.
Kadoi Y, Hinohara H, Kunimoto F, Saito S, Goto F, Kosaka T, Ieta K. Pilot study of the effects of ONO-5046 in patients with acute respiratory distress syndrome. Anesth Analg. 2004;99:872–877.
Hoshi K, Kurosawa S, Kato M, Andoh K, Satoh D, Kaise A. Sivelestat, a neutrophil elastase inhibitor, reduces mortality rate of critically ill patients. Tohoku J Exp Med. 2005;207:143–148.
Okayama N, Kakihana Y, Setoguchi D, Imabayashi T, Omae T, Matsunaga A, Kanmura Y. Clinical effects of a neutrophil elastase inhibitor, sivelestat, in patients with acute respiratory distress syndrome. J Anesth. 2006;20:6–10.
Ikeda T, Endo S, Miura M, Matsushita M, Sato N, Nakamura F. Investigation on sivelestat sodium efficacy for acute lung injury at three different ICUs (in Japanese with English abstract). J Jpn Soc Intensive Care Med. 2006;13:479–480.
Zeiher BG, Artigas A, Vincent JL, Dmitrienko A, Jackson K, Thompson BT, Bernard G; STRIVE Study Group. Neutrophil elastase inhibition in acute lung injury: results of the STRIVE study. Crit Care Med. 2004;32:1695–1702.
Matsuzaki K, Hiramatsu Y, Homma S, Sato S, Shigeta O, Sakakibara Y. Sivelestat reduces inflammatory mediators and preserves neutrophil deformability during simulated extracorporeal circulation. Ann Thorac Surg. 2005;80:611–617.
Wakayama F, Fukuda I, Suzuki Y, Kondo N. Neutrophil elastase inhibitor, sivelestat, attenuates acute lung injury after cardiopulmonary bypass in the rabbit endotoxemia model. Ann Thorac Surg. 2007;83:153–160.
Minami T, Kito K. Prophylactic effects of neutrophil elastase inhibitor for patients undergoing surgery for thoracic aortic aneurysm: a retrospective study (in Japanese with English abstract). Masui (Jpn J Anesthesiol). 2006;55:977–982.
Ryugo M, Sawa Y, Takano H, Matsumiya G, Iwai S, Ono M, Hata H, Yamauchi T, Nishimura M, Fujino Y, Matsuda H. Effect of a polymorphonuclear elastase inhibitor (sivelestat sodium) on acute lung injury after cardiopulmonary bypass: findings of a double-blind randomized study. Surg Today. 2006;36:321–326.
Furusawa T, Tsukioka K, Fukui D, Sakaguchi M, Seto T, Wada Y, Amano J. The effects of a neutrophil elastase inhibitor on the postoperative respiratory failure of acute aortic dissection. Thorac Cardiovasc Surg. 2006;54:404–407.
Tomashefski JF Jr, Davies P, Boggis C, Greene R, Zapol WM, Leid LM. The pulmonary vascular lesions of the adult respiratory distress syndrome. Am J Pathol. 1983;112:112–126.
Prewitt RM, MacCarthy J, Wood LD. Treatment of acute low pressure pulmonary edema in dogs. J Clin Invest. 1981;67:409–418.
Brix-Christensen V. The systemic inflammatory response after cardiac surgery with cardiopulmonary bypass in children. Acta Anesthesiol Scand. 2001;45:671–679.
Gessler P, Pfenninger J, Pfammatter JP, Carrel T, Baenziger O, Dahinden C. Plasma levels of interleukin-8 and expression of interleukin-8 receptors on circulating neutrophils and monocytes after cardiopulmonary bypass in children. J Thorac Cardiovasc Surg. 2003;126:718–725.
Seghaye MC. The clinical implications of the systemic inflammatory reaction related to cardiac operations in children. Cardiol Young. 2003;13:228–239.
Harig F, Feyrer R, Mahmoud FO, Blum U, von der Emde J. Reducing the post-pump syndrome by using heparin-coated circuit, steroids, or aprotinin. Thorac Cardiovasc Surg. 1999;47:111–118.
Miyaji K, Hannan RL, Ojito J, Jacobs JP, White JA, Burke RP. Heparin-coated cardiopulmonary bypass circuits: clinical effects in pediatric cardiac surgery. J Card Surg. 2000;15:194–198.
Berdat PA, Eichenberger E, Ebell J, Pfammatter JP, Pavlovic M, Zobrist C, Gygax E, Nydegger U, Carrel T. Elimination of proinflammatory cytokines in pediatric cardiac surgery: analysis of ultrafiltration method and filter type. J Thorac Cardiovasc Surg. 2004;127:1688–1696.
Mossinger H, Dietrich W, Braun SL, Jochum M, Meisner H, Richter JA. High-dose aprotinin reduces activation of hemostasis, allogenic blood requirement, and duration of postoperative ventilation in pediatric cardiac surgery. Ann Thorac Surg. 2003;75:430–437.
Wippermann CF, Schmid FX, Eberle B, Huth RG, Kampmann C, Schranz D, Oelert H. Reduced inotropic support after aprotinin therapy during pediatric cardiac operations. Ann Thorac Surg. 1999;67:173–176.
Schroeder VA, Pearl JM, Schwartz SM, Shanley TP, Manning PB, Nelson DP. Combined steroid treatment for congenital heart surgery improves oxygen delivery and reduces postbypass inflammatory mediator expression. Circulation. 2003;107:2823–2828.
Bronicki RA, Backer CL, Baden HP, Mavroudis C, Vrawford SE, Green TP. Dexamethasone reduces the inflammatory response to cardiopulmonary bypass in children. Ann Thorac Surg. 2000;69:1490–1495.
Morishima S, Fujiwara T, Oshitomi T, Aotsuka H, Okajima Y, Tohyama T. Echocardiographic evaluation of left ventricular function and volume immediately after intracardiac repair for ventricular septal defect (in Japanese with English abstract). Ped Cardiol Cardiac Surg. 2004;20:86–93.
Hennein HA, Ebba H, Rodriguez JL, Merrick SH, Keith FM, Bronstein MH, Leung JM, Mangano DT, Greenfield LJ, Lankin JS. Relationship of the proinflammatory cytokines to myocardial ischemia and dysfunction after uncomplicated coronary revascularization. J Thorac Surg. 1994;108:626–635.
Yokoyama T, Vaca L, Rossen RD, Durante W, Hazarika P, Mann DL. Cellular basis for negative inotropic effects of tumor necrosis factor-alpha in the adult mammalian heart. J Clin Invest. 1993;92:2303–2312.
Hovels-Gurich HH, Vazquez-Jimenez JF, Silvestri A, Schumacher K, Minkenberg R, Duchateau J, Messmer BJ, von Bernuth G, Seghaye MC. Production of proinflammatory cytokines and myocardial dysfunction after arterial switch operation in neonates with transposition of great arteries. J Thorac Cardiovasc Surg. 2002;124:811–820.
Author information
Authors and Affiliations
About this article
Cite this article
Toyama, S., Hatori, F., Shimizu, A. et al. A neutrophil elastase inhibitor, sivelestat, improved respiratory and cardiac function in pediatric cardiovascular surgery with cardiopulmonary bypass. J Anesth 22, 341–346 (2008). https://doi.org/10.1007/s00540-008-0645-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-008-0645-z