Abstract
Background
Taste and smell abnormalities (TSA) commonly occur in cancer and are associated with anorexia, early satiety, malnutrition, weight loss and reduced quality of life. A recent study found a high TSA prevalence in newly diagnosed cancer patients before treatment. This suggests that TSA may originate from the tumour itself. No previous study has examined TSA, both subjectively and objectively, in newly diagnosed, treatment-naïve cancer patients. This study aimed to address this gap.
Methods
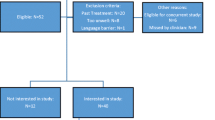
This prospective observational study recruited consecutive, newly diagnosed, treatment-naïve patients with solid tumours at Radiation Oncology Out-patients. Self-reported taste and smell changes since becoming ill were evaluated using modified Taste and Smell Survey, and objective taste and smell tests were conducted using ‘Sniffin’ Sticks Olfactory Test® and Burghart Taste Strips®. Nutritional status was assessed with abridged Patient-Generated Subjective Global Assessment.
Results
Thirty completed the study. Seventy-four per cent had at least one TSA. Taste changes and/or abnormalities were more prevalent than smell, and subjective taste changes more common than objective abnormalities. Although less common, smell abnormalities impacted quality of life more. TSA characteristics were heterogeneous. Forty-seven per cent were at malnutrition risk. No association was found between TSA and nutritional status.
Conclusions
Over two thirds had at least one TSA and almost half were at malnutrition risk. Self-reported TSA included changes in taste and smell perception, and most commonly persistent bad taste. This study demonstrated the complexity of TSA assessment and the prevalence, severity and impact of these and related symptoms in treatment-naïve cancer patients.


Similar content being viewed by others
References
Hummel T, Landis BN, Hüttenbrink KB (2011) Smell and taste disorders. GMS Curr Top Otorhinolaryngol Head Neck Surg 10:1–15
Wrobel BB, Leopold DA (2004) Clinical assessment of patients with taste and smell disorder. Otolaryngol Clin N Am 37(6):1127–1142
Stinton N, Ali Atif M, Barkat N et al (2010) Influence of smell loss on taste function. Behav Neurosci 124(2):256–264
Boseveldt S, de Graff K (2017) The differential role of smell and taste for eating behaviour. Perception 46(3–4):307–319
Henkin RI, Levy LM, Fordyce A (2013) Taste and smell function in chronic disease: a review of clinical and biochemical evaluations of taste and smell dysfunction in over 5000 patients at the taste and smell Clinic in Washington, DC. Am J Otolaryngol 34(5):477–489
Yoshinaka M, Ikebe K, Uota M, Ogawa T, Okada T, Inomata C, Takeshita H, Mihara Y, Gondo Y, Masui Y, Kamide K, Arai Y, Takahashi R, Maeda Y (2016) Age and sex differences in the taste sensitivity of young adult, young-old and old-old Japanese. Geriatr Gerontol Int 16(12):1281–1288
Kirkova J, Walsh D, Rybicki L, Davis MP, Aktas A, Tao Jin, Homsi J (2010) Symptom severity and distress in advanced cancer. Palliat Med 24(3):330–339
Mahmoud FA, Aktas A, Walsh D, Hullihen B (2011) A pilot study of taste changes among hospice inpatients with advanced cancer. Am J Hosp Palliat Care 28:487–492
McLoughlin L, Mahon SM (2012) Understanding taste dysfunction in patients with cancer. Clin J Oncol Nurs 16(2):171–178
Epstein JB, Barasch A (2010) Taste disorders in cancer patients: pathogenesis, and approach to assessment and management. Oral Oncol 46:77–81
Gamper E, Zabernigg A, Wintner LM et al (2012) Coming to your senses: detecting taste and smell alterations in chemotherapy patients. A systematic review. J Pain Symptom Manag 44:880–895
Hutton JL, Baracos VE, Wismer WV (2007) Chemosensory dysfunction is a primary factor in the evolution of declining nutritional status and quality of life in patients with advanced cancer. J Pain Symptom Manag 33:156–165
Newell S, Sanson-Fisher R, Girgis A et al (1998) How well do medical oncologists' perceptions reflect their patients’ reported physical and psychosocial problems? Data from a survey of five oncologists. Cancer J 83:1640–1651
Zabernigg A, Gamper EM, Giesinger JM, Rumpold G, Kemmler G, Gattringer K, Sperner-Unterweger B, Holzner B (2010) Taste alterations in cancer patients receiving chemotherapy: a neglected side effect? Oncologist 15:913–920
Ovesen L, Hannibal J, Sorensen M et al (1991) Food intake, eating-related complaints, and smell and taste sensations in patients with cancer of the lung, ovary and breast undergoing chemotherapy. Clin Nutr 10(6):336–334
Steinbach S, Hummel T, Bohner C et al (2009) Qualitative and quantitative assessment of taste and smell changes in patients undergoing chemotherapy for breast cancer or gynecologic malignancies. J Clin Oncol 27(11):1899–1905
Cohen J, Laing DG, Wilkes FJ, Chan A, Gabriel M, Cohn RJ (2014) Taste and smell dysfunction in childhood cancer survivors. Appetite 75:135–140
Brisbois TD, Hutton JL, Baracos VE, Wismer WV (2006) Taste and smell abnormalities as an independent cause of failure of food intake in patients with advanced cancer - an argument for the application of sensory science. J Palliat Care 22:111–114
Spotten L, Corish C, Lorton C et al (2017) Subjective and objective taste and smell changes in cancer: a narrative review. Ann Oncol 28:969–984
Huldij A, Giesbers A, Klein Poelhuis EH et al (1986) Alterations in taste appreciation in cancer patients during treatment. Cancer Nurs 9(1):38–42
Bernhardson BM, Tischlman C, Rutquist LE (2008) Self–reported taste and smell changes during cancer chemotherapy. Support Care Cancer 16(3):275–283
Boltong A, Campbell K (2013) Taste changes: a problem for patients and their dietitians. Nutr Diet 70:262–269
Spotten L, Corish C, Lorton C et al (2016) Subjective taste and smell changes in treatment-naïve people with solid tumours. Support Cancer Care 24:3201–3208
Schalk P, Kohl M, Herrmann HJ, Schwappacher R, Rimmele ME, Buettner A, Siebler J, Neurath MF, Zopf Y (2018) Influence of cancer and acute inflammatory disease on taste perception: a clinical pilot study. Support Care Cancer 26:843–851
Williams LR, Cohen MH (1978) Altered taste thresholds in lung cancer. Am J Clin Nut 31(1):122–125
Kamath S, Booth P, Lad TE, Kohrs MB, McGuire WP (1983) Taste thresholds of patients with cancer of the esophagus. Cancer 52(2):386–389
Belqaid K, Orrevall Y, McGreevy J, Månsson-Brahme E, Wismer W, Tishelman C, Bernhardson BM (2014) Self-reported taste and smell alterations in patients under investigation for lung cancer. Acta Oncol 53:1405–1412
Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, Carbone PP (1982) Toxicity and response criteria of the eastern cooperative oncology group. Am J Clin Oncol 5:649–655
Heald AE, Pieper CF, Schiffman SS (1998) Taste and smell complaints in HIV-infected patients. AIDS 12:1667–1674
Leyrer CM, Chan MD, Peiffer AM, Horne E, Harmon M, Carter AF, Hinson WH, Mirlohi S, Duncan SE, Dietrich AM, Lesser GJ (2014) Taste and smell disturbances after brain irradiation: a dose-volume histogram analysis of a prospective observational study. Pract Radiat Oncol 4(2):130–135
Gabrielson DK, Scaffidi D, Leung E, Stoyanoff L, Robinson J, Nisenbaum R, Brezden-Masley C, Darling PB (2013) Use of an abridged scored patient-generated subjective global assessment (abPG-SGA) as a nutritional screening tool for cancer patients in an outpatient setting. Nutr and Cancer 65(2):234–239
World Health Organisation: BMI Classification. (Internet: http://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi - accessed 31 October 2017)
Landis BN, Welge-Luessen A, Brämerson A, Bende M, Mueller CA, Nordin S, Hummel T (2009) "taste strips" - a rapid, lateralized, gustatory bedside identification test based on impregnated filter papers. J Neurol 256(2):242–248
Kobal G, Hummel TH, Sekinger B et al (1996) Sniffin’sticks’: screening of olfactory performance. Rhinology 34:222–226
Hummel T, Sekinger B, Wolf SR, Pauli E, Kobal G (1997) Sniffin’ sticks – olfactory performance assessed by the combined testing of odour identification, odour discrimination and olfactory threshold. Chem Senses 22:39–52
Gerkin RC, Castro JB (2015) The number of olfactory stimuli that humans can discriminate is still unknown. eLife 4 https://elifesciences.org/articles/08127
Bernhardson BM, Tishelman C, Rytqvist LE (2007) Chemosensory changes experienced by patients undergoing cancer chemotherapy: a qualitative interview study. J Pain Symptom Manag 34(4):403–412
Steinbach S, Hundt W, Schmalfeldt B, Böhner C, Berktold S, Wolf P, Harbeck N (2012) Effect of platinum-containing chemotherapy on olfactory, gustatory, and hearing function in ovarian cancer patients. Arch Gynecol Obstet 286(2):473–480
Sánchez-Lara K, Sosa-Sánchez R, Green-Renner D, Rodríguez C, Laviano A, Motola-Kuba D, Arrieta O (2010) Influence of taste disorders on dietary behaviors in cancer patients under chemotherapy. Nutr J 9:15
Acknowledgements
We would like to acknowledge the nursing and administration staff at the Radiation Oncology Out-patient at St James Hospital for their assistance with data collection processes.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
This study has not been previously published.
We have full control of all primary data and can allow the journal to review the data if requested.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Uí Dhuibhir, P., Barrett, M., O’Donoghue, N. et al. Self-reported and objective taste and smell evaluation in treatment-naive solid tumour patients. Support Care Cancer 28, 2389–2396 (2020). https://doi.org/10.1007/s00520-019-05017-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-019-05017-3