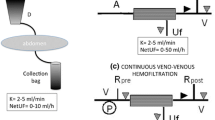
Abstract
Children requiring long-term kidney replacement therapy are a “rare disease” cohort. While the basic technical requirements for hemodialysis (HD) are similar in children and adults, key aspects of the child’s cardiovascular anatomy and hemodynamic specifications must be considered. In this article, we describe the technical requirements for long-term HD therapy for children and the devices that are currently available around the world. We highlight the characteristics and major technical shortcomings of permanent central venous catheters, dialyzers, dialysis machines, and software available to clinicians who care for children. We show that currently available HD machines are not equipped with appropriately small circuits and sensitive control mechanisms to perform safe and effective HD in the youngest patients. Manufacturers limit their liability, and health regulatory agencies permit the use of devices, only in children according to the manufacturers’ pre-specified weight limitations. Although registries show that 6–23% of children starting long-term HD weigh less than 15 kg, currently, there is only one long-term HD device that is cleared for use in children weighing 10 to 15 kg and none is available and labelled for use in children weighing less than 10 kg anywhere in the world. Thus, many children are being treated “off-label” and are subject to interventions delivered by medical devices that lack pediatric safety and efficacy data. Moreover, recent improvements in dialysis technology offered to adult patients are denied to most children. We, in turn, advocate for concerted action by pediatric nephrologists, industry, and health regulatory agencies to increase the development of dedicated HD machines and equipment for children.
Similar content being viewed by others
References
Fine RN, De Palma JR, Lieberman E et al (1968) Extended hemodialysis in children with chronic renal failure. J Pediatr 73:706–713. https://doi.org/10.1016/S0022-3476(68)80176-3
Broyer M, Loirat C, Kleinknecht C (1972) Technical aspects and results of regular hemodialysis in children. Acta Paediatr 61:677–684. https://doi.org/10.1111/j.1651-2227.1972.tb15966.x
Agence de la Biomédecine (2019) Rapport 2019. https://www.agence-biomedecine.fr/IMG/pdf/rapport_rein_2019_2021-10-14.pdf. Accessed 5 July 2023
Bonthuis M, Vidal E, Bjerre A et al (2021) Ten-year trends in epidemiology and outcomes of pediatric kidney replacement therapy in Europe: data from the ESPN/ERA-EDTA Registry. Pediatr Nephrol 36:2337–2348. https://doi.org/10.1007/s00467-021-04928-w
United States Renal Data System (2021) USRDS Annual Data Report: epidemiology of kidney disease in the United States. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, MD https://adr.usrds.org/2021. Accessed 25 July 2022
International pediatric dialysis Network. http://www.pedpd.org/. Accessed 11 September 2022
Shroff R, Calder F, Bakkaloğlu S et al (2019) Vascular access in children requiring maintenance haemodialysis: a consensus document by the European Society for Paediatric Nephrology Dialysis Working Group. Nephrol Dial Transplant 34:1746–1765. https://doi.org/10.1093/ndt/gfz011
Borzych-Duzalka D, Shroff R, Ariceta G et al (2019) Vascular access choice, complications, and outcomes in children on maintenance hemodialysis: findings from the International Pediatric Hemodialysis Network (IPHN) Registry. Am J Kidney Dis 74:193–202. https://doi.org/10.1053/j.ajkd.2019.02.014
Lok CE, Huber TS, Lee T et al (2020) KDOQI clinical practice guideline for vascular access: 2019 update. Am J Kidney Dis 75:S1–S164. https://doi.org/10.1053/j.ajkd.2019.12.001
United States Renal Data System (2022) USRDS Annual Data Report: Epidemiology of kidney disease in the United States. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, MD https://adr.usrds.org/2022. Accessed 5 July 2023
Bruno C, Moumneh R, Sauvage E, Stronach L, Waters K, Simcock I, Arthurs O, Schievano S, Capelli C, Shroff R (2022) Central venous catheter malfunction in children: a bioengineering approach. Clin J Am Soc Nephrol 17:1382–1384. https://doi.org/10.2215/CJN.01470222
Bruno C, Sauvage E, Simcock I et al (2022) Computational investigation of the haemodynamics shows criticalities of central venous lines used for chronic haemodialysis in children. Front Pediatr 10:1055212. https://doi.org/10.3389/fped.2022.1055212
Fischbach M, Edefonti A, Schröder C et al (2005) Hemodialysis in children: general practical guidelines. Pediatr Nephrol 20:1054–1066. https://doi.org/10.1007/s00467-005-1876-y
Leffell MS, Kim D, Vega RM et al (2014) Red blood cell transfusions and the risk of allosensitization in patients awaiting primary kidney transplantation. Transplantation 97:525–533. https://doi.org/10.1097/01.tp.0000437435.19980.8f
Raina R, Lam S, Raheja H et al (2019) Pediatric intradialytic hypotension: recommendations from the Pediatric Continuous Renal Replacement Therapy (PCRRT) Workgroup. Pediatr Nephrol 34:925–941. https://doi.org/10.1007/s00467-018-4190-1
Hothi DK, Rees L, Marek J et al (2009) Pediatric myocardial stunning underscores the cardiac toxicity of conventional hemodialysis treatments. Clin J Am Soc Nephrol 4:790–797. https://doi.org/10.2215/CJN.05921108
Idrovo A, Pignatelli R, Loar R et al (2021) Preserved cerebral oxygenation with worsening global myocardial strain during pediatric chronic hemodialysis. J Am Soc Nephrol 32:2912–2919. https://doi.org/10.1681/ASN.2021020193
Anazodo UC, Wong DY, Théberge J et al (2023) Hemodialysis-related acute brain injury demonstrated by application of intradialytic magnetic resonance imaging and spectroscopy. J Am Soc Nephrol 34:1090–1104. https://doi.org/10.1681/ASN.0000000000000105
Peters SAE, Bots ML, Canaud B et al (2016) Haemodiafiltration and mortality in end-stage kidney disease patients: a pooled individual participant data analysis from four randomized controlled trials. Nephrol Dial Transplant 31:978–984. https://doi.org/10.1093/ndt/gfv349
Shroff R, Smith C, Ranchin B et al (2019) Effects of hemodiafiltration versus conventional hemodialysis in children with ESKD: the HDF, heart and height study. J Am Soc Nephrol 30:678–691. https://doi.org/10.1681/ASN.2018100990
Blankestijn PJ, Vernooij RWM, Hockham C et al (2023) Effect of hemodiafiltration or hemodialysis on mortality in kidney failure. N Engl J Med 389:700–709. https://doi.org/10.1056/NEJMoa2304820
Shroff R, Basile C, Van Der Sande F et al (2023) Haemodiafiltration for all: are we CONVINCEd? Nephrol Dial Transplant. https://doi.org/10.1093/ndt/gfad136
Ronco C, Marchionna N, Brendolan A et al (2018) Expanded haemodialysis: from operational mechanism to clinical results. Nephrol Dial Transplant 33:iii41–iii47. https://doi.org/10.1093/ndt/gfy202
García-Prieto A, De La Flor JC, Coll E et al (2023) Expanded hemodialysis: what’s up, Doc? Clin Kidney J 16:1071–1080. https://doi.org/10.1093/ckj/sfad033
Ağbaş A, Canpolat N, Çalışkan S et al (2018) Hemodiafiltration is associated with reduced inflammation, oxidative stress and improved endothelial risk profile compared to high-flux hemodialysis in children. PLoS One 13:e0198320. https://doi.org/10.1371/journal.pone.0198320
Fadel FI, Makar SH, Zekri H, Ahmed DH, Aon AH (2015) The effect of on-line hemodiafiltration on improving the cardiovascular function parameters in children on regular dialysis. Saudi J Kidney Dis Transpl 26:39–46. https://doi.org/10.4103/1319-2442.148731
Maduell F, Broseta JJ, Rodríguez-Espinosa D et al (2022) Comparison of four medium cut-off dialyzers. Clin Kidney J 15:2292–2299. https://doi.org/10.1093/ckj/sfac167
Martínez-Miguel P, Gimena Muñoz R, Barrionuevo González M et al (2023) Comparison of 2 medium cutoff dialyzers versus on-line hemodiafiltration regarding depurative ability and albumin loss: an uncontrolled clinical research. Artif Organs 47:589–594. https://doi.org/10.1111/aor.14466
Morgenstern BZ, Wühl E, Nair KS et al (2006) Anthropometric prediction of total body water in children who are on pediatric peritoneal dialysis. J Am Soc Nephrol 17:285–293. https://doi.org/10.1681/ASN.2005050568
Zaloszyc A, Fischbach M, Schaefer B et al (2016) Body composition monitoring-derived urea distribution volume in children on chronic hemodialysis. Pediatr Nephrol 31:991–999. https://doi.org/10.1007/s00467-015-3283-3
Preciado P, Zhang H, Thijssen S et al (2019) All-cause mortality in relation to changes in relative blood volume during hemodialysis. Nephrol Dial Transplant 34:1401–1408. https://doi.org/10.1093/ndt/gfy286
Flythe JE, Chang TI, Gallagher MP et al (2020) Blood pressure and volume management in dialysis: conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) Controversies Conference. Kidney Int 97:861–876. https://doi.org/10.1016/j.kint.2020.01.046
Geer JJ, Shah S, Williams E et al (2017) Faster rate of blood volume change in pediatric hemodialysis patients impairs cardiac index. Pediatr Nephrol 32:341–345. https://doi.org/10.1007/s00467-016-3486-2
Paglialonga F, Consolo S, Edefonti A, Montini G (2018) The first hour refill index: a promising marker of volume overload in children and young adults on chronic hemodialysis. Pediatr Nephrol 33:1209–1214. https://doi.org/10.1007/s00467-018-3915-5
Garzoni D, Keusch G, Kleinoeder T et al (2007) Reduced complications during hemodialysis by automatic blood volume controlled ultrafiltration. Int J Artif Organs 30:16–24. https://doi.org/10.1177/039139880703000104
Patel HP, Goldstein SL, Mahan JD et al (2007) A standard, noninvasive monitoring of hematocrit algorithm improves blood pressure control in pediatric hemodialysis patients. Clin J Am Soc Nephrol 2:252–257. https://doi.org/10.2215/CJN.02410706
Fadel FI, Makar SH, Eskander AE, Aon AH (2014) Decreasing intra-dialytic morbid events and assessment of dry weight in children on chronic hemodialysis using non-invasive changes in hematocrit. Saudi J Kidney Dis Transpl 25:1030–1037. https://doi.org/10.4103/1319-2442.139916
Maggiore Q, Pizzarelli F, Santoro A et al (2002) The effects of control of thermal balance on vascular stability in hemodialysis patients: results of the European randomized clinical trial. Am J Kidney Dis 40:280–290. https://doi.org/10.1053/ajkd.2002.34506
Tsujimoto Y, Tsujimoto H, Nakata Y et al (2019) Dialysate temperature reduction for intradialytic hypotension for people with chronic kidney disease requiring haemodialysis. Cochrane Database Syst Rev 7:CD012598. https://doi.org/10.1002/14651858.CD012598.pub2
Garg AX, Al-Jaishi AA, Dixon SN et al (2022) Personalised cooler dialysate for patients receiving maintenance haemodialysis (MyTEMP): a pragmatic, cluster-randomised trial. Lancet 400:1693–1703. https://doi.org/10.1016/S0140-6736(22)01805-0
Badr B, Bories P, Marais R et al (2014) Transonic, thermodilution, or ionic dialysance to manage vascular access: which method is best?: Managing vascular access. Hemodial Int 18:127–135. https://doi.org/10.1111/hdi.12092
Schneditz D (1999) Validation of haemodialysis recirculation and access blood flow measured by thermodilution. Nephrol Dial Transplant 14:376–383. https://doi.org/10.1093/ndt/14.2.376
Karava V, Kwon T, Franco G et al (2019) Ultrasound dilution and thermodilution versus color Doppler ultrasound for arteriovenous fistula assessment in children on hemodialysis. Pediatr Nephrol 34:2381–2387. https://doi.org/10.1007/s00467-019-04297-5
Ságová M, Wojke R, Maierhofer A et al (2019) Automated individualization of dialysate sodium concentration reduces intradialytic plasma sodium changes in hemodialysis. Artif Organs 43:1002–1013. https://doi.org/10.1111/aor.13463
Rodriguez A, Morena M, Bargnoux A et al (2021) Quantitative assessment of sodium mass removal using ionic dialysance and sodium gradient as a proxy tool: comparison of high-flux hemodialysis versus online hemodiafiltration. Artif Organs 45:E280–E292. https://doi.org/10.1111/aor.13923
Maduell F, Broseta JJ, Rodríguez-Espinosa D et al (2023) Practical implementation and clinical benefits of the new automated dialysate sodium control biosensor. Clin Kidney J 16:859–867. https://doi.org/10.1093/ckj/sfad013
Fujisaki K, Joki N, Tanaka S et al (2021) Pre-dialysis hyponatremia and change in serum sodium concentration during a dialysis session are significant predictors of mortality in patients undergoing hemodialysis. Kidney Int Rep 6:342–350. https://doi.org/10.1016/j.ekir.2020.11.009
Locatelli F, Stefoni S, Petitclerc T et al (2012) Effect of a plasma sodium biofeedback system applied to HFR on the intradialytic cardiovascular stability. Results from a randomized controlled study. Nephrol Dial Transplant 27:3935–3942. https://doi.org/10.1093/ndt/gfs091
Petitclerc T, Mercadal L (2023) Automated adjustment of dialysate sodium by the hemodialysis monitor: Rationale, implementation, and clinical benefits. Semin Dial 36:184–192. https://doi.org/10.1111/sdi.13132
Mosca M, Kouajip-Mabou A, De Mul A et al (2023) Daily practice evaluation of the paediatric set of a next-generation long-term haemodialysis machine. Pediatr Nephrol 38:3863–3866. https://doi.org/10.1007/s00467-023-05943-9
Assimon MM, Wenger JB, Wang L, Flythe JE (2016) Ultrafiltration rate and mortality in maintenance hemodialysis patients. Am J Kidney Dis 68:911–922. https://doi.org/10.1053/j.ajkd.2016.06.020
Karava V, Benzouid C, Kwon T et al (2018) Interdialytic weight gain and vasculopathy in children on hemodialysis: a single center study. Pediatr Nephrol 33:2329–2336. https://doi.org/10.1007/s00467-018-4026-z
Paglialonga F, Consolo S, Galli MA et al (2015) Interdialytic weight gain in oligoanuric children and adolescents on chronic hemodialysis. Pediatr Nephrol 30:999–1005. https://doi.org/10.1007/s00467-014-3005-2
Ranchin B, Schmitt CP, Warady B et al (2023) Devices for long-term hemodialysis in small children—a plea for action. Kidney Int 103:1038–1040. https://doi.org/10.1016/j.kint.2023.03.018
Espinoza J, Shah P, Nagendra G et al (2022) Pediatric medical device development and regulation: current state, barriers, and opportunities. Pediatrics 149:e2021053390. https://doi.org/10.1542/peds.2021-053390
Bourgeois FT, Espinoza JC (2023) Advancing equity in medical device development for children. JAMA Pediatr 177:561–562. https://doi.org/10.1001/jamapediatrics.2023.0790
Acknowledgements
We would like to thank the nurses on our teams for their knowledge of dialysis machines and their commitment to children’s care, the patients and their families for their trust, and the manufacturers’ representatives for their support in gathering information about the devices. We also gratefully acknowledge the following colleagues for help with data extraction: Marjolein Bonthuis for the ESPN/ERA registry, Kimberly Nieman for the USRDS registry, and Dagmara Borzych-Duzalka for the IPHN registry.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
RS has received research grants from Fresenius Medical Care, speaker honoraria from Fresenius Medical Care and Amgen, and consulting fees from AstraZeneca and Humacyte. CPS has received consulting fees from Baxter, Iperboreal, and Stadapharma; speaker honoraria from Fresenius Medical Care; and research grants from Baxter and Invizius. The other authors do not declare any conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ranchin, B., Schmitt, C.P., Warady, B.A. et al. Technical requirements and devices available for long-term hemodialysis in children—mind the gap!. Pediatr Nephrol (2023). https://doi.org/10.1007/s00467-023-06233-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00467-023-06233-0