Abstract
Background
Hemolytic uremic syndrome secondary to Shiga-toxin-producing Escherichia coli infection (STEC-HUS) generally shows a favorable outcome. Few cases develop extra-renal complications, since neurological involvement is an important cause of morbidity and mortality. The role of complement in STEC-HUS has been recently highlighted, and the use of eculizumab in severe cases has been communicated. HUS results from environmental and genetic factors, but the simultaneous occurrence of STEC and complement mutations remains undetermined.
Methods
A pediatric case with severe STEC-HUS carrying CFH mutations, with favorable response to eculizumab is analyzed.
Results
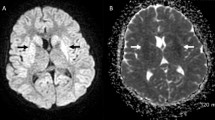
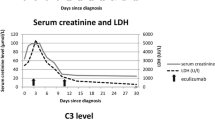
STEC-HUS was diagnosed in a 4-year-old girl with classic HUS, including low C3. Peritoneal dialysis was started due to hypertension, oligoanuria, and pleural effusion. She evolved with generalized tonic–clonic seizures and required mechanical ventilation. MRI reported multiple supra- and infratentorial ischemic lesions with laminar/striatal cortical necrosis and leukoencephalopathy. After two eculizumab doses, a significative stabilization in diuresis, blood pressure, creatinine, and C3 was achieved. At the third week, episodes of massive digestive bleeding and a life-threatening condition required a colectomy thus preserving the ileocecal valve. Due to atypical evolution, a genetic study was considered, identifying two heterozygous variants (CFH S1191L/V1197A).
Conclusion
STEC-HUS in patients with a genetic predisposition has been previously reported, but the low frequency of occurrence makes it a rare disease. As in the present case, patients with atypical course might benefit from genetic analysis to evaluate early eculizumab initiation and to better understand its phenotype.
Graphical abstract
A higher resolution version of the Graphical abstract is available as Supplementary information



Similar content being viewed by others
References
Grisaru S (2014) Management of hemolytic-uremic syndrome in children. Int J Nephrol Renovasc Dis 7:231–239. https://doi.org/10.2147/IJNRD.S41837
Tarr PI, Gordon CA, Chandler WL (2005) Shiga-toxin-producing Escherichia coli and haemolytic uraemic syndrome. Lancet 365:1073–1086. https://doi.org/10.1016/S0140-6736(05)71144-2
Walsh PR, Johnson S (2018) Treatment and management of children with haemolytic uraemic syndrome. Arch Dis Child 103:285–291. https://doi.org/10.1136/archdischild-2016-311377
Brocklebank V, Wood KM, Kavanagh D (2018) Thrombotic microangiopathy and the kidney. Clin J Am Soc Nephrol 13:300–317. https://doi.org/10.2215/CJN.00620117
Percheron L, Gramada R, Tellier S, Salomon R, Harambat J, Llanas B, Fila M, Allain-Launay E, Lapeyraque AL, Leroy V, Adra AL, Bérard E, Bourdat-Michel G, Chehade H, Eckart P, Merieau E, Piètrement C, Sellier-Leclerc AL, Frémeaux-Bacchi V, Dimeglio C, Garnier A (2018) Eculizumab treatment in severe pediatric STEC-HUS: a multicenter retrospective study. Pediatr Nephrol 33:1385–1394. https://doi.org/10.1007/s00467-018-3903-9
Keir LS, Saleem MA (2014) Current evidence for the role of complement in the pathogenesis of Shiga toxin haemolytic uraemic syndrome. Pediatr Nephrol 29:1895–1902. https://doi.org/10.1007/s00467-013-2561-1
Lynn RM, O’Brien SJ, Taylor CM, Adak GK, Chart H, Cheasty T, Coia JE, Gillespie IA, Locking ME, Reilly WJ, Smith HR, Waters A, Willshaw GA (2005) Childhood hemolytic uremic syndrome, United Kingdom and Ireland. Emerg Infect Dis 11:590–596. https://doi.org/10.3201/EID1104.040833
Poolpol K, Orth-Höller D, Speth C, Zipfel PF, Skerka C, de Córdoba SR, Brockmeyer J, Bielaszewska M, Würzner R (2014) Interaction of Shiga toxin 2 with complement regulators of the factor H protein family. Mol Immunol 58:77–84. https://doi.org/10.1016/j.molimm.2013.11.009
Spinale JM, Ruebner RL, Copelovitch L, Kaplan BS (2013) Long-term outcomes of Shiga toxin hemolytic uremic syndrome. Pediatr Nephrol 28:2097–2105. https://doi.org/10.1007/s00467-012-2383-6
Gerber A, Karch H, Allerberger F, Verweyen HM, Zimmerhackl LB (2002) Clinical course and the role of shiga toxin-producing Escherichia coli infection in the hemolytic-uremic syndrome in pediatric patients, 1997–2000, in Germany and Austria: a prospective study. J Infect Dis 186:493–500. https://doi.org/10.1086/341940
Loirat C, Fakhouri F, Ariceta G, Besbas N, Bitzan M, Bjerre A, Coppo R, Emma F, Johnson S, Karpman D, Landau D, Langman CB, Lapeyraque AL, Licht C, Nester C, Pecoraro C, Riedl M, van de Kar NC, Van de Walle J, Vivarelli M, Frémeaux-Bacchi V; HUS International (2016) An international consensus approach to the management of atypical hemolytic uremic syndrome in children. Pediatr Nephrol 31:15–39. https://doi.org/10.1007/s00467-015-3076-8
Kim Y, Miller K, Michael AF (1977) Breakdown products of C3 and factor B in hemolytic-uremic syndrome. J Lab Clin Med 89:845–850. https://doi.org/10.5555/uri:pii:0022214377900713
Orth-Höller D, Riedl M, Würzner R (2011) Inhibition of terminal complement activation in severe Shiga toxin-associated HUS—perfect example for a fast track from bench to bedside. EMBO Mol Med 3:617–619. https://doi.org/10.1002/EMMM.201100169
Orth-Höller D, Würzner R (2014) Role of complement in enterohemorrhagic Escherichia coli-Induced hemolytic uremic syndrome. Semin Thromb Hemost 40:503–507. https://doi.org/10.1055/S-0034-1375295
Orth D, Khan AB, Naim A, Grif K, Brockmeyer J, Karch H, Joannidis M, Clark SJ, Day AJ, Fidanzi S, Stoiber H, Dierich MP, Zimmerhackl LB, Würzner R (2009) Shiga toxin activates complement and binds factor H: evidence for an active role of complement in hemolytic uremic syndrome. J Immunol 182:6394–6400. https://doi.org/10.4049/jimmunol.0900151
Kellnerová S, Chatterjee S, Bayarri-Olmos R, Justesen L, Talasz H, Posch W, Kenno S, Garred P, Orth-Höller D, Grasse M, Würzner R (2020) Shiga toxin 2a binds to complement components C3b and C5 and upregulates their gene expression in human cell lines. Toxins (Basel) 13:8. https://doi.org/10.3390/toxins13010008
Arvidsson I, Rebetz J, Loos S, Hertelious M, Kristoffersson AC, Englund E, Chromek M, Karpman D (2016) Early terminal complement blockade and C6 deficiency are protective in enterohemorrhagic Escherichia coli-infected mice. J Immunol 197:1276–1286. https://doi.org/10.4049/jimmunol.1502377
Zoja C, Buelli S, Morigi M (2019) Shiga toxin triggers endothelial and podocyte injury: the role of complement activation. Pediatr Nephrol 34:379–388. https://doi.org/10.1007/s00467-017-3850-x
Ehrlenbach S, Rosales A, Posch W, Wilflingseder D, Hermann M, Brockmeyer J, Karch H, Satchell SC, Würzner R, Orth-Höller D (2013) Shiga toxin 2 reduces complement inhibitor CD59 expression on human renal tubular epithelial and glomerular endothelial cells. Infect Immun 81:2678–2685. https://doi.org/10.1128/IAI.01079-12
Balestracci A, Meni Bataglia L, Toledo I, Beaudoin L, Alvarado C (2020) C3 levels and acute outcomes in Shiga toxin–related hemolytic uremic syndrome. Pediatr Nephrol 35:331–339. https://doi.org/10.1007/s00467-019-04334-3
Netti GS, Santangelo L, Paulucci L, Piscopo G, Torres DD, Carbone V, Giordano P, Spadaccino F, Castellano G, Stallone G, Gesualdo L, Chironna M, Ranieri E, Giordano M (2020) Low C3 serum levels predict severe forms of STEC-HUS with neurologic involvement. Front Med 7:357. https://doi.org/10.3389/fmed.2020.00357
Lapeyraque AL, Malina M, Fremeaux-Bacchi V, Boppel T, Kirschfink M, Oualha M, Proulx F, Clermont MJ, Le Deist F, Niaudet P, Schaefer F (2011) Eculizumab in Severe Shiga-Toxin–Associated HUS. N Eng J Med 364:2561–2563. https://doi.org/10.1056/nejmc1100859
Pape L, Hartmann H, Bange Christoph Bange F, Suerbaum S, Bueltmann E, Ahlenstiel-Grunow T (2015) Eculizumab in Typical Hemolytic Uremic Syndrome (HUS) With Neurological Involvement. Medicine 94:e1000. https://doi.org/10.1097/MD.0000000000001000
Kielstein JT, Beutel G, Fleig S, Steinhoff J, Meyer TN, Hafer C, Kuhlmann U, Bramstedt J, Panzer U, Vischedyk M, Busch V, Ries W, Mitzner S, Mees S, Stracke S, Nürnberger J, Gerke P, Wiesner M, Sucke B, Abu-Tair M, Kribben A, Klause N, Schindler R, Merkel F, Schnatter S, Dorresteijn EM, Samuelsson O, Brunkhorst R; Collaborators of the DGfN STEC-HUS registry (2012) Best supportive care and therapeutic plasma exchange with or without eculizumab in Shiga-toxin-producing E. coli O104:H4 induced haemolytic-uraemic syndrome: An analysis of the German STEC-HUS registry. Nephrol Dial Transplant 27:3807–3815. https://doi.org/10.1093/ndt/gfs394
Walsh PR, Johnson S (2019) Eculizumab in the treatment of Shiga toxin haemolytic uraemic syndrome. Pediatr Nephrol 34:1485–1492. https://doi.org/10.1007/s00467-018-4025-0
De S, Waters AM, Segal AO, Trautmann A, Harvey EA, Licht C (2010) Severe atypical HUS caused by CFH S1191L-case presentation and review of treatment options. Pediatr Nephrol 25:97–104. https://doi.org/10.1007/s00467-009-1306-7
Heinen S, Sanchez-Corral P, Jackson MS, Strain L, Goodship JA, Kemp EJ, Skerka C, Jokiranta TS, Meyers K, Wagner E, Robitaille P, Esparza-Gordillo J, Rodriguez de Cordoba S, Zipfel PF, Goodship TH (2006) De novo gene conversion in the RCA gene cluster (1q32) causes mutations in complement factor H associated with atypical hemolytic uremic syndrome. Hum Mutat 27:292–293. https://doi.org/10.1002/humu.9408
Frémeaux-Bacchi V, Sellier-Leclerc AL, Vieira-Martins P, Limou S, Kwon T, Lahoche A, Novo R, Llanas B, Nobili F, Roussey G, Cailliez M, Ulinski T, Deschênes G, Alberti C, Weill FX, Mariani P, Loirat C (2019) Complement gene variants and shiga toxin-producing escherichia coli-associated hemolytic uremic syndrome retrospective genetic and clinical study. Clin J Am Soc Nephrol 14:364–377. https://doi.org/10.2215/CJN.05830518
Morigi M, Galbusera M, Gastoldi S, Locatelli M, Buelli S, Pezzotta A, Pagani C, Noris M, Gobbi M, Stravalaci M, Rottoli D, Tedesco F, Remuzzi G, Zoja C (2011) Alternative pathway activation of complement by Shiga toxin promotes exuberant C3a formation that triggers microvascular thrombosis. J Immunol 187:172–180. https://doi.org/10.4049/jimmunol.1100491
Robson WLM, Leung AKC, Fick GH, McKenna AI (1992) Hypocomplementemia and leukocytosis in diarrhea-associated hemolytic uremic syndrome. Nephron 62:296–299. https://doi.org/10.1159/000187063
Thurman JM, Marians R, Emlen W, Wood S, Smith C, Akana H, Holers VM, Lesser M, Kline M, Hoffman C, Christen E, Trachtman H (2009) Alternative pathway of complement in children with diarrhea-associated hemolytic uremic syndrome. Clin J Am Soc Nephrol 4:1920–1924. https://doi.org/10.2215/CJN.02730409
Ferraris JR, Ferraris V, Acquier AB, Sorroche PB, Saez MS, Ginaca A, Mendez CF (2015) Activation of the alternative pathway of complement during the acute phase of typical haemolytic uraemic syndrome. Clin Exp Immunol 181:118–125. https://doi.org/10.1111/cei.12601
Alconcher LF, Balestracci A, Coccia PA, Suarez ADC, Ramírez FB, Monteverde ML, Perez Y Gutiérrez MG, Carlopio PM, Principi I, Estrella P, Micelli S, Leroy DC, Quijada NE, Seminara C, Giordano MI, Hidalgo Solís SB, Saurit M, Caminitti A, Arias A, Liern M, Rivas M (2021) Hemolytic uremic syndrome associated with Shiga toxin-producing Escherichia coli infection in Argentina: update of serotypes and genotypes and their relationship with severity of the disease. Pediatr Nephrol 36:2811–2817. https://doi.org/10.1007/s00467-021-04988-y
Muff-Luett M, Sanderson KR, Engen RM, Zahr RS, Wenderfer SE, Tran CL, Sharma S, Cai Y, Ingraham S, Winnicki E, Weaver DJ, Hunley TE, Kiessling SG, Seamon M, Woroniecki R, Miyashita Y, Xiao N, Omoloja AA, Kizilbash SJ, Mansuri A, Kallash M, Yu Y, Sherman AK, Srivastava T, Nester CM (2021) Eculizumab exposure in children and young adults: indications, practice patterns, and outcomes-a Pediatric Nephrology Research Consortium study. Pediatr Nephrol 36:2349–2360. https://doi.org/10.1007/s00467-021-04965-5
Castañeda-Sanabria J, Hajage D, le Jouan M, Perozziello A, Tubach F (2016) Off-label use of the expensive orphan drug eculizumab in France 2009–2013 and the impact of literature: focus on the transplantation field. Eur J Clin Pharmacol 72:737–746. https://doi.org/10.1007/s00228-016-2027-z
Mahat U, Matar RB, Rotz SJ (2019) Use of complement monoclonal antibody eculizumab in Shiga toxin producing Escherichia coli associated hemolytic uremic syndrome: A review of current evidence. Pediatr Blood Cancer 66:e27913. https://doi.org/10.1002/pbc.27913
Gitiaux C, Krug P, Grevent D, Kossorotoff M, Poncet S, Eisermann M, Oualha M, Boddaert N, Salomon R, Desguerre I (2013) Brain magnetic resonance imaging pattern and outcome in children with haemolytic-uraemic syndrome and neurological impairment treated with eculizumab. Dev Med Child Neurol 55:758–765. https://doi.org/10.1111/DMCN.12161
Menne J, Nitschke M, Stingele R, Abu-Tair M, Beneke J, Bramstedt J, Bremer JP, Brunkhorst R, Busch V, Dengler R, Deuschl G, Fellermann K, Fickenscher H, Gerigk C, Goettsche A, Greeve J, Hafer C, Hagenmüller F, Haller H, Herget-Rosenthal S, Hertenstein B, Hofmann C, Lang M, Kielstein JT, Klostermeier UC, Knobloch J, Kuehbacher M, Kunzendorf U, Lehnert H, Manns MP, Menne TF, Meyer TN, Michael C, Münte T, Neumann-Grutzeck C, Nuernberger J, Pavenstaedt H, Ramazan L, Renders L, Repenthin J, Ries W, Rohr A, Rump LC, Samuelsson O, Sayk F, Schmidt BM, Schnatter S, Schöcklmann H, Schreiber S, von Seydewitz CU, Steinhoff J, Stracke S, Suerbaum S, van de Loo A, Vischedyk M, Weissenborn K, Wellhöner P, Wiesner M, Zeissig S, Büning J, Schiffer M, Kuehbacher T; EHEC-HUS consortium (2012) Validation of treatment strategies for enterohaemorrhagic Escherichia coli O104:H4 induced haemolytic uraemic syndrome: case-control study. BMJ 345:e4565. https://doi.org/10.1136/BMJ.E4565
Raina R, Sethi S, Dragon-Durey M, Khooblall A, Sharma D, Khandelwal P, Shapiro R, Boyer O, Yap HK, Bagga A, Licht C (2022) Systematic review of atypical hemolytic uremic syndrome biomarkers. Pediatr Nephrol 37:1479–1493. https://doi.org/10.1007/s00467-022-05451-2
Noris M, Galbusera M, Gastoldi S, Macor P, Banterla F, Bresin E, Tripodo C, Bettoni S, Donadelli R, Valoti E, Tedesco F, Amore A, Coppo R, Ruggenenti P, Gotti E, Remuzzi G (2014) Dynamics of complement activation in aHUS and how to monitor eculizumab therapy. Blood 124:1715–1726. https://doi.org/10.1182/blood-2014-02-558296
Volokhina EB, Westra D, van der Velden TJAM, van de Kar NCAJ, Mollnes TE, van den Heuvel LP (2015) Complement activation patterns in atypical haemolytic uraemic syndrome during acute phase and in remission. Clin Exp Immunol 181:306–313. https://doi.org/10.1111/CEI.12426
Cataland SR, Holers VM, Geyer S, Yang S, Wu HM (2014) Biomarkers of terminal complement activation confirm the diagnosis of aHUS and differentiate aHUS from TTP. Blood 123:3733–3738. https://doi.org/10.1182/BLOOD-2013-12-547067
Bu F, Zhang Y, Thomas C, Smith RJH (2015) Soluble C5b–9 as a biomarker for complement activation in atypical hemolytic uremic syndrome. Am J Kidney Dis 65:968–969. https://doi.org/10.1053/j.ajkd.2015.02.326
Karnisova L, Hradsky O, Blahova K, Fencl F, Dolezel Z, Zaoral T, Zieg J (2018) Complement activation is associated with more severe course of diarrhea-associated hemolytic uremic syndrome, a preliminary study. Eur J Pediatr 177:1837–1844. https://doi.org/10.1007/S00431-018-3255-2
Palomo M, Blasco M, Molina P, Losano M, Praga M, Torramade-Moix S, Martinez-Sanchez J, Cid J, Escolar G, Carreras E, Paules C, Crispi F, Quintana LF, Poch E, Rodas L, Goma E, Morelle J, Espinosa M, Morales E, Avila A, Cabello V, Ariceta G, Chocron S, Manrique J, Barros X, Martin N, Huerta A, Fraga-Rodriguez GM, Cao M, Martin M, Romera AM, Moreso F, Manonelles A, Gratacos E, Pereira A, Campistol JM, Diaz-Ricart M (2022) Complement activation and thrombotic microangiopathies. Clin J Am Soc Nephrol 14:1719–1732. https://doi.org/10.2215/CJN.05830519
Fakhouri F, Schwotzer N, Golshayan D, Frémeaux-Bacchi V (2022) The Rational Use of Complement Inhibitors in Kidney Diseases. Kidney Int Rep 7:1165–1178. https://doi.org/10.1016/J.EKIR.2022.02.021
Bianchi L, Gaiani F, Vincenzi F, Kayali S, Di Mario F, Leandro G, De' Angelis GL, Ruberto C (2018) Hemolytic uremic syndrome: Differential diagnosis with the onset of inflammatory bowel diseases. Acta Biomed 89:153–157. https://doi.org/10.23750/abm.v89i9-S.7911
de Buys Roessingh AS, de Lagausie P, Baudoin V, Loirat C, Aigrain Y (2007) Gastrointestinal complications of post-diarrheal hemolytic uremic syndrome. Eur J Pediatr Surg 17:328–334. https://doi.org/10.1055/s-2007-965013
Dragon-Durey MA, Sethi SK, Bagga A, Blanc C, Blouin J, Ranchin B, André JL, Takagi N, Cheong HI, Hari P, Le Quintrec M, Niaudet P, Loirat C, Fridman WH, Frémeaux-Bacchi V (2010) Clinical features of anti-factor H autoantibody-associated hemolytic uremic syndrome. J Am Soc Nephrol 21:2180–2187. https://doi.org/10.1681/ASN.2010030315
Herbert AP, Kavanagh D, Johansson C, Morgan HP, Blaum BS, Hannan JP, Barlow PN, Uhrín D (2012) Structural and functional characterization of the product of disease-related factor H gene conversion. Biochemistry 51:1874–1884. https://doi.org/10.1021/BI201689J
Venables JP, Strain L, Routledge D, Bourn D, Powell HM, Warwicker P, Diaz-Torres ML, Sampson A, Mead P, Webb M, Pirson Y, Jackson MS, Hughes A, Wood KM, Goodship JA, Goodship TH (2006) Atypical haemolytic uraemic syndrome associated with a hybrid complement gene. PLoS Med 3:1957–1967. https://doi.org/10.1371/journal.pmed.0030431
Krid S, Roumenina LT, Beury D, Charbit M, Boyer O, Frémeaux-Bacchi V, Niaudet P (2012) Renal transplantation under prophylactic eculizumab in atypical hemolytic uremic syndrome with CFH/CFHR1 hybrid protein. Am J Transplant 12:1938–1944. https://doi.org/10.1111/j.1600-6143.2012.04051.x
Bouwmeester RN, van de Kar NCAJ, Wetzels JFM (2021) Enough is enough: targeted eculizumab withdrawal in atypical hemolytic uremic syndrome. Kidney Int 100:265–268. https://doi.org/10.1016/j.kint.2021.02.033
Acosta-Medina A, Moyer A, Go R, Willrich MA, Alkhateeb HB, Chen D, Heikal NM, Leung N, Marshall AL, Tran CL, Winters JL, Sridharan M (2020) Determination of relapse risk by complement gene variants after eculizumab discontinuation in complement-mediated thrombotic microangiopathy: a retrospective review. Blood 136(Suppl 1):25–26. https://doi.org/10.1182/blood-2020-136617
Acknowledgements
Carla Galvez is a resident fellow of the Pediatric Nephrology Program at the Faculty of Medicine at Universidad de Chile. The genetic CFH/CFI analysis was funded by the Grant FICR19-20 from the Gobierno Regional de la Región de Los Ríos.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Galvez, C., Krall, P., Rojas, A. et al. HUS with mutations in CFH and STEC infection treated with eculizumab in a 4-year-old girl. Pediatr Nephrol 38, 1195–1203 (2023). https://doi.org/10.1007/s00467-022-05694-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-022-05694-z