Abstract
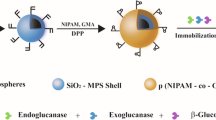
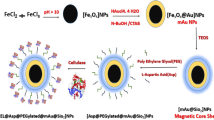
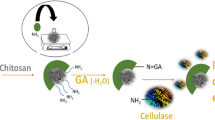
Immobilization of cellulases on magnetic nanoparticles, especially magnetite nanoparticles, has been the main approach studied to make this enzyme, economically and industrially, more attractive. However, magnetite nanoparticles tend to agglomerate, are very reactive and easily oxidized in air, which has strong impact on their useful life. Thus, it is very important to provide proper surface coating to avoid the mentioned problems. This study aimed to investigate the immobilization of cellulase on magnetic nanoparticles encapsulated in polymeric nanospheres. The support was characterized in terms of morphology, average diameter, magnetic behavior and thermal decomposition analyses. The polymer nanospheres containing encapsulated magnetic nanoparticles showed superparamagnetic behavior and intensity average diameter about 150 nm. Immobilized cellulase exhibited broader temperature stability than in the free form and great reusability capacity, 69% of the initial enzyme activity was maintained after eight cycles of use. The magnetic support showed potential for cellulase immobilization and allowed fast and easy biocatalyst recovery through a single magnet.










Similar content being viewed by others
References
Bhat MK (2000) Cellulases and related enzymes in biotechnology. Biotechnol Adv 18:355–383
Cavaco-Paulo A (1998) Mechanism of cellulase action in textile processes. Carbohydr Polym 37:273–277
Chiaradia V, Valério A, de Oliveira D, Araújo PHH, Sayer C (2016) Simultaneous single-step immobilization of Candida antarctica lipase B and incorporation of magnetic nanoparticles on poly(urea-urethane) nanoparticles by interfacial miniemulsion polymerization. J Mol Catal B Enzym 131:31–35
Abraham RE, Verma ML, Barrow CJ, Puri M (2014) Suitability of magnetic nanoparticle immobilised cellulases in enhancing enzymatic saccharification of pretreated hemp biomass. Biotechnol Biofuels 7:1–12
Sahoo B, Sahu SK, Pramanik P (2011) A novel method for the immobilization of urease on phosphonate grafted iron oxide nanoparticle. J Mol Catal B Enzym 69:95–102
Wei W, Zhaohui W, Taekyung Y, Changzhong J, Woo-Sik K (2015) Recent progress on magnetic iron oxide nanoparticles: synthesis, surface functional strategies and biomedical applications. Sci Technol Adv Mater 16:023501
Liao H, Chen D, Yuan L, Zheng M, Zhu Y, Liu X (2010) Immobilized cellulase by polyvinyl alcohol/Fe2O3 magnetic nanoparticle to degrade microcrystalline cellulose. Carbohydr Polym 82:600–604
Khoshnevisan K, Bordbar A-K, Zare D, Davoodi D, Noruzi M, Barkhi M, Tabatabaei M (2011) Immobilization of cellulase enzyme on superparamagnetic nanoparticles and determination of its activity and stability. Chem Eng J 171(2):669–673
Jordan J, Kumar CSSR, Theegala C (2011) Preparation and characterization of cellulase-bound magnetite nanoparticles. J Mol Catal B Enzym 68:139–146
Zang L, Qiu J, Wu X, Zhang W, Sakai E, Wei Y (2014) Preparation of magnetic chitosan nanoparticles as support for cellulase immobilization. Ind Eng Chem Res 53:3448–3454
Zhang W, Qiu J, Feng H, Zang L, Sakai E (2015) Increase in stability of cellulase immobilized on functionalized magnetic nanospheres. J Magn Magn Mater 375:117–123
Lu A-H, Salabas EL, Schüth F (2007) Magnetic nanoparticles: synthesis, protection, functionalization, and application. Angew Chem Int Ed 46:1222–1244
Mahdavi M, Ahmad M, Haron M, Namvar F, Nadi B, Rahman M, Amin J (2013) Synthesis, surface modification and characterisation of biocompatible magnetic iron oxide nanoparticles for biomedical applications. Molecules 18:7533
Chiaradia V, Soares NS, Valério A, de Oliveira D, Araújo PHH, Sayer C (2016) Immobilization of Candida antarctica lipase B on magnetic poly(urea-urethane) nanoparticles. Appl Biochem Biotechnol 1:1–18
Cunha AG, Besteti MD, Manoel EA, da Silva AAT, Almeida RV, Simas ABC, Fernandez-Lafuente R, Pinto JC, Freire DMG (2014) Preparation of core–shell polymer supports to immobilize lipase B from Candida antarctica: effect of the support nature on catalytic properties. J Mol Catal B Enzym 100:59–67
Li S, Hu J, Liu B (2004) Use of chemically modified PMMA microspheres for enzyme immobilization. Biosystems 77:25–32
Valério A, Nicoletti G, Cipolatti EP, Ninow JL, Araújo PHH, Sayer C, de Oliveira D (2015) Kinetic study of Candida antarctica lipase B immobilization using poly(methyl methacrylate) nanoparticles obtained by miniemulsion polymerization as support. Appl Biochem Biotechnol 175:2961–2971
Feuser PE, Bubniak LdS, Silva MCdS, Viegas AdC, Castilho Fernandes A, Ricci-Junior E, Nele M, Tedesco AC, Sayer C, de Araújo PHH (2015) Encapsulation of magnetic nanoparticles in poly(methyl methacrylate) by miniemulsion and evaluation of hyperthermia in U87MG cells. Eur Polymer J 68:355–365
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cullity BD, Graham CD (2011) Introduction to magnetic materials. Wiley, Oxford
Zheng W, Gao F, Gu H (2005) Magnetic polymer nanospheres with high and uniform magnetite content. J Magn Magn Mater 288:403–410
Landfester K, Ramírez LP (2003) Encapsulated magnetite particles for biomedical application. J Phys Condens Matter 15:S1345
Neunzert H, Siddiqi AH (2013) Topics in industrial mathematics: case studies and related mathematical methods. Springer, US
Hirsh SL, Bilek MMM, Nosworthy NJ, Kondyurin A, dos Remedios CG, McKenzie DR (2010) A comparison of covalent immobilization and physical adsorption of a cellulase enzyme mixture. Langmuir 26:14380–14388
Linder M, Mattinen ML, Kontteli M, Lindeberg G, Ståhlberg J, Drakenberg T, Reinikainen T, Pettersson G, Annila A (1995) Identification of functionally important amino acids in the cellulose-binding domain of Trichoderma reesei cellobiohydrolase I. Protein Sci Publ Protein Soc 4:1056–1064
Xu J, Huo S, Yuan Z, Zhang Y, Xu H, Guo Y, Liang C, Zhuang X (2011) Characterization of direct cellulase immobilization with superparamagnetic nanoparticles. Biocatal Biotransform 1:29–35
Bandekar J (1992) Amide modes and protein conformation. Biochim Biophys Acta (BBA) Protein Struct Mol Enzymol 1120:123–143
Mishra A, Sardar M (2015) Cellulase assisted synthesis of nano-silver and gold: application as immobilization matrix for biocatalysis. Int J Biol Macromol 77:105–113
Zhang D, Hegab HE, Lvov Y, Dale Snow L, Palmer J (2016) Immobilization of cellulase on a silica gel substrate modified using a 3-APTES self-assembled monolayer. SpringerPlus 5:1–20
Yu Y, Yuan J, Wang Q, Fan X, Wang P, Cui L (2015) Noncovalent immobilization of cellulases using the reversibly soluble polymers for biopolishing of cotton fabric. Biotechnol Appl Biochem 62:494–501
Acknowledgements
The authors would like to thank Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for financial support as well as Laboratório Central de Microscopia Eletrônica da UFSC (LCME-UFSC) for TEM images.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lima, J.S., Araújo, P.H.H., Sayer, C. et al. Cellulase immobilization on magnetic nanoparticles encapsulated in polymer nanospheres. Bioprocess Biosyst Eng 40, 511–518 (2017). https://doi.org/10.1007/s00449-016-1716-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-016-1716-4