Abstract
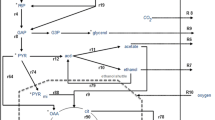
Production of xylitol from xylose in batch fermentations of Candida mogii ATCC 18364 is discussed in the presence of glucose as the cosubstrate. Various initial ratios of glucose and xylose concentrations are assessed for their impact on yield and rate of production of xylitol. Supplementation with glucose at the beginning of the fermentation increased the specific growth rate, biomass yield and volumetric productivity of xylitol compared with fermentation that used xylose as the sole carbon source. A mathematical model is developed for eventual use in predicting the product formation rate and yield. The model parameters were estimated from experimental observations, using a genetic algorithm. Batch fermentations, which were carried out with xylose alone and a mixture of xylose and glucose, were used to validate the model. The model fitted well with the experimental data of cell growth, substrate consumption and xylitol production.



Similar content being viewed by others
Abbreviations
- a cell :
-
Specific surface area of the cell (m2 g DCW−1)
- C glc :
-
Glucose concentration (g glucose l−1)
- C x :
-
Biomass concentration (g DCW l−1)
- C exxit :
-
Extracellular concentration of xylitol (g xylitol l−1)
- C inxit :
-
Intracellular concentration of xylitol (g xylitol l cell volume−1)
- C xyl :
-
Xylose concentration (g xylose l−1)
- d mem :
-
Thickness of the cell membrane (m)
- D mem :
-
Diffusion coefficient of xylitol in the lipid bilayer (m2 s−1)
- HMP:
-
Hexose monophosphate
- K i, glc :
-
Inhibition constant by glucose (g glucose l−1)
- K i, xyl :
-
Inhibition constant by xylose (g xylose l−1)
- K s, glc :
-
Saturation constant based on glucose (g glucose l−1)
- K s, xit :
-
Saturation constant based on xylitol (g xylitol l−1)
- K s, xyl :
-
Saturation constant based on xylose (g xylose l−1)
- K par :
-
Partition coefficient (-)
- K r :
-
Repression constant by glucose (g glucose l−1)
- min J :
-
Minimum error of the objective function (-)
- M :
-
Number of experimentally observable state variables (-)
- MWxit :
-
Molecular weight of xylitol (g xylitol mole−1)
- MWxyl :
-
Molecular weight of xylose (g xylose mole−1)
- N :
-
Number of sampling points of experimental data (-)
- P :
-
Permeability coefficient (m s−1)
- P xit :
-
Permeability coefficient for xylitol (m s−1)
- q glc :
-
Specific uptake rate of glucose (g glucose g DCW-1 h−1)
- q maxglc :
-
Maximum specific uptake rate of glucose (g glucose g DCW−1 h−1)
- q xit :
-
Specific production rate of xylitol (g xylitol g DCW−1 h−1)
- q xyl :
-
Specific uptake rate of xylose (g xylose g DCW−1 h−1)
- q maxxyl :
-
Maximum specific uptake rate of xylose (g xylose g DCW−1 h−1)
- Q xit :
-
Volumetric production rate of xylitol (g xylitol l−1 h−1)
- Q xyl :
-
Volumetric uptake rate of xylose (g xylose l−1 h−1)
- r f,xit :
-
Specific rate of formation of xylitol (g xylitol g DCW−1 h−1)
- r u,xit :
-
Consumption rate of intracellular xylitol (g xylitol g DCW−1 h−1)
- r t,xit :
-
Mass flux of xylitol based on dry cell weight (g xylitol g DCW−1 h−1)
- t :
-
Cultivation time (h)
- W i :
-
Weighting coefficient (-)
- y ij :
-
Experimental value of variable (-)
- \( \ifmmode\expandafter\hat\else\expandafter\^\fi{y}_{ij} \) :
-
Model predicted value of variable (-)
- Y x/xit :
-
Biomass yield on xylitol (g DCW g xylitol−1)
- Y x/(xyl+glc) :
-
Biomass yield on xylose and glucose (g DCW g sugar−1)
- Y xit/xyl :
-
Xylitol yield on xylose (g xylitol g xylose−1)
- μ:
-
Specific growth rate (h−1)
- μ maxglc :
-
Maximum specific growth rate on glucose (h−1)
- μ maxxit :
-
Maximum specific growth rate on xylitol (h−1)
- ρ x :
-
Mass density of cells (g DCW l cell−1 volume)
- η:
-
Energy yield coefficient for biomass production (-)
- ξp :
-
Energy yield coefficient for product formation (-)
- σcell :
-
Weight fraction of carbon in biomass (g atom-C g DCW−1)
- σxit :
-
Weight fraction of carbon in xylitol (g atom-C g xylitol−1)
- γcell :
-
Reductance degree of biomass (-)
- γxit :
-
Reductance degree of xylitol (-)
References
Winkelhausen E, Kuzmanova S (1998) Microbial conversion of D-xylose to xylitol. J Ferment Bioeng 86:1–14
Kim JH, Ryu YW, Seo JH (1999) Analysis and optimization of a two-substrate fermentation for xylitol production using Candida tropicalis. J Ind Microbiol Biotechnol 22:181–186
Oh DK, Kim SY (1998) Increase of xylitol yield by feeding xylose and glucose in Candida tropicalis. Appl Microbiol Biotechnol 50:419–425
Tavares JM, Duarte LC, Amaral-Collaço MT, Gírio FM (2000) The influence of hexoses addition on the fermentation of D-xylose in Debaryomyces hansenii under continuous cultivation. Enzyme Microb Technol 26:743–747
Kilian SG, van Uden N (1988) Transport of xylose and glucose in the xylose-fermenting yeast Pichia stipitis. Appl Microbiol Biotechnol 27:545–548
Lee H, Sopher CR, Yau KYF (1996) Induction of xylose reductase and xylitol dehydrogenase activities on mixed sugars in Candida guilliermondii. J Chem Technol Biotechnol 66:375–379
Rosa SMA, Felipe MGA, Silva SS, Vitolo M (1998) Xylose reductase production by Candida guilliermondii. Appl Biochem Biotechnol 70/72:127–135
Aguiar Jr WB, Faria LFF, Couto MAPG, Araujo OQF, Pereira N Jr (2002) Growth model and prediction of oxygen transfer rate for xylitol production from D-xylose by C. guilliermondii. Biochem Eng J 12:49–59
Aranda-Barradas JS, Delia ML, Riba JP (2000) Kinetic study and modelling of the xylitol production using Candida parapsilosis in oxygen-limited culture conditions. Bioprocess Eng 22:219–225
Sirisansaneeyakul S, Staniszewski S, Rizzi M (1995) Screening of yeasts for production of xylitol form D-xylose. J Ferment Bioeng 80:565–570
Mayerhoff ZDVL, Roberto IC, Silva SS (1998) Production of xylitol by Candida mogii from rice straw hydrolysate. Appl Biochem Biotechnol 70/72:149–159
Baishan F, Hongwen C, Xiaolan X, Ning W, Zongding H (2003) Using genetic algorithms coupling neural networks in a study of xylitol production: medium optimization. Process Biochem 38:979–985
Grootjen DRJ, van der Lans RGJM, Luyben KCAM (1991) Conversion of glucose/xylose mixtures by Pichia stipitis under oxygen-limited conditions. Enzyme Microb Technol 13:648–654
Nakamura Y, Sawada T, Inoue E (2001) Mathematical model for ethanol production from mixed sugars by Pichia stipitis. J Chem Technol Biotechnol 76:586–592
Bisswanger H (2002) Enzyme kinetics: principles and methods. Wiley-VCH, Weinheim
Ligthelm ME, Prior BA, du Preez JC, Brandt V (1988) An investigation of D-{1−13C} xylose metabolism in Pichia stipitis under aerobic and anaerobic conditions. Appl Microbiol Biotechnol 28:293–296
Walker GM (1998) Yeast physiology and biotechnology. Wiley, Chichester
Stephanopoulos GN, Aristidou AA, Nielsen J (1998) Metabolic engineering: principles and methodologies. Academic, San Diego
Deschatelets L, Yu EKC (1986) A simple pentose assay for biomass conversion studies. Appl Microbiol Biotechnol 24:379–397
Adler L, Gustafsson L (1980) Polyhydric alcohol production and intracellular amino acid pool in relation to halotolerance of the yeast Debaromyces hansenii. Arch Microbiol 124:123–130
Houck CR, Joines JA, Kay MG (1995) A genetic algorithm for function optimization: a matlab implementation. Technical Report NCSU-IE Technical Report 95–09, North Carolina State University
Lee H (1992) Reversible inactivation of D-xylose utilization by D-glucose in the pentose-fermenting yeast Pachysolen tannophilus. FEMS Microbiol Lett 92:1–4
Jeppsson H, Holmgren K, Hahn-Hägerdal B (1999) Oxygen-dependent xylitol metabolism in Pichia stipitis. Appl Microbiol Biotechnol 53:92–97
Leathers TD, Gupta SC (1997) Xylitol and riboflavin accumulation in xylose-grown cultures of Pichia guilliermondii. Appl Microbiol Biotechnol 47:58–61
Barbosa MFS, de Medeiros MB, Mancilha IM, Schneider H, Lee H (1988) Screening of yeasts for production of xylitol from D-xylose and some factors which affect xylitol yield in Candida guilliermondii. J Ind Microbiol 3:241–251
Sirisansaneeyakul S (1993) Reaktionstechnische Untersuchungen zur mikrobiellen Produktion von Xylit. PhD Thesis, Universitaet Stuttgart, Stuttgart
Kocková-Kratochvílová A (1990) Yeasts and yeast-like organisms. VCH, New York
Voleskey B, Votruba J (1992) Modeling and optimization of fermentation processes. Elsevier, Amsterdam
Roels JA (1983) Energetics and kinetics in biotechnology. Elsevier, Amsterdam
Lucas C, van Uden N (1986) Transport of hemicellulose monomers in the xylose-fermenting yeast Candida shehatae. Appl Microbiol Biotechnol 23:491–495
Nobre A, Lucas C, Leăo C (1999) Transport and utilization of hexoses and pentoses in the halotolerant yeast Debaryomyces hansenii. Appl Environ Microbiol 65:3594–3598
Rogers HJ, Perkins HR (1969) Cell walls and membranes. Spon, London
Jain MK (1972) The biomolecular lipid membrane. Litton, New York
Acknowledgement
This research was supported by the Commission on Higher Education, Ministry of Education, Royal Thai Government, under the Agro-industry PhD Program Consortium.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tochampa, W., Sirisansaneeyakul, S., Vanichsriratana, W. et al. A model of xylitol production by the yeast Candida mogii . Bioprocess Biosyst Eng 28, 175–183 (2005). https://doi.org/10.1007/s00449-005-0025-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-005-0025-0