Abstract
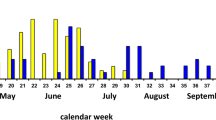
Hybridization leading to reproductively isolated, novel genotypes is poorly understood as a means of speciation and few empirical examples have been studied. In 1999, a previously non-existent delayed flight of what appeared to be the Canadian tiger swallowtail butterfly, Papilio canadensis, was observed in the Battenkill River Valley, USA. Allozyme frequencies and morphology suggest that this delayed flight was the product of hybridization between Papilio canadensis and its sibling species Papilio glaucus. The mitochondrial DNA (mtDNA) restriction fragment length polymorphisms presented here indicate that only P. canadensis-like mtDNA occurs in this population, suggesting that introgression likely occurred from hybrid males mating with P. canadensis females. Preliminary studies of this population indicated that delayed post-diapause pupal emergence in this hybrid genotype was the root cause behind the observed delayed flight, which suggests a potential empirical example of a mechanism leading to reproductive isolation. Here we provide further evidence of the role of adult pupal emergence as a reproductive barrier likely leading to reproductive isolation. In particular, we present results from pupal emergence studies using four different spring and two different winter temperature treatments. The results indicate a clear separation of adult emergences between the hybrid population and both parental species. However, our results indicate that exceptionally hot springs are likely to lead to greater potential for overlap between the local parental species, P. canadensis, and this delayed population with hybrid origins. Conversely, our results also show that warmer winters are likely to increase the temporal separation of the hybrid population and the parental species. Finally, we report recently collected evidence that this hybrid population remains morphologically distinct.



Similar content being viewed by others
References
Abbot P, Withgott JH (2004) Phylogenetic and molecular evidence for allochronic speciation in gall-forming aphids (Pemphigus). Evolution 58:539–553
Abrahamson WG, Blair CP (2008) Sequential radiation through host-race formation: herbivore diversity leads to diversity in natural enemies. In: Tilmon KJ (ed) Specialization, speciation, and radiation: the evolutionary biology of herbivorous insects. University of California Press, Berkley, pp 188–202
Alexander RD, Bigelow RS (1960) Allochronic speciation in field crickets and a new species, Acheta veletis. Evolution 14:334–346
Andolfatto P, Scriber JM, Charlesworth B (2003) No association between mitochondrial DNA haplotypes and a female-limited mimicry phenotype in Papilio glaucus. Evolution 57:305–316
Berlocher SH, Feder JL (2002) Sympatric speciation in phytophagous insects: moving beyond controversy? Annu Rev Entomol 47:773–815
Blair CP, Abrahamson WG, Jackman JA, Tyrrell L (2005) Cryptic speciation and host race formation in a purportedly generalist tumbling flower beetle. Evolution 59:304–316
Bush GL (1994) Sympatric speciation in animals: new wine in old bottles. Trends Ecol Evol 9:285–288
Carling MD, Brumfield RT (2009) Haldane’s rule in an avian system: using cline theory and divergence population genetics to test for differential introgression of mitochondrial, autosomal, and sex-linked loci across the passerine bunting hybrid zone. Evolution 10:2600–2615
Carroll SP, Boyd C (1992) Host race radiation in the soapberry bug: natural history with the history. Evolution 46:1052–1069
Caterino MS, Sperling FAH (1999) Papilio phylogeny based on mitochondrial cytochrome oxidase I and II genes. Mol Phylogenet Evol 11:122–137
Cooley JR, Simon CS, Marshall DC, Slon K, Ehrhardt C (2001) Allochronic speciation, secondary contact, and reproductive character displacement in periodical cicadas (Hemiptera: Magicicada spp.): genetic, morphological, and behavioural evidence. Mol Ecol 10:661–671
Coyne JA, Orr HA (2004) Speciation. Sinauer, Sunderland
Dopman EB, Perez L, Bogdanowicz SM, Harrison RG (2005) Consequences of reproductive barriers for genealogical discordance in the European corn borer. Proc Natl Acad Sci USA 102:14706–14711
Dowling TE, Secor CL (1997) The role of hybridization and introgression in the diversification of animals. Annu Rev Ecol Syst 28:593–619
Eckenrode CJ, Robbins PS, Andaloro JT (1983) Variations in flight patterns of European corn borer (Lepidoptera: Pyralidae) in New York. Environ Entomol 12:393–396
Emalianov I, Simpson F, Narang P, Mallet J (2003) Host choice promotes reproductive isolation between host races of the larch budmoth, Zeiraphera diniana. J Evol Biol 16:208–218
Feder JL, Roethele JB, Filchak K, Niedbalski J, Romero-Severson J (2003) Evidence for inversion polymorphism related to sympatric host race formation in the apple maggot fly, Rhagoletis pomonella. Genetics 163:939–954
Forbes AA, Powell THQ, Stelinski LL, Smith JJ, Feder JL (2009) Sequential speciation across trophic levels. Science 323:776–779
Funk DJ, Nosil P (2008) Comparative analyses of ecological speciation. In: Tilmon KJ (ed) Specialization, speciation, and radiation: the evolutionary biology of herbivorous insects. University of California Press, Berkeley, pp 117–135
Futuyma DJ (2008) Ecology, speciation, and adaptive radiation: the long view. Evolution 62(9):2446–2449
Glover TJ, Robbins PS, Eckenrode CJ, Roelofs WL (1992) Genetic control of voltinism characteristics in European corn borer races assessed with a marker gene. Arch Insect Biochem 20:107–117
Gompert Z, Fordyce JA, Forister ML, Shapiro AM, Nice CC (2006) Homoploid hybrid speciation in an extreme habitat. Science 314:1923–1925
Gompert Z, Forister ML, Fordyce JA, Nice CC (2008) Widespread mito-nuclear discordance with evidence for introgressive hybridization ands selective sweeps in Lycaedes. Mol Ecol 17:5231–5244
Hagen RH, Lederhouse RC (1985) Polymodal emergence of the tiger swallowtail, Papilio glaucus (Lepidoptera: Papilionidae): source of a false second generation in central New York State. Ecol Entomol 10:19–28
Hagen RH, Scriber JM (1989) Sex linked diapause, color, and allozyme loci in Papilio glaucus: linkage analysis and significance in a hybrid zone. Heredity 80:179–185
Hagen RH, Lederhouse RC, Bossart JL, Scriber JM (1991) Papilio canadensis and P. glaucus (Papilionidae) are distinct species. J Lep Soc 45:245–258
Harrison RG, Bogdanowicz SM (1995) Mitochondrial DNA phylogeny of North American field crickets: perspectives on the evolution of life cycles, songs, and habitat associations. J Evol Biol 8:209–232
Lederhouse RC, Ayres MP, Scriber JM (1990) Adult nutrition affects male virility in Papilio glaucus L. Ecology 72:1428–1435
Luebke HJ, Scriber JM, Yandell BS (1988) Use of multivariate discriminant analysis of male wing morphometrics to delineate the Wisconsin hybrid zone for Papilio glaucus glaucus and P. g. canadensis. Am Midl Nat 119:366–379
Mavarez J, Salazar CA, Bermingham E, Salcedo C, Jiggins CD, Linares M (2006) Speciation by hybridization in Heliconius butterflies. Nature 441:868–871
McMillan WO, Jiggins CD, Mallet J (1997) What initiates speciation in passion-vine butterflies? Proc Natl Acad Sci USA 94:8628–8633
Mercader RJ, Scriber JM (2007) Diversification of host use in two polyphagous butterflies: differences in oviposition specificity or host rank hierarchy? Entomol Exp Appl 125:89–101
Mercader RJ, Scriber JM (2008) Asymmetrical thermal constraints on the parapatric species boundaries of two widespread generalist butterflies. Ecol Entomol 33:537–545
Mercader RJ, Aardema ML, Scriber JM (2009) Hybridization leads to host use divergence in a polyphagous butterfly sibling species pair. Oecologia 158:65–662
Morbey YE, Ydenberg RC (2001) Protandrous arrival timing to breeding areas: a review. Ecol Lett 4:663–673
Mullen SP, Dopman EB, Harrison RG (2008) Hybrid zone origins, species boundaries, and the evolution of wing-pattern diversity in a polytypic species complex of North American Admiral butterflies (Nymphalidae: Limenitis). Evolution 62:1401–1417
Nosil P, Vines TH, Funk DJ (2005) Perspective: reproductive isolation caused by natural selection against immigrants from divergent habitats. Evolution 59:705–719
Nosil P, Funk DJ, Ortiz-Barrientos D (2009) Divergent selection and heterogeneous genomic divergence. Mol Ecol 18:375–402
Ording GJ (2008) An analysis of climate induced hybrid speciation in tiger swallowtail butterflies (Papilio). PhD dissertation, Michigan State University
Pavulaan H, Wright DM (2002) Pterourus appalachiensis (Papilionidae: Papilioninae), a new swallowtail butterfly from the Appalachian region of the United States. Taxonomic Rep 3:1–20
Putnam AS, Scriber JM, Andolfatto P (2007) Discordant divergence times among Z chromosome regions between two ecologically distinct swallowtail butterfly species. Evolution 61:912–927
Rockey SJ, Hainze JH, Scriber JM (1987) Evidence of a sex-linked diapause response in Papilio glaucus subspecies and their hybrids. Physiol Entomol 12:181–184
Roelofs W, Glover T, Tang XH, Streng I, Robbins P, Eckenrode C, Lofstedt C, Bengtsson B (1987) Sex pheromone production and perception in European corn borer moths is determined by both autosomal and sex-linked genes. Proc Natl Acad Sci USA 84:7585–7589
Roelofs WL, Liu W, Hao G, Jiao H, Rooney AP, Linn CE Jr (2002) Evolution of moth sex pheromones via ancestral genes. Proc Natl Acad Sci USA 99:13621–13626
Rothschild W, Jordan K (1906) A revision of the American Papilios. Nov Zool 13:411–752
Rundle HD, Nosil P (2005) Ecological speciation. Ecol Lett 8:336–352
Santos H, Rousselet J, Magnoux E, Paiva MR, Branco M, Kerdelhue C (2007) Genetic isolation through time: allochronic differentiation of a phenotypically atypical population of the pine processionary moth. Proc R Soc Lond Ser B 274:935–941
Schwarz D, Matta BJ, Shakir-Botteri NL, McPheron BA (2005) Host shift to an invasive plant triggers rapid animal hybrid speciation. Nature 436:546–549
Schwartz MD, Ahas R, Aasa A (2006) Onset of spring starting earlier across the Northern Hemisphere. Glob Change Biol 12:343–351
Schwarz D, Shoemaker KD, Botteri NL, McPheron BA (2007) A novel preference for an invasive plant as a mechanism for animal hybrid speciation. Evolution 61:245–256
Scriber JM (1982) Food plants and speciation in the Papilio glaucus group. In: Visser JH, Minks AK (eds) Proceedings of the 5th international symposium on insect plant relationships, PUDOC, Wageningen, pp 307–314
Scriber JM (1990) Interaction of introgression from Papilio glaucus canadensis and diapause in producing ‘spring form’ Eastern tiger swallowtail butterflies, P. glaucus. Gt Lakes Entomol 23:127–138
Scriber JM (1994) Climatic legacies and sex chromosomes: latitudinal patterns of voltinism, diapause size and host-plant selection in 2 species of swallowtail butterflies at their hybrid zone. In: Danks HV (ed) 1. Insect life-cycle polymorphism: theory, evolution and ecological consequences for seasonality and diapause control. Kluwer, Dordrecht, pp 17–133
Scriber JM, Lederhouse RC (1992) The thermal environment as a resource dictating geographic patterns of feeding specialization of insect herbivores. In: Hunter MR, Ohgushi T, Price PW (eds) Effects of resource distribution on animal-plant interactions. Academic Press, New York, pp 429–466
Scriber JM, Ording GJ (2005) Ecological speciation without host plant specialization: possible origins of a recently described cryptic Papilio species (Lepidoptera: Papilionidae). Entomol Exp Appl 115:247–263
Scriber JM, Giebink BL, Snider D (1991) Reciprocal latitudinal clines in oviposition behavior of Papilio glaucus and P. canadensis across the Great Lakes hybrid zone: possible sex-linkage of oviposition preferences. Oecologia 87:360–368
Scriber JM, Hagen RH, Lederhouse RC (1996) Genetics of mimicry in the tiger swallowtail butterflies, Papilio glaucus and P. canadensis. (Lepidoptera: Papilionidae). Evolution 50:222–236
Scriber JM, Stump A, Deering M (2003) Hybrid zone ecology and tiger swallowtail trait clines in North America. In: Boggs C, Watt W, Ehrlich P (eds) Ecology and evolution taking flight: butterflies as model study systems. University of Chicago Press, Chicago, USA, pp 367–391
Scriber JM, Ording GJ, Mercader RJ (2008) Hybrid introgression and parapatric speciation in a hybrid zone. In: Tilmon KJ (ed) Specialization, speciation, and radiation: the evolutionary biology of herbivorous insects. University of California Press, Berkeley, pp 69–87
Seehausen O (2004a) Hybridization and adaptive radiation. Trends Ecol Evol 19:198–207
Seehausen O (2004b) Response to Schilthuizen et al.: Hybridization, rare alleles, and adaptive radiation. Trends Ecol Evol 19:405–406
Simon C, Jiaming T, Dalwadi S, Gregory S, Deniega J, Unuaschi T (2000) Genetic evidence for assortative mating between 13-year cicadas and sympatric “17-year cicadas with 13 year life cycles” provides support for allochronic speciation. Evolution 54:1326–1336
Sperling FAH, Hickey DA (1995) Amplified mitochondrial DNA as a diagnostic marker for species of conifer-feeding Choristoneura (Lepidoptera: Tortricidae). Can Entomol 127:277–288
Stump AD, Sperling FAH, Crim A, Scriber JM (2003) Gene flow between great lakes region populations of the Canadian tiger swallowtail butterfly, Papilio canadensis, near the hybrid zone with P. glaucus (Lepidoptera: Papiliondiae). Gt Lakes Entomol 36:41–53
Tauber CA, Tauber MJ, Nechols JR (1977) Two genes control seasonal isolation in sibling species. Science 197:592–593
R Development Core Team (2008) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna (ISBN 3-900051-07-0)
Thomas Y, Bethenod MT, Pelozuelo L, Frérot B, Bourguet D (2003) Genetic isolation between two sympatric host-plantraces of the European corn borer, Ostrinia nubilalis Hübner. I. Sex pheromone, moth emergence timing, and parasitism. Evolution 57:261–273
Via S, Hawthorne DJ (2002) Genetic architecture of ecological specialization and incipient speciation in divergent races of pea aphids. Am Nat 159:S76–S88
Vines TH, Schluter D (2006) Strong assortative mating between allopatric sticklebacks as a byproduct of adaptation to different environments. Proc R Soc Lond Ser B 273:911–916
Wiklund C (2003) Sexual selection and the evolution of butterfly mating systems. In: Boggs CL, Watt WB, Ehrlich PR (eds) Butterflies: ecology and evolution taking flight. University of Chicago Press, Chicago, pp 67–90
Wood TK, Keese M (1990) Host-plant-induced assortative mating in Enchenopa treehoppers. Evolution 44:619–628
Wood TK, Tilmon KJ, Shantz AB, Harris CK, Pesek J (1999) The role of host plant fidelity in initiating insect race formation. Evol Ecol Res 1:317–332
Xie X, Rull J, Michel AP, Velez S, Forbes AA, Lobo NL, Aluja M, Feder JL (2007) Hawthorne infesting populations of Rhagoletis pomonella in Mexico and speciation mode plurality. Evolution 61:1091–1105
Acknowledgments
This research was supported in part by the College of Natural Science and the Michigan Agricultural Experiment Station (project no. 1644), and the National Science Foundation (DEB-0716683). Thanks are extended to various students and research assistants, including Mark and Jessica Deering, Jen Donovan, Barb Gary, Holly Hereau, Angeline Kosnik, Matt Lehnert, Emily Maher, Michelle Oberlin, Laura Palombi, and Aram Stump. Special thanks to Bill Houtz, James Maudsley, Howard Romack, Harry Pavulaan and David Wright for providing eastern specimens. Drs Jim Smith and Jessica Hellmann provided guidance and lab facilities for mtDNA analyses. Matthew Aardema was supported in part by a Scriber Scholars award in Butterfly Biology and Conservation [Department of Entomology, Michigan State University (MSU)], an MSU Plant Sciences Fellowship, and an National Science Foundation (NSF) research experience for undergraduates (NSF-REU, DEB-0821958) award.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Matthias Schaefer.
Rights and permissions
About this article
Cite this article
Ording, G.J., Mercader, R.J., Aardema, M.L. et al. Allochronic isolation and incipient hybrid speciation in tiger swallowtail butterflies. Oecologia 162, 523–531 (2010). https://doi.org/10.1007/s00442-009-1493-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-009-1493-8