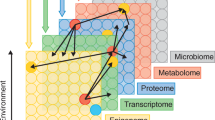
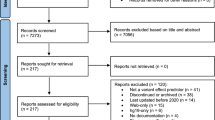
Abstract
Comprehending the molecular basis of quantitative genetic variation is a principal goal for complex diseases or traits. Molecular quantitative trait loci (molQTLs) have made it possible to investigate the effects of genetic variants hiding behind large-scale omics data. A deeper understanding of molQTL is urgently required in light of the multi-dimensionalization of omics data to more fully elucidate the pertinent biological mechanisms. Herein, we reviewed molQTLs with the corresponding resource from the omics perspective and further discussed the integrative strategy of GWAS-molQTL to infer their causal effects. Subsequently, we described the opportunities and challenges encountered by molQTL. The case studies showed that molQTL is essential for complex diseases and traits, whether single- or multi-omics QTLs. Overall, we highlighted the functional significance of genetic variants to employ the discovery of molQTL in complex diseases and traits.




Similar content being viewed by others
Availability of data and materials
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
References
Alasoo K et al (2018) Shared genetic effects on chromatin and gene expression indicate a role for enhancer priming in immune response. Nat Genet 50(3):424–431
Astle WJ et al (2016) The allelic landscape of human blood cell trait variation and links to common complex disease. Cell 167(5):1415–1429
Aznaourova M et al (2022) Single-cell RNA sequencing uncovers the nuclear decoy lincRNA PIRAT as a regulator of systemic monocyte immunity during COVID-19. Proc Natl Acad Sci USA 119(36):e2120680119
Bannister AJ, Kouzarides T (2011) Regulation of chromatin by histone modifications. Cell Res 21(3):381–395
Barbeira AN et al (2018) Exploring the phenotypic consequences of tissue specific gene expression variation inferred from GWAS summary statistics. Nat Commun 9(1):1825
Beckmann ND et al (2020) Multiscale causal networks identify VGF as a key regulator of Alzheimer’s disease. Nat Commun 11(1):3942
Benner C et al (2016) FINEMAP: efficient variable selection using summary data from genome-wide association studies. Bioinformatics 32(10):1493–1501
Bonder MJ et al (2017) Disease variants alter transcription factor levels and methylation of their binding sites. Nat Genet 49(1):131–138
Bruscadin JJ et al (2021) Muscle allele-specific expression QTLs may affect meat quality traits in Bos indicus. Sci Rep 11(1):7321
Byars SG et al (2017) Genetic loci associated with coronary artery disease harbor evidence of selection and antagonistic pleiotropy. PLoS Genet 13(6):e1006328
Chamberlain LH, Shipston MJ (2015) The physiology of protein S-acylation. Physiol Rev 95(2):341–376
Chandra V et al (2021) Promoter-interacting expression quantitative trait loci are enriched for functional genetic variants. Nat Genet 53(1):110–119
Chang L, Zhou G, Xia J (2023) mGWAS-Explorer 20: causal analysis and interpretation of metabolite-phenotype associations. Metabolites 13(7):826
Chen W et al (2015) Fine mapping causal variants with an approximate Bayesian method using marginal test statistics. Genetics 200(3):719–736
Chen L et al (2016) Genetic drivers of epigenetic and transcriptional variation in human immune cells. Cell 167(5):1398–1414
Chen C et al (2022) Pan-cancer analysis of microbiome quantitative trait loci. Cancer Res 82(19):3449–3456
Chun S et al (2017) Limited statistical evidence for shared genetic effects of eQTLs and autoimmune-disease-associated loci in three major immune-cell types. Nat Genet 49(4):600–605
Consortium G.T (2015) Human genomics. The Genotype-Tissue Expression (GTEx) pilot analysis: multitissue gene regulation in humans. Science 348(6235):648–660
Consortium, G.T (2013) The Genotype-Tissue Expression (GTEx) project. Nat Genet 45(6):580–585
Cookson W et al (2009) Mapping complex disease traits with global gene expression. Nat Rev Genet 10(3):184–194
Corces MR et al (2020) Single-cell epigenomic analyses implicate candidate causal variants at inherited risk loci for Alzheimer’s and Parkinson’s diseases. Nat Genet 52(11):1158–1168
Cuddleston WH et al (2022) Cellular and genetic drivers of RNA editing variation in the human brain. Nat Commun 13(1):2997
Cui Y et al (2022) 3’aQTL-atlas: an atlas of 3’UTR alternative polyadenylation quantitative trait loci across human normal tissues. Nucleic Acids Res 50(D1):D39–D45
Currin KW et al (2021) Genetic effects on liver chromatin accessibility identify disease regulatory variants. Am J Hum Genet 108(7):1169–1189
Degner JF et al (2012) DNase I sensitivity QTLs are a major determinant of human expression variation. Nature 482(7385):390–394
Denault WRP, Jugessur A (2021) Detecting differentially methylated regions using a fast wavelet-based approach to functional association analysis. BMC Bioinformatics 22(1):61
Dowell RD (2010) Transcription factor binding variation in the evolution of gene regulation. Trends Genet 26(11):468–475
Draisma HHM et al (2015) Genome-wide association study identifies novel genetic variants contributing to variation in blood metabolite levels. Nat Commun 6:7208
Elkon R, Ugalde AP, Agami R (2013) Alternative cleavage and polyadenylation: extent, regulation and function. Nat Rev Genet 14(7):496–506
Emdin CA, Khera AV, Kathiresan S (2017) Mendelian randomization. JAMA 318(19):1925–1926
Ernst CW, Steibel JP (2013) Molecular advances in QTL discovery and application in pig breeding. Trends Genet 29(4):215–224
Evans DM, Davey Smith G (2015) Mendelian randomization: new applications in the coming age of hypothesis-free causality. Annu Rev Genomics Hum Genet 16:327–350
Farashi S et al (2019) Post-GWAS in prostate cancer: from genetic association to biological contribution. Nat Rev Cancer 19(1):46–59
Fazel-Najafabadi M et al (2022) Discovery and functional characterization of two regulatory variants underlying lupus susceptibility at 2p13.1. Genes (basel) 13(6):1016
Fleige S, Pfaffl MW (2006) RNA integrity and the effect on the real-time qRT-PCR performance. Mol Aspects Med 27(2–3):126–139
Flint J, Mott R (2001) Finding the molecular basis of quantitative traits: successes and pitfalls. Nat Rev Genet 2(6):437–445
Flynn ED et al (2022) Transcription factor regulation of eQTL activity across individuals and tissues. PLoS Genet 18(1):e1009719
Folkersen L et al (2017) Mapping of 79 loci for 83 plasma protein biomarkers in cardiovascular disease. PLoS Genet 13(4):e1006706
Franzen O et al (2018) Global analysis of A-to-I RNA editing reveals association with common disease variants. PeerJ 6:e4466
Gallagher MD, Chen-Plotkin AS (2018) The Post-GWAS Era: from association to function. Am J Hum Genet 102(5):717–730
Gamazon ER et al (2015) A gene-based association method for mapping traits using reference transcriptome data. Nat Genet 47(9):1091–1098
Garieri M et al (2017) The effect of genetic variation on promoter usage and enhancer activity. Nat Commun 8(1):1358
Gaunt TR et al (2016) Systematic identification of genetic influences on methylation across the human life course. Genome Biol 17:61
Geeleher P et al (2018) Cancer expression quantitative trait loci (eQTLs) can be determined from heterogeneous tumor gene expression data by modeling variation in tumor purity. Genome Biol 19(1):130
Giambartolomei C et al (2014) Bayesian test for colocalisation between pairs of genetic association studies using summary statistics. PLoS Genet 10(5):e1004383
Gillies CE et al (2018) An eQTL Landscape of kidney tissue in human nephrotic syndrome. Am J Hum Genet 103(2):232–244
Gong J et al (2018) PancanQTL: systematic identification of cis-eQTLs and trans-eQTLs in 33 cancer types. Nucleic Acids Res 46(D1):D971–D976
Gong J et al (2019) Pancan-meQTL: a database to systematically evaluate the effects of genetic variants on methylation in human cancer. Nucleic Acids Res 47(D1):D1066–D1072
Grishin D, Gusev A (2022) Allelic imbalance of chromatin accessibility in cancer identifies candidate causal risk variants and their mechanisms. Nat Genet 54(6):837–849
Grubert F et al (2015) Genetic control of chromatin states in humans involves local and distal chromosomal interactions. Cell 162(5):1051–1065
Guo X et al (2018) A comprehensive cis-eQTL analysis revealed target genes in breast cancer susceptibility loci identified in genome-wide association studies. Am J Hum Genet 102(5):890–903
Gusev A et al (2016) Integrative approaches for large-scale transcriptome-wide association studies. Nat Genet 48(3):245–252
Hannon E et al (2018) Leveraging DNA-Methylation quantitative-trait loci to characterize the relationship between methylomic variation, gene expression, and complex traits. Am J Hum Genet 103(5):654–665
Hauberg ME et al (2017) Large-scale identification of common trait and disease variants affecting gene expression. Am J Hum Genet 100(6):885–894
He JW et al (2021) Associations of genetic variants contributing to gut microbiota composition in immunoglobin a nephropathy. mSystems. https://doi.org/10.1128/mSystems.00819-20
He L, Loika Y, Kulminski AM (2022) Allele-specific analysis reveals exon- and cell-type-specific regulatory effects of Alzheimer’s disease-associated genetic variants. Transl Psychiatry 12(1):163
Hoque M et al (2013) Analysis of alternative cleavage and polyadenylation by 3’ region extraction and deep sequencing. Nat Methods 10(2):133–139
Hormozdiari F et al (2014) Identifying causal variants at loci with multiple signals of association. Genetics 198(2):497–508
Hormozdiari F et al (2015) Identification of causal genes for complex traits. Bioinformatics 31(12):i206–i213
Hormozdiari F et al (2016) Colocalization of GWAS and eQTL signals detects target genes. Am J Hum Genet 99(6):1245–1260
Hu Y et al (2019) A statistical framework for cross-tissue transcriptome-wide association analysis. Nat Genet 51(3):568–576
Hu S et al (2021) Whole exome sequencing analyses reveal gene-microbiota interactions in the context of IBD. Gut 70(2):285–296
Huan T et al (2019) Genome-wide identification of DNA methylation QTLs in whole blood highlights pathways for cardiovascular disease. Nat Commun 10(1):4267
Imaizumi A et al (2019) Genetic basis for plasma amino acid concentrations based on absolute quantification: a genome-wide association study in the Japanese population. Eur J Hum Genet 27(4):621–630
Initiative C-HG (2021) Mapping the human genetic architecture of COVID-19. Nature 600(7889):472–477
Jansen R et al (2017) Conditional eQTL analysis reveals allelic heterogeneity of gene expression. Hum Mol Genet 26(8):1444–1451
Johnson RK et al (2022) Discovering metabolite quantitative trait loci in asthma using an isolated population. J Allergy Clin Immunol 149(5):1807–1811
Judge A, Dodd MS (2020) Metabolism. Essays Biochem 64(4):607–647
Kalayci S et al (2019) ImmuneRegulation: a web-based tool for identifying human immune regulatory elements. Nucleic Acids Res 47(W1):W142–W150
Khetan S et al (2018) Type 2 diabetes-associated genetic variants regulate chromatin accessibility in human islets. Diabetes 67(11):2466–2477
Kim-Hellmuth S et al (2017) Genetic regulatory effects modified by immune activation contribute to autoimmune disease associations. Nat Commun 8(1):266
Klemm SL, Shipony Z, Greenleaf WJ (2019) Chromatin accessibility and the regulatory epigenome. Nat Rev Genet 20(4):207–220
Kraus WE et al (2015) Metabolomic quantitative trait loci (mQTL) mapping implicates the ubiquitin proteasome system in cardiovascular disease pathogenesis. PLoS Genet 11(11):e1005553
Krause MD et al (2018) Genetic variant at coronary artery disease and ischemic stroke locus 1p32.2 regulates endothelial responses to hemodynamics. Proc Natl Acad Sci USA 115(48):E11349–E11358
Kumasaka N, Knights AJ, Gaffney DJ (2016) Fine-mapping cellular QTLs with RASQUAL and ATAC-seq. Nat Genet 48(2):206–213
Kurilshikov A et al (2021) Large-scale association analyses identify host factors influencing human gut microbiome composition. Nat Genet 53(2):156–165
Lackner DH et al (2007) A network of multiple regulatory layers shapes gene expression in fission yeast. Mol Cell 26(1):145–155
Lee Y, Rio DC (2015) Mechanisms and regulation of alternative Pre-mRNA splicing. Annu Rev Biochem 84:291–323
Lemire M et al (2015) Long-range epigenetic regulation is conferred by genetic variation located at thousands of independent loci. Nat Commun 6:6326
Leslie R, O’Donnell CJ, Johnson AD (2014) GRASP: analysis of genotype-phenotype results from 1390 genome-wide association studies and corresponding open access database. Bioinformatics 30(12):i185–i194
Li Q et al (2013) Genome-wide search for exonic variants affecting translational efficiency. Nat Commun 4:2260
Li MJ et al (2017) Exploring genetic associations with ceRNA regulation in the human genome. Nucleic Acids Res 45(10):5653–5665
Li J et al (2020) ncRNA-eQTL: a database to systematically evaluate the effects of SNPs on non-coding RNA expression across cancer types. Nucleic Acids Res 48(D1):D956–D963
Li L et al (2021) An atlas of alternative polyadenylation quantitative trait loci contributing to complex trait and disease heritability. Nat Genet 53(7):994–1005
Li JR et al (2022) Genetic variants associated mRNA stability in lung. BMC Genomics 23(1):196
Liu Z, Zhang J (2018) Human C-to-U coding RNA editing is largely nonadaptive. Mol Biol Evol 35(4):963–969
Liu ZX et al (2018) Link between m6A modification and cancers. Front Bioeng Biotechnol 6:89
Liu Z et al (2019) Detection of circular RNA expression and related quantitative trait loci in the human dorsolateral prefrontal cortex. Genome Biol 20(1):99
Liu RK et al (2021) Identification of novel functional CpG-SNPs associated with Type 2 diabetes and birth weight. Aging (albany NY) 13(7):10619–10658
Ma X et al (2023) ipaQTL-atlas: an atlas of intronic polyadenylation quantitative trait loci across human tissues. Nucleic Acids Res 51(D1):D1046–D1052
Mak TSH et al (2017) Polygenic scores via penalized regression on summary statistics. Genet Epidemiol 41(6):469–480
Mangialasche F et al (2010) Alzheimer’s disease: clinical trials and drug development. Lancet Neurol 9(7):702–716
Manolio TA et al (2009) Finding the missing heritability of complex diseases. Nature 461(7265):747–753
McVicker G et al (2013) Identification of genetic variants that affect histone modifications in human cells. Science 342(6159):747–749
Melzer D et al (2008) A genome-wide association study identifies protein quantitative trait loci (pQTLs). PLoS Genet 4(5):e1000072
Min JL et al (2021) Genomic and phenotypic insights from an atlas of genetic effects on DNA methylation. Nat Genet 53(9):1311–1321
Moore LD, Le T, Fan G (2013) DNA methylation and its basic function. Neuropsychopharmacology 38(1):23–38
Moreno V et al (2018) Colon-specific eQTL analysis to inform on functional SNPs. Br J Cancer 119(8):971–977
Nathan A et al (2022) Single-cell eQTL models reveal dynamic T cell state dependence of disease loci. Nature 606(7912):120–128
Neumeyer S, Hemani G, Zeggini E (2020) Strengthening causal inference for complex disease using molecular quantitative trait loci. Trends Mol Med 26(2):232–241
Newman JRB et al (2017) Disease-specific biases in alternative splicing and tissue-specific dysregulation revealed by multitissue profiling of lymphocyte gene expression in type 1 diabetes. Genome Res 27(11):1807–1815
Newton-Cheh C et al (2009) Genome-wide association study identifies eight loci associated with blood pressure. Nat Genet 41(6):666–676
Ng B et al (2017) An xQTL map integrates the genetic architecture of the human brain’s transcriptome and epigenome. Nat Neurosci 20(10):1418–1426
Nica AC et al (2010) Candidate causal regulatory effects by integration of expression QTLs with complex trait genetic associations. PLoS Genet 6(4):e1000895
Numata S et al (2014) DNA methylation changes in the postmortem dorsolateral prefrontal cortex of patients with schizophrenia. Front Genet 5:280
Olayinka OA et al (2022) Molecular quantitative trait locus mapping in human complex diseases. Curr Protoc 2(5):e426
Oliva M et al (2020) The impact of sex on gene expression across human tissues. Science 369(6509):eaba3066
Ota M et al (2021) Dynamic landscape of immune cell-specific gene regulation in immune-mediated diseases. Cell 184(11):3006–3021
Palanisamy V et al (2012) Control of cytokine mRNA expression by RNA-binding proteins and microRNAs. J Dent Res 91(7):651–658
Park E et al (2021) Genetic variation and microRNA targeting of A-to-I RNA editing fine tune human tissue transcriptomes. Genome Biol 22(1):77
Pavlides JM et al (2016) Predicting gene targets from integrative analyses of summary data from GWAS and eQTL studies for 28 human complex traits. Genome Med 8(1):84
Pelikan RC et al (2018) Enhancer histone-QTLs are enriched on autoimmune risk haplotypes and influence gene expression within chromatin networks. Nat Commun 9(1):2905
Perrin HJ et al (2021) Chromatin accessibility and gene expression during adipocyte differentiation identify context-dependent effects at cardiometabolic GWAS loci. PLoS Genet 17(10):e1009865
Pietzner M et al (2021) Mapping the proteo-genomic convergence of human diseases. Science 374(6569):eabj1541
Porcu E et al (2019) Mendelian randomization integrating GWAS and eQTL data reveals genetic determinants of complex and clinical traits. Nat Commun 10(1):3300
Raj T et al (2018) Integrative transcriptome analyses of the aging brain implicate altered splicing in Alzheimer’s disease susceptibility. Nat Genet 50(11):1584–1592
Rohde K et al (2019) (Epi)genetic regulation of CRTC1 in human eating behaviour and fat distribution. EBioMedicine 44:476–488
Ruan H et al (2022) GPEdit: the genetic and pharmacogenomic landscape of A-to-I RNA editing in cancers. Nucleic Acids Res 50(D1):D1231–D1237
Sainsbury S, Bernecky C, Cramer P (2015) Structural basis of transcription initiation by RNA polymerase II. Nat Rev Mol Cell Biol 16(3):129–143
Schadt EE et al (2005) An integrative genomics approach to infer causal associations between gene expression and disease. Nat Genet 37(7):710–717
Schmiedel BJ et al (2018) Impact of Genetic Polymorphisms on Human Immune Cell Gene Expression. Cell 175(6):1701–1715
Sheng X et al (2021) Mapping the genetic architecture of human traits to cell types in the kidney identifies mechanisms of disease and potential treatments. Nat Genet 53(9):1322–1333
Shetty A et al (2021) Allele-specific epigenetic activity in prostate cancer and normal prostate tissue implicates prostate cancer risk mechanisms. Am J Hum Genet 108(11):2071–2085
Shin SY et al (2014) An atlas of genetic influences on human blood metabolites. Nat Genet 46(6):543–550
Sonehara K et al (2022) Genetic architecture of microRNA expression and its link to complex diseases in the Japanese population. Hum Mol Genet 31(11):1806–1820
Soskic B et al (2022) Immune disease risk variants regulate gene expression dynamics during CD4(+) T cell activation. Nat Genet 54(6):817–826
Soubrier F (2013) From an ACE polymorphism to genome-wide searches for eQTL. J Clin Invest 123(1):111–112
Stein JL et al (2012) Identification of common variants associated with human hippocampal and intracranial volumes. Nat Genet 44(5):552–561
Sun YV, Hu YJ (2016) Integrative analysis of multi-omics data for discovery and functional studies of complex human diseases. Adv Genet 93:147–190
Sun W et al (2016) Common genetic polymorphisms influence blood biomarker measurements in COPD. PLoS Genet 12(8):e1006011
Sun W et al (2016) Histone acetylome-wide association study of autism spectrum disorder. Cell 167(5):1385–1397
Tam V et al (2019) Benefits and limitations of genome-wide association studies. Nat Rev Genet 20(8):467–484
Tan Z et al (2007) Allele-specific targeting of microRNAs to HLA-G and risk of asthma. Am J Hum Genet 81(4):829–834
Taylor DL et al (2019) Integrative analysis of gene expression, DNA methylation, physiological traits, and genetic variation in human skeletal muscle. Proc Natl Acad Sci USA 116(22):10883–10888
Tian J et al (2019) CancerSplicingQTL: a database for genome-wide identification of splicing QTLs in human cancer. Nucleic Acids Res 47(D1):D909–D916
Tian J et al (2021) CancerImmunityQTL: a database to systematically evaluate the impact of genetic variants on immune infiltration in human cancer. Nucleic Acids Res 49(D1):D1065–D1073
Toropainen A et al (2022) Functional noncoding SNPs in human endothelial cells fine-map vascular trait associations. Genome Res 32(3):409–424
Uffelmann E, Posthuma D (2021) Emerging methods and resources for biological interrogation of neuropsychiatric polygenic signal. Biol Psychiatry 89(1):41–53
Umans BD, Battle A, Gilad Y (2021) Where are the disease-associated eQTLs? Trends Genet 37(2):109–124
Vallejo DD et al (2022) Mass spectrometry methods for measuring protein stability. Chem Rev 122(8):7690–7719
van Arensbergen J, van Steensel B, Bussemaker HJ (2014) In search of the determinants of enhancer-promoter interaction specificity. Trends Cell Biol 24(11):695–702
van der Wijst M et al (2020) The single-cell eQTLGen consortium. Elife. https://doi.org/10.7554/eLife.52155
Vandiedonck C (2018) Genetic association of molecular traits: a help to identify causative variants in complex diseases. Clin Genet 93(3):520–532
Vialle RA et al (2022) Integrating whole-genome sequencing with multi-omic data reveals the impact of structural variants on gene regulation in the human brain. Nat Neurosci 25(4):504–514
Vosa U et al (2021) Large-scale cis- and trans-eQTL analyses identify thousands of genetic loci and polygenic scores that regulate blood gene expression. Nat Genet 53(9):1300–1310
Wainberg M et al (2019) Opportunities and challenges for transcriptome-wide association studies. Nat Genet 51(4):592–599
Walker RL et al (2019) Genetic control of expression and splicing in developing human brain informs disease mechanisms. Cell 179(3):750–771
Wang M et al (2018) The Mount Sinai cohort of large-scale genomic, transcriptomic and proteomic data in Alzheimer’s disease. Sci Data 5:180185
Wang J et al (2019) ncRNA-Encoded peptides or proteins and cancer. Mol Ther 27(10):1718–1725
Wang T et al (2022) Enhancing discoveries of molecular QTL studies with small sample size using summary statistic imputation. Brief Bioinform. https://doi.org/10.1093/bib/bbab370
Waszak SM et al (2015) Population variation and genetic control of modular chromatin architecture in humans. Cell 162(5):1039–1050
Wen X, Pique-Regi R, Luca F (2017) Integrating molecular QTL data into genome-wide genetic association analysis: Probabilistic assessment of enrichment and colocalization. PLoS Genet 13(3):e1006646
Westra HJ et al (2013) Systematic identification of trans eQTLs as putative drivers of known disease associations. Nat Genet 45(10):1238–1243
Williamson VS et al (2015) Expression quantitative trait loci (eQTLs) in microRNA genes are enriched for schizophrenia and bipolar disorder association signals. Psychol Med 45(12):2557–2569
Xia K et al (2012) seeQTL: a searchable database for human eQTLs. Bioinformatics 28(3):451–452
Xiong X et al (2021) Genetic drivers of m(6)A methylation in human brain, lung, heart and muscle. Nat Genet 53(8):1156–1165
Xuan JJ et al (2018) RMBase v2.0: deciphering the map of RNA modifications from epitranscriptome sequencing data. Nucleic Acids Res 46(D1):D327–D334
Yan H et al (2002) Allelic variation in human gene expression. Science 297(5584):1143
Yang C et al (2019) CoMM: a collaborative mixed model to dissecting genetic contributions to complex traits by leveraging regulatory information. Bioinformatics 35(10):1644–1652
Yang Y et al (2020a) CoMM-S2: a collaborative mixed model using summary statistics in transcriptome-wide association studies. Bioinformatics 36(7):2009–2016
Yang Y et al (2020b) SNP2APA: a database for evaluating effects of genetic variants on alternative polyadenylation in human cancers. Nucleic Acids Res 48(D1):D226–D232
Yang C et al (2021a) Genomic atlas of the proteome from brain, CSF and plasma prioritizes proteins implicated in neurological disorders. Nat Neurosci 24(9):1302–1312
Yang Y, Yeung KF, Liu J (2021b) CoMM-S(4): a collaborative mixed model using summary-Level eQTL and GWAS datasets in transcriptome-wide association studies. Front Genet 12:704538
Yang H et al (2022) ABO genotype alters the gut microbiota by regulating GalNAc levels in pigs. Nature 606(7913):358–367
Yao C et al (2018) Genome-wide mapping of plasma protein QTLs identifies putatively causal genes and pathways for cardiovascular disease. Nat Commun 9(1):3268
Ye Y et al (2020) A multi-omics perspective of quantitative trait loci in precision medicine. Trends Genet 36(5):318–336
Yu CH, Pal LR, Moult J (2016) Consensus genome-wide expression quantitative trait loci and their relationship with human complex trait disease. OMICS 20(7):400–414
Zhang Z et al (2020) Genetic analyses support the contribution of mRNA N(6)-methyladenosine (m(6)A) modification to human disease heritability. Nat Genet 52(9):939–949
Zhang Z et al (2022a) Genetic, pharmacogenomic, and immune landscapes of enhancer RNAs across human cancers. Cancer Res 82(5):785–790
Zhang Z et al (2022b) SUMMIT: an integrative approach for better transcriptomic data imputation improves causal gene identification. Nat Commun 13(1):6336
Zhao Q et al (2020) Molecular mechanisms of coronary disease revealed using quantitative trait loci for TCF21 binding, chromatin accessibility, and chromosomal looping. Genome Biol 21(1):135
Zheng Z et al (2020) QTLbase: an integrative resource for quantitative trait loci across multiple human molecular phenotypes. Nucleic Acids Res 48(D1):D983–D991
Zhou D et al (2020) A unified framework for joint-tissue transcriptome-wide association and Mendelian randomization analysis. Nat Genet 52(11):1239–1246
Zhu Z et al (2016) Integration of summary data from GWAS and eQTL studies predicts complex trait gene targets. Nat Genet 48(5):481–487
Acknowledgements
Not applicable.
Funding
This work was supported by the Tou-Yan Innovation Team Program of the Heilongjiang Province (2019-15), the National Natural Science Foundation of China (62222104, 62172130), and Heilongjiang Postdoctoral Fund (LBH-Q20030).
Author information
Authors and Affiliations
Contributions
All the authors made significant contributions: ZZ, XZ and LC designed the research; SZ, CQ, and RY performed the research; and ZZ and XC wrote the paper.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no conflict of interest, financial or otherwise.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhu, Z., Chen, X., Zhang, S. et al. Leveraging molecular quantitative trait loci to comprehend complex diseases/traits from the omics perspective. Hum. Genet. 142, 1543–1560 (2023). https://doi.org/10.1007/s00439-023-02602-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-023-02602-9