Abstract
Usher syndrome (USH) is an autosomal recessive disorder characterized by sensorineural hearing loss, progressive pigmentary retinopathy, and vestibular dysfunction. The degree and onset of hearing loss vary among subtypes I, II, and III, while blindness often occurs in the second to fourth decades of life. Usher type III (USH3), characterized by postlingual progressive sensorineural hearing loss, varying levels of vestibular dysfunction, and varying degrees of visual impairment, typically manifests in the first to second decades of life. While USH3 is rare, it is highly prevalent in certain populations. RP61, USH3, and USH3A symbolize the same disorder, with the latter symbol used more frequently in recent literature. This review focuses on the clinical features, epidemiology, molecular genetics, treatment, and research advances for sensory deficits in USH3A.

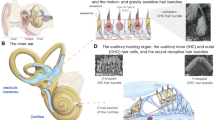
adapted from Mammano et al. (2007), panel C’’ reprinted from Geng et al (2017), panels D and E reprinted from Neurosciences: Exploring the Brain (Bear et al. 2007), and panel F reprinted from Physiology and Pathology of Chloride Transporters and Channels in the Nervous System (Alvarez-Leefmans and Delpire 2009) with permission


modified from Tian et al. (2009) under Creative Common license BY-NC-SA 4.0 and panel D modified from Lukacs et al. (2016) with permission


Similar content being viewed by others
Data availability
The manuscript does not have associated data.
Abbreviations
- CLRN1:
-
The clarin-1 protein in mice or human
- CLRN1 :
-
Human clarin-1 gene
- Clrn1 :
-
Mice clarin-1 gene
- USH3A :
-
Former designation for the gene associated with USHIII/IIIA locus; replaced by the clarin-1 gene.
- USH3A:
-
Associated with pathogenic variants in CLRN1
- IHCs:
-
Inner hair cells
- OHCs:
-
Outer hair cells
- USH:
-
Usher syndrome
- USH1:
-
Usher type I
- USH2:
-
Usher type II
- USH3:
-
Usher type III
- ENG:
-
Electronystagmography
- VNG:
-
Videonystagmography
- RP:
-
Retinitis Pigmentosa
- ERG:
-
Electroretinogram
- UTRs:
-
Untranslated regions
- MET:
-
Mechanoelectrical transduction
- INL:
-
Inner nuclear layer
- CI:
-
Cochlear implantation
- P:
-
Postnatal
- PTA:
-
Pure tone average
- PHL:
-
Progressive hearing loss
- iPSCs:
-
Induced pluripotent stem cells
- MSCs:
-
Mesenchymal stem cells
References
Abbott JA, Guth E, Kim C, Regan C, Siu VM, Rupar CA, Demeler B, Francklyn CS, Robey-Bond SM (2017) The usher syndrome type IIIB Histidyl-tRNA synthetase mutation confers temperature sensitivity. Biochemistry 56:3619–3631. https://doi.org/10.1021/acs.biochem.7b00114
Adato A, Vreugde S, Joensuu T, Avidan N, Hamalainen R, Belenkiy O, Olender T, Bonne-Tamir B, Ben-Asher E, Espinos C, Millán JM, Lehesjoki AE, Flannery JG, Avraham KB, Pietrokovski S, Sankila EM, Beckmann JS, Lancet D (2002) USH3A transcripts encode clarin-1, a four-transmembrane-domain protein with a possible role in sensory synapses. Eur J Hum Genet 10:339–350. https://doi.org/10.1038/sj.ejhg.5200831
Adjei GO, Kurtzhals JA, Rodrigues OP, Alifrangis M, Hoegberg LC, Kitcher ED, Badoe EV, Lamptey R, Goka BQ (2008) Amodiaquine-artesunate vs artemether-lumefantrine for uncomplicated malaria in Ghanaian children: a randomized efficacy and safety trial with one year follow-up. Malar J 7:127. https://doi.org/10.1186/1475-2875-7-127
Akoury E, El Zir E, Mansour A, Mégarbané A, Majewski J, Slim R (2011) A novel 5-bp deletion in Clarin 1 in a family with Usher syndrome. Ophthalmic Genet 32:245–249. https://doi.org/10.3109/13816810.2011.587083
Alagramam KN, Gopal SR, Geng R, Chen DH, Nemet I, Lee R, Tian G, Miyagi M, Malagu KF, Lock CJ, Esmieu WR, Owens AP, Lindsay NA, Ouwehand K, Albertus F, Fischer DF, Bürli RW, MacLeod AM, Harte WE, Palczewski K, Imanishi Y (2016) A small molecule mitigates hearing loss in a mouse model of Usher syndrome III. Nat Chem Biol 12:444–451. https://doi.org/10.1038/nchembio.2069
Aller E, Jaijo T, Oltra S, Alió J, Galán F, Nájera C, Beneyto M, Millán JM (2004) Mutation screening of USH3 gene (clarin-1) in Spanish patients with Usher syndrome: low prevalence and phenotypic variability. Clin Genet 66:525–529. https://doi.org/10.1111/j.1399-0004.2004.00352.x
Alvarez-Leefmans FJ, Delpire E (2009). Cochlear and Vestibular Function and Dysfunction. In Alvarez-Leefmans, F.J. and Delpire, E (eds) Physiology and Pathology of Chloride Transporters and Channels in the Nervous System. Elsevier, Amsterdam, Chapter 21, pp. 425–437.
Baloh R, Honrubia V, Kerber KA (2011) Laboratory Examination of the Vestibular System. In Baloh R, Honrubia V, Kerber KA (eds) Baloh and Honrubia's clinical neurophysiology of the vestibular system, 4th edn. Oxford, UK, Chapter 7, pp. 171–214.
Bear MF, Connors BW, Paradiso MA (2007) Audotiry and Vestibular Systems. In Bear MF, Connors BW, Paradiso MA (eds) Neuroscience : exploring the brain, 3rd edn. Lippincott Williams & Wilkins, Philadelphia, PA, Chapter 11.
Boughman JA, Vernon M, Shaver KA (1983) Usher syndrome: definition and estimate of prevalence from two high-risk populations. J Chronic Dis 36:595–603. https://doi.org/10.1016/0021-9681(83)90147-9
Carrara VI, Phyo AP, Nwee P, Soe M, Htoo H, Arunkamomkiri J, Singhasivanon P, Nosten F (2008) Auditory assessment of patients with acute uncomplicated Plasmodium falciparum malaria treated with three-day mefloquine-artesunate on the north-western border of Thailand. Malar J 7:233. https://doi.org/10.1186/1475-2875-7-233
Carrasquilla G, Barón C, Monsell EM, Cousin M, Walter V, Lefèvre G, Sander O, Fisher LM (2012) Randomized, prospective, three-arm study to confirm the auditory safety and efficacy of artemether-lumefantrine in Colombian patients with uncomplicated Plasmodium falciparum malaria. Am J Trop Med Hyg 86:75–83. https://doi.org/10.4269/ajtmh.2012.11-0192
Chen L, Chetkovich DM, Petralia RS, Sweeney NT, Kawasaki Y, Wenthold RJ, Bredt DS, Nicoll RA (2000) Stargazin regulates synaptic targeting of AMPA receptors by two distinct mechanisms. Nature 408:936–943. https://doi.org/10.1038/35050030
Dad S, Rendtorff ND, Tranebjærg L, Grønskov K, Karstensen HG, Brox V, Nilssen Ø, Roux AF, Rosenberg T, Jensen H, Møller LB (2016) Usher syndrome in Denmark: mutation spectrum and some clinical observations. Mol Genet Genomic Med 4:527–539. https://doi.org/10.1002/mgg3.228
Davies C, Bergman J, Misztal C, Ramchandran R, Mittal J, Bulut E, Shah V, Mittal R, Eshraghi AA (2021) The outcomes of cochlear implantation in Usher syndrome: a systematic review. J Clin Med. https://doi.org/10.3390/jcm10132915
Dinculescu A, Link BA, Saperstein DA (2021) Retinal gene therapy for usher syndrome: current developments, challenges, and perspectives. Int Ophthalmol Clin 61:109–124. https://doi.org/10.1097/IIO.0000000000000378
Dulon D, Papal S, Patni P, Cortese M, Vincent PF, Tertrais M, Emptoz A, Tlili A, Bouleau Y, Michel V, Delmaghani S, Aghaie A, Pepermans E, Alegria-Prevot O, Akil O, Lustig L, Avan P, Safieddine S, Petit C, El-Amraoui A (2018) Clarin-1 gene transfer rescues auditory synaptopathy in model of Usher syndrome. J Clin Invest 128:3382–3401. https://doi.org/10.1172/JCI94351
Dunbar LA, Patni P, Aguilar C, Mburu P, Corns L, Wells HR, Delmaghani S, Parker A, Johnson S, Williams D, Esapa CT, Simon MM, Chessum L, Newton S, Dorning J, Jeyarajan P, Morse S, Lelli A, Codner GF, Peineau T, Gopal SR, Alagramam KN, Hertzano R, Dulon D, Wells S, Williams FM, Petit C, Dawson SJ, Brown SD, Marcotti W, El-Amraoui A, Bowl MR (2019) Clarin-2 is essential for hearing by maintaining stereocilia integrity and function. EMBO Mol Med 11:e10288. https://doi.org/10.15252/emmm.201910288
Ebermann I, Wilke R, Lauhoff T, Lübben D, Zrenner E, Bolz HJ (2007) Two truncating USH3A mutations, including one novel, in a German family with Usher syndrome. Mol Vis 13:1539–1547
El-Amraoui A, Petit C (2014) The retinal phenotype of Usher syndrome: pathophysiological insights from animal models. C R Biol 337:167–177. https://doi.org/10.1016/j.crvi.2013.12.004
Fahim A (2018) Retinitis pigmentosa: recent advances and future directions in diagnosis and management. Curr Opin Pediatr 30:725–733. https://doi.org/10.1097/MOP.0000000000000690
Fettiplace R (2017) Hair Cell Transduction, Tuning, and Synaptic Transmission in the Mammalian Cochlea. Compr Physiol 7:1197–1227. https://doi.org/10.1002/cphy.c160049
Fields RR, Zhou G, Huang D, Davis JR, Möller C, Jacobson SG, Kimberling WJ, Sumegi J (2002) Usher syndrome type III: revised genomic structure of the USH3 gene and identification of novel mutations. Am J Hum Genet 71:607–617. https://doi.org/10.1086/342098
García-García G, Aparisi MJ, Rodrigo R, Sequedo MD, Espinós C, Rosell J, Olea JL, Mendívil MP, Ramos-Arroyo MA, Ayuso C, Jaijo T, Aller E, Millán JM (2012) Two novel disease-causing mutations in the CLRN1 gene in patients with Usher syndrome type 3. Mol Vis 18:3070–3078
Gasparini P, De Fazio A, Croce AI, Stanziale P, Zelante L (1998) Usher syndrome type III (USH3) linked to chromosome 3q in an Italian family. J Med Genet 35:666–667. https://doi.org/10.1136/jmg.35.8.666
Geller SF, Guerin KI, Visel M, Pham A, Lee ES, Dror AA, Avraham KB, Hayashi T, Ray CA, Reh TA, Bermingham-McDonogh O, Triffo WJ, Bao S, Isosomppi J, Västinsalo H, Sankila EM, Flannery JG (2009) CLRN1 is nonessential in the mouse retina but is required for cochlear hair cell development. PLoS Genet 5:e1000607. https://doi.org/10.1371/journal.pgen.1000607
Geng R, Geller SF, Hayashi T, Ray CA, Reh TA, Bermingham-McDonogh O, Jones SM, Wright CG, Melki S, Imanishi Y, Palczewski K, Alagramam KN, Flannery JG (2009) Usher syndrome IIIA gene clarin-1 is essential for hair cell function and associated neural activation. Hum Mol Genet 18:2748–2760. https://doi.org/10.1093/hmg/ddp210
Geng R, Melki S, Chen DH, Tian G, Furness DN, Oshima-Takago T, Neef J, Moser T, Askew C, Horwitz G, Holt JR, Imanishi Y, Alagramam KN (2012) The mechanosensory structure of the hair cell requires clarin-1, a protein encoded by Usher syndrome III causative gene. J Neurosci 32:9485–9498. https://doi.org/10.1523/JNEUROSCI.0311-12.2012
Geng R, Omar A, Gopal SR, Chen DH, Stepanyan R, Basch ML, Dinculescu A, Furness DN, Saperstein D, Hauswirth W, Lustig LR, Alagramam KN (2017) Modeling and preventing progressive hearing loss in Usher syndrome III. Sci Rep 7:13480. https://doi.org/10.1038/s41598-017-13620-9
Gopal SR, Chen DH, Chou SW, Zang J, Neuhauss SC, Stepanyan R, McDermott BM, Alagramam KN (2015) Zebrafish models for the mechanosensory hair cell dysfunction in usher syndrome 3 reveal that Clarin-1 Is an essential hair bundle protein. J Neurosci 35:10188–10201. https://doi.org/10.1523/JNEUROSCI.1096-15.2015
Gopal SR, Lee YT, Stepanyan R, McDermott BM, Alagramam KN (2019) Unconventional secretory pathway activation restores hair cell mechanotransduction in an USH3A model. Proc Natl Acad Sci USA 116:11000–11009. https://doi.org/10.1073/pnas.1817500116
Grossniklaus HE, Geisert EE, Nickerson JM (2015) Introduction to the Retina. Prog Mol Biol Transl Sci 134:383–396. https://doi.org/10.1016/bs.pmbts.2015.06.001
Grøndahl J, Mjøen S (1986) Usher syndrome in four Norwegian counties. Clin Genet 30:14–28. https://doi.org/10.1111/j.1399-0004.1986.tb00564.x
György B, Meijer EJ, Ivanchenko MV, Tenneson K, Emond F, Hanlon KS, Indzhykulian AA, Volak A, Karavitaki KD, Tamvakologos PI, Vezina M, Berezovskii VK, Born RT, O’Brien M, Lafond JF, Arsenijevic Y, Kenna MA, Maguire CA, Corey DP (2019) Gene transfer with AAV9-PHP.B rescues hearing in a mouse model of usher syndrome 3A and transduces hair cells in a non-human primate. Mol Ther Methods Clin Dev 13:1–13. https://doi.org/10.1016/j.omtm.2018.11.003
Haynes RK, Vonwiller SC (1994) Extraction of artemisinin and artemisinic acid: preparation of artemether and new analogues. Trans R Soc Trop Med Hyg 88(Suppl 1):S23–S26. https://doi.org/10.1016/0035-9203(94)90466-9
Herrera W, Aleman TS, Cideciyan AV, Roman AJ, Banin E, Ben-Yosef T, Gardner LM, Sumaroka A, Windsor EA, Schwartz SB, Stone EM, Liu XZ, Kimberling WJ, Jacobson SG (2008) Retinal disease in Usher syndrome III caused by mutations in the clarin-1 gene. Invest Ophthalmol Vis Sci 49:2651–2660. https://doi.org/10.1167/iovs.07-1505
Hien TT, Davis TM, Chuong LV, Ilett KF, Sinh DX, Phu NH, Agus C, Chiswell GM, White NJ, Farrar J (2004) Comparative pharmacokinetics of intramuscular artesunate and artemether in patients with severe falciparum malaria. Antimicrob Agents Chemother 48:4234–4239
Hien TT, Arnold K, Vinh H, Cuong BM, Phu NH, Chau TT, Hoa NT, Chuong LV, Mai NT, Vinh NN, Trang TT (1992) Comparison of artemisinin suppositories with intravenous artesunate and intravenous quinine in the treatment of cerebral malaria. Trans R Soc Trop Med Hyg 86:582–583. https://doi.org/10.1016/0035-9203(92)90137-2
Holan V, Palacka K, Hermankova B (2021) Mesenchymal stem cell-based therapy for retinal degenerative diseases: experimental models and clinical trials. Cells 10:588. https://doi.org/10.3390/cells10030588
Holden LK, Finley CC, Firszt JB, Holden TA, Brenner C, Potts LG, Gotter BD, Vanderhoof SS, Mispagel K, Heydebrand G, Skinner MW (2013) Factors affecting open-set word recognition in adults with cochlear implants. Ear Hear 34:342–360. https://doi.org/10.1097/AUD.0b013e3182741aa7
Hope CI, Bundey S, Proops D, Fielder AR (1997) Usher syndrome in the city of Birmingham–prevalence and clinical classification. Br J Ophthalmol 81:46–53. https://doi.org/10.1136/bjo.81.1.46
Hutagalung R, Htoo H, Nwee PA, Arunkamomkiri J, Zwang J, Carrara VI, Ashley E, Singhasivanon P, White NJ, Nosten F (2006) A case-control auditory evaluation of patients treated with artemether-lumefantrine. Am J Trop Med Hyg 74:211–214. https://doi.org/10.4269/ajtmh.2006.74.211
Isosomppi J, Västinsalo H, Geller SF, Heon E, Flannery JG, Sankila EM (2009) Disease-causing mutations in the CLRN1 gene alter normal CLRN1 protein trafficking to the plasma membrane. Mol Vis 15:1806–1818
Ivanchenko MV, Hanlon KS, Hathaway DM, Klein AJ, Peters CW, Li Y, Tamvakologos PI, Nammour J, Maguire CA, Corey DP (2021) AAV-S: a versatile capsid variant for transduction of mouse and primate inner ear. Mol Ther Methods Clin Dev 21:382–398. https://doi.org/10.1016/j.omtm.2021.03.019
Joensuu T, Blanco G, Pakarinen L, Sistonen P, Kääriäinen H, Brown S, de la Chapelle A, Sankila EM (1996) Refined mapping of the Usher syndrome type III locus on chromosome 3, exclusion of candidate genes, and identification of the putative mouse homologous region. Genomics 38:255–263. https://doi.org/10.1006/geno.1996.0626
Joensuu T, Hämäläinen R, Lehesjoki AE, de la Chapelle A, Sankila EM (2000) A sequence-ready map of the Usher syndrome type III critical region on chromosome 3q. Genomics 63:409–416. https://doi.org/10.1006/geno.1999.6096
Joensuu T, Hämäläinen R, Yuan B, Johnson C, Tegelberg S, Gasparini P, Zelante L, Pirvola U, Pakarinen L, Lehesjoki AE, de la Chapelle A, Sankila EM (2001) Mutations in a novel gene with transmembrane domains underlie Usher syndrome type 3. Am J Hum Genet 69:673–684. https://doi.org/10.1086/323610
Khan AO, Becirovic E, Betz C, Neuhaus C, Altmüller J, Maria Riedmayr L, Motameny S, Nürnberg G, Nürnberg P, Bolz HJ (2017) A deep intronic CLRN1 (USH3A) founder mutation generates an aberrant exon and underlies severe Usher syndrome on the Arabian Peninsula. Sci Rep 7:1411. https://doi.org/10.1038/s41598-017-01577-8
Khan S, Chang R (2013) Anatomy of the vestibular system: a review. NeuroRehabilitation 32:437–443. https://doi.org/10.3233/NRE-130866
Kim HH, Barrs DM (2006) Hearing aids: a review of what’s new. Otolaryngol Head Neck Surg 134:1043–1050. https://doi.org/10.1016/j.otohns.2006.03.010
Kimberling WJ, Hildebrand MS, Shearer AE, Jensen ML, Halder JA, Trzupek K, Cohn ES, Weleber RG, Stone EM, Smith RJH (2010) Frequency of Usher syndrome in two pediatric populations: Implications for genetic screening of deaf and hard of hearing children. Genet Med 12:512–516. https://doi.org/10.1097/GIM.0b013e3181e5afb8
Kissinger E, Hien TT, Hung NT, Nam ND, Tuyen NL, Dinh BV, Mann C, Phu NH, Loc PP, Simpson JA, White NJ, Farrar JJ (2000) Clinical and neurophysiological study of the effects of multiple doses of artemisinin on brain-stem function in Vietnamese patients. Am J Trop Med Hyg 63:48–55. https://doi.org/10.4269/ajtmh.2000.63.48
Le Quesne Stabej P, Saihan Z, Rangesh N, Steele-Stallard HB, Ambrose J, Coffey A, Emmerson J, Haralambous E, Hughes Y, Steel KP, Luxon LM, Webster AR, Bitner-Glindzicz M (2012) Comprehensive sequence analysis of nine Usher syndrome genes in the UK National Collaborative Usher Study. J Med Genet 49:27–36. https://doi.org/10.1136/jmedgenet-2011-100468
LeMasurier M, Gillespie PG (2005) Hair-cell mechanotransduction and cochlear amplification. Neuron 48:403–415. https://doi.org/10.1016/j.neuron.2005.10.017
Li Q, Remich S, Miller SR, Ogutu B, Otieno W, Melendez V, Teja-Isavadharm P, Weina PJ, Hickman MR, Smith B, Polhemus M (2014) Pharmacokinetic evaluation of intravenous artesunate in adults with uncomplicated falciparum malaria in Kenya: a phase II study. Malar J 13:281. https://doi.org/10.1186/1475-2875-13-281
Lieu JEC, Kenna M, Anne S, Davidson L (2020) Hearing Loss in Children: A Review. JAMA 324:2195–2205. https://doi.org/10.1001/jama.2020.17647
Litovsky RY, Gordon K (2016) Bilateral cochlear implants in children: effects of auditory experience and deprivation on auditory perception. Hear Res 338:76–87. https://doi.org/10.1016/j.heares.2016.01.003
Loundon N, Marlin S, Busquet D, Denoyelle F, Roger G, Renaud F, Garabedian EN (2003) Usher syndrome and cochlear implantation. Otol Neurotol 24:216–221. https://doi.org/10.1097/00129492-200303000-00015
Lukacs GL (2016) Proteostasis: Chaperoning for hearing loss. Nat Chem Biol 12:388–389. https://doi.org/10.1038/nchembio.2091
Lumpkin EA, Collisson T, Parab P, Omer-Abdalla A, Haeberle H, Chen P, Doetzlhofer A, White P, Groves A, Segil N, Johnson JE (2003) Math1-driven GFP expression in the developing nervous system of transgenic mice. Gene Expr Patterns 3:389–395. https://doi.org/10.1016/s1567-133x(03)00089-9
Malm E, Ponjavic V, Möller C, Kimberling WJ, Stone ES, Andréasson S (2011) Alteration of rod and cone function in children with Usher syndrome. Eur J Ophthalmol 21:30–38. https://doi.org/10.5301/ejo.2010.5433
Mammano F, Bortolozzi M, Ortolano S, Anselmi F (2007) Ca2+ signaling in the inner ear. Physiology (bethesda) 22:131–144. https://doi.org/10.1152/physiol.00040.2006
Maude RJ, Plewes K, Faiz MA, Hanson J, Charunwatthana P, Lee SJ, Tärning J, Yunus EB, Hoque MG, Hasan MU, Hossain A, Lindegardh N, Day NP, White NJ, Dondorp AM (2009) Does artesunate prolong the electrocardiograph QT interval in patients with severe malaria? Am J Trop Med Hyg 80:126–132. https://doi.org/10.4269/ajtmh.2009.08-0326
Naples JG, Ruckenstein MJ (2020) Cochlear implant. Otolaryngol Clin N Am 53:87–102. https://doi.org/10.1016/j.otc.2019.09.004
Ness SL, Ben-Yosef T, Bar-Lev A, Madeo AC, Brewer CC, Avraham KB, Kornreich R, Desnick RJ, Willner JP, Friedman TB, Griffith AJ (2003) Genetic homogeneity and phenotypic variability among Ashkenazi Jews with Usher syndrome type III. J Med Genet 40:767–772. https://doi.org/10.1136/jmg.40.10.767
Ogun O, Zallocchi M (2014) Clarin-1 acts as a modulator of mechanotransduction activity and presynaptic ribbon assembly. J Cell Biol 207:375–391. https://doi.org/10.1083/jcb.201404016
Pakarinen L, Sankila EM, Tuppurainen K, Karjalaninen S, Helena K (1995a) Usher syndrome type III (USH3): The clinical manifestations in 42 patients. Scand J Log Phoniatr 20:141–150. https://doi.org/10.3109/14015439509098741
Pakarinen L, Karjalainen S, Simola KO, Laippala P, Kaitalo H (1995b) Usher’s syndrome type 3 in Finland. Laryngoscope 105:613–617. https://doi.org/10.1288/00005537-199506000-00010
Pakarinen L, Tuppurainen K, Laippala P, Mäntyjärvi M, Puhakka H (1995c) The ophthalmological course of Usher syndrome type III. Int Ophthalmol 19:307–311. https://doi.org/10.1007/BF00130927
Pennings RJ, Deutman AF, Fields RR, Kimberling WJ, Huygen PL, Cremers WR (2003) Usher syndrome type III can mimic other types of Usher syndrome. Ann Otol Rhinol Laryngol 112:525–530. https://doi.org/10.1177/000348940311200608
Phillips JB, Västinsalo H, Wegner J, Clément A, Sankila EM, Westerfield M (2013) The cone-dominant retina and the inner ear of zebrafish express the ortholog of CLRN1, the causative gene of human Usher syndrome type 3A. Gene Expr Patterns 13:473–481. https://doi.org/10.1016/j.gep.2013.09.001
Pietola L, Aarnisalo AA, Abdel-Rahman A, Västinsalo H, Isosomppi J, Löppönen H, Kentala E, Johansson R, Valtonen H, Vasama JP, Sankila EM, Jero J (2012) Speech recognition and communication outcomes with cochlear implantation in Usher syndrome type 3. Otol Neurotol 33:38–41. https://doi.org/10.1097/MAO.0b013e31823dbc56
Plantinga RF, Kleemola L, Huygen PL, Joensuu T, Sankila EM, Pennings RJ, Cremers CW (2005) Serial audiometry and speech recognition findings in Finnish Usher syndrome type III patients. Audiol Neurootol 10:79–89. https://doi.org/10.1159/000083363
Plantinga RF, Pennings RJ, Huygen PL, Sankila EM, Tuppurainen K, Kleemola L, Cremers CW, Deutman AF (2006) Visual impairment in Finnish Usher syndrome type III. Acta Ophthalmol Scand 84:36–41. https://doi.org/10.1111/j.1600-0420.2005.00507.x
Puffenberger EG, Jinks RN, Sougnez C, Cibulskis K, Willert RA, Achilly NP, Cassidy RP, Fiorentini CJ, Heiken KF, Lawrence JJ, Mahoney MH, Miller CJ, Nair DT, Politi KA, Worcester KN, Setton RA, Dipiazza R, Sherman EA, Eastman JT, Francklyn C, Robey-Bond S, Rider NL, Gabriel S, Morton DH, Strauss KA (2012) Genetic mapping and exome sequencing identify variants associated with five novel diseases. PLoS ONE 7:e28936. https://doi.org/10.1371/journal.pone.0028936
Ratnam K, Västinsalo H, Roorda A, Sankila EM, Duncan JL (2013) Cone structure in patients with usher syndrome type III and mutations in the Clarin 1 gene. JAMA Ophthalmol 131:67–74. https://doi.org/10.1001/2013.jamaophthalmol.2
Rees HA, Liu DR (2018) Base editing: precision chemistry on the genome and transcriptome of living cells. Nat Rev Genet 19:770–788. https://doi.org/10.1038/s41576-018-0059-1
Rehfeld A, Nylander M, Karnov K (2017) The Eye. In Rehfeld A, Nylander M, Karnov K (eds) Compendium of histology. Springer, Cham, Chapter 28, pp 603–638
Reiss LA (2020) Cochlear implants and other inner ear prostheses: today and tomorrow. Curr Opin Physiol 18:49–55. https://doi.org/10.1016/j.cophys.2020.08.001
Rosenberg T, Haim M, Hauch AM, Parving A (1997) The prevalence of Usher syndrome and other retinal dystrophy-hearing impairment associations. Clin Genet 51:314–321. https://doi.org/10.1111/j.1399-0004.1997.tb02480.x
Sadeghi M, Cohn ES, Kimberling WJ, Tranebjaerg L, Möller C (2005) Audiological and vestibular features in affected subjects with USH3: a genotype/phenotype correlation. Int J Audiol 44:307–316. https://doi.org/10.1080/14992020500060610
Sadeghi M, William K, Tranebjœrg L, Möller C (2004) The Prevalence of Usher Syndrome in Sweden: a Nationwide Epidemiological and Clinical Survey 2:220–228. https://doi.org/10.1080/16513860410003093
Schmitz F, Königstorfer A, Südhof TC (2000) RIBEYE, a component of synaptic ribbons: a protein’s journey through evolution provides insight into synaptic ribbon function. Neuron 28:857–872. https://doi.org/10.1016/s0896-6273(00)00159-8
Shiomi Y, Naito Y, Hirano S, Fujiki N, Honjo I (1997) Cortical activity of a patient with Usher’s syndrome using a cochlear implant. Am J Otolaryngol 18:412–414. https://doi.org/10.1016/s0196-0709(97)90063-6
Spandau UH, Rohrschneider K (2002) Prevalence and geographical distribution of Usher syndrome in Germany. Graefes Arch Clin Exp Ophthalmol 240:495–498. https://doi.org/10.1007/s00417-002-0485-8
Starkov D, Strupp M, Pleshkov M, Kingma H, van de Berg R (2021) Diagnosing vestibular hypofunction: an update. J Neurol 268:377–385. https://doi.org/10.1007/s00415-020-10139-4
Stemerdink M, García-Bohórquez B, Schellens R, Garcia-Garcia G, Van Wijk E, Millan JM (2021) Genetics, pathogenesis and therapeutic developments for Usher syndrome type 2. Hum Genet. https://doi.org/10.1007/s00439-021-02324-w
Stingl K, Kurtenbach A, Hahn G, Kernstock C, Hipp S, Zobor D, Kohl S, Bonnet C, Mohand-Saïd S, Audo I, Fakin A, Hawlina M, Testa F, Simonelli F, Petit C, Sahel JA, Zrenner E (2019) Full-field electroretinography, visual acuity and visual fields in Usher syndrome: a multicentre European study. Doc Ophthalmol 139:151–160. https://doi.org/10.1007/s10633-019-09704-8
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126:663–676. https://doi.org/10.1016/j.cell.2006.07.024
Tamayo ML, Bernal JE, Tamayo GE, Frias JL, Alvira G, Vergara O, Rodriguez V, Uribe JI, Silva JC (1991) Usher syndrome: results of a screening program in Colombia. Clin Genet 40:304–311. https://doi.org/10.1111/j.1399-0004.1991.tb03100.x
Tian G, Lee R, Ropelewski P, Imanishi Y (2016) Impairment of vision in a mouse model of usher syndrome type III. Invest Ophthalmol Vis Sci 57:866–875. https://doi.org/10.1167/iovs.15-16946
Tian G, Zhou Y, Hajkova D, Miyagi M, Dinculescu A, Hauswirth WW, Palczewski K, Geng R, Alagramam KN, Isosomppi J, Sankila EM, Flannery JG, Imanishi Y (2009) Clarin-1, encoded by the Usher Syndrome III causative gene, forms a membranous microdomain: possible role of clarin-1 in organizing the actin cytoskeleton. J Biol Chem 284:18980–18993. https://doi.org/10.1074/jbc.M109.003160
Toms M, Pagarkar W, Moosajee M (2020) Usher syndrome: clinical features, molecular genetics and advancing therapeutics. Ther Adv Ophthalmol 12:2515841420952194. https://doi.org/10.1177/2515841420952194
Toovey S, Jamieson A (2004) Audiometric changes associated with the treatment of uncomplicated falciparum malaria with co-artemether. Trans R Soc Trop Med Hyg 98: 261–7; discussion 268–9. https://doi.org/10.1016/j.trstmh.2003.11.001
Toulis V, Marfany G (2020) By the Tips of Your Cilia: Ciliogenesis in the Retina and the Ubiquitin-Proteasome System. In Barrio R, Sutherland JD, Rodriguez MS (eds) Proteostasis and Disease: From Basic Mechanisms to Clinics. Springer, Cham, Chapter 13, pp 303–318
Tsang SH, Aycinena ARP, Sharma T (2018) Ciliopathy: Usher syndrome. Adv Exp Med Biol 1085:167–170. https://doi.org/10.1007/978-3-319-95046-4_32
Van Camp G, Smith RJH (2021) Hereditary Hearing Loss Homepage. https://hereditaryhearingloss.org.
van de Berg R, van Tilburg M, Kingma H (2015) Bilateral vestibular hypofunction: challenges in establishing the diagnosis in adults. ORL J Otorhinolaryngol Relat Spec 77:197–218. https://doi.org/10.1159/000433549
Vona B, Mazaheri N, Lin SJ, Dunbar LA, Maroofian R, Azaiez H, Booth KT, Vitry S, Rad A, Rüschendorf F, Varshney P, Fowler B, Beetz C, Alagramam KN, Murphy D, Shariati G, Sedaghat A, Houlden H, Petree C, VijayKumar S, Smith RJH, Haaf T, El-Amraoui A, Bowl MR, Varshney GK, Galehdari H (2021) A biallelic variant in CLRN2 causes non-syndromic hearing loss in humans. Hum Genet 140:915–931. https://doi.org/10.1007/s00439-020-02254-z
Västinsalo H, Jalkanen R, Dinculescu A, Isosomppi J, Geller S, Flannery JG, Hauswirth WW, Sankila EM (2011) Alternative splice variants of the USH3A gene Clarin 1 (CLRN1). Eur J Hum Genet 19:30–35. https://doi.org/10.1038/ejhg.2010.140
Wang J, Puel JL (2018) Toward cochlear therapies. Physiol Rev 98:2477–2522. https://doi.org/10.1152/physrev.00053.2017
Xu L, Bolch SN, Santiago CP, Dyka FM, Akil O, Lobanova ES, Wang Y, Martemyanov KA, Hauswirth WW, Smith WC, Handa JT, Blackshaw S, Ash JD, Dinculescu A (2020) Clarin-1 expression in adult mouse and human retina highlights a role of Müller glia in Usher syndrome. J Pathol 250:195–204. https://doi.org/10.1002/path.5360
Yang D, Prieskorn D, Song J, Beyer L, Dolan D, Xu J, Zhang J, Raphael Y, Chen YE (2022) Rabbit Models of USH3A Have Progressive Hearing Loss .The Association for Research in Otolaryngology (ARO) - The 45th Annual MidWinter Meeting, Virtual.
Yoshimura H, Oshikawa C, Nakayama J, Moteki H, Usami S (2015) Identification of a novel CLRN1 gene mutation in Usher syndrome type 3: two case reports. Ann Otol Rhinol Laryngol 124(Suppl 1):94S-S99. https://doi.org/10.1177/0003489415574069
Zallocchi M, Meehan DT, Delimont D, Askew C, Garige S, Gratton MA, Rothermund-Franklin CA, Cosgrove D (2009) Localization and expression of clarin-1, the Clrn1 gene product, in auditory hair cells and photoreceptors. Hear Res 255:109–120. https://doi.org/10.1016/j.heares.2009.06.006
Acknowledgements
The authors acknowledge the support of Usher III Initiative, Chicago, which played a pioneering role in bringing together a team of scientists and experts around the world to develop therapies to treat both vision and hearing loss in individuals diagnosed with USH3A. Basic science research related clarin-1 in KNA’s lab was supported by the National Institutes of Health Grants (R01-DC010816) and the Maniglia Endowed Chair, University Hospitals Cleveland Medical Center.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
This study was exempt from IRB review, as it is a review article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Marouf, A., Johnson, B. & Alagramam, K.N. Usher syndrome IIIA: a review of the disorder and preclinical research advances in therapeutic approaches. Hum Genet 141, 759–783 (2022). https://doi.org/10.1007/s00439-022-02446-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-022-02446-9