Abstract
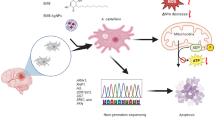
Acanthamoeba castellanii, a ubiquitous protozoan, is responsible for significant diseases such as Acanthamoeba keratitis and granulomatous amoebic encephalitis. A crucial survival strategy of A. castellanii involves the formation of highly resistant cysts during adverse conditions. This study delves into the cellular processes underpinning encystment, focusing on gene expression changes related to reactive oxygen species (ROS) balance, with a particular emphasis on mitochondrial processes. Our findings reveal a dynamic response within the mitochondria during encystment, with the downregulation of key enzymes involved in oxidative phosphorylation (COX, AOX, and NADHalt) during the initial 48 h, followed by their overexpression at 72 h. This orchestrated response likely creates a pro-oxidative environment, facilitating encystment. Analysis of other ROS processing enzymes across the cell reveals differential expression patterns. Notably, antioxidant enzymes, such as catalases, glutaredoxins, glutathione S-transferases, peroxiredoxins, and thioredoxins, mirror the mitochondrial trend of downregulation followed by upregulation. Additionally, glycolysis and gluconeogenesis are downregulated during the early stages in order to potentially balance the metabolic requirement of the cyst. Our study underscores the importance of ROS regulation in Acanthamoeba encystment. Understanding these mechanisms offers insights into infection control and identifies potential therapeutic targets. This work contributes to unraveling the complex biology of A. castellanii and may aid in combatting Acanthamoeba-related infections. Further research into ROS and oxidase enzymes is warranted, given the organism’s remarkable respiratory versatility.





Similar content being viewed by others
Data availability
The information of the discussed genes can be found in the supplementary materials. Everything else can be provided upon request.
References
Antos-Krzeminska N, Jarmuszkiewicz W (2014) External NAD(P)H dehydrogenases in Acanthamoeba castellanii mitochondria. Protist 165:580–593. https://doi.org/10.1016/j.protis.2014.07.002
Antos-Krzeminska N, Jarmuszkiewicz W (2019) Alternative type II NAD(P)H dehydrogenases in the mitochondria of protists and fungi. Protist 170:21–37. https://doi.org/10.1016/j.protis.2018.11.001
Aurrecoechea C, Barreto A, Brestelli J et al (2011) AmoebaDB and MicrosporidiaDB: functional genomic resources for Amoebozoa and Microsporidia species. Nucleic Acids Res 39:612–619. https://doi.org/10.1093/nar/gkq1006
Bahr JT, Bonner WD (1973) Cyanide-insensitive respiration. II. Control of the alternate pathway. J Biol Chem 248:3446–3450
Band RN, Mohrlok S (1969) The respiratory metabolism of Acanthamoeba rhysodes during encystation. J Gen Microbiol 59:351–358. https://doi.org/10.1099/00221287-59-3-351
Barnstable CJ, Zhang M, Tombran-Tink J (2022) Uncoupling proteins as therapeutic targets for neurodegenerative diseases. Int J Mol Sci 23
Bernard C, Locard-Paulet M, Noël C et al (2022) A time-resolved multi-omics atlas of Acanthamoeba castellanii encystment. Nat Commun 13:4104. https://doi.org/10.1038/s41467-022-31832-0
Bowers B, Korn ED (1968) The fine structure of Acanthamoeba castellanii. J Cell Biol 39:95–111
Brigelius-Flohé R, Maiorino M (2013) Glutathione peroxidases. Biochimica et Biophysica Acta (BBA) - General Subjects 1830:3289–3303. https://doi.org/10.1016/j.bbagen.2012.11.020
Chen L, Orfeo T, Gilmartin G, Bateman E (2004) Mechanism of cyst specific protein 21 mRNA induction during Acanthamoeba differentiation. Biochim Biophys Acta 1691:23–31. https://doi.org/10.1016/j.bbamcr.2003.11.005
Clarkson AB, Bienen EJ, Pollakis G, Grady RW (1989) Respiration of bloodstream forms of the parasite Trypanosoma brucei brucei is dependent on a plant-like alternative oxidase. J Biol Chem 264:17770–17776. https://doi.org/10.1016/S0021-9258(19)84639-2
Czarna M, Jarmuszkiewicz W (2005) Activation of alternative oxidase and uncoupling protein lowers hydrogen peroxide formation in amoeba Acanthamoeba castellanii mitochondria. FEBS Lett 579:3136–3140. https://doi.org/10.1016/j.febslet.2005.04.081
Czarna M, Sluse FE, Jarmuszkiewicz W (2007) Mitochondrial function plasticity in Acanthamoeba castellanii during growth in batch culture. J Bioenerg Biomembr 39:149–157. https://doi.org/10.1007/s10863-007-9073-2
Day DA, Whelan J, Millar AH et al (1995) Regulation of the alternative oxidase in plants and fungi. Aust J Plant Physiol 22:497–509. https://doi.org/10.1071/PP9950497
de Alves DSMM, Alves LM, da Costa TL et al (2017) Anaerobic metabolism in T4 Acanthamoeba genotype. Curr Microbiol 74:685–690. https://doi.org/10.1007/s00284-017-1223-0
de Obeso Fernández del Valle A (2018) Protein secretion and encystation in Acanthamoeba. The University of Edinburgh
de Obeso Fernández del Valle A, Gómez-Montalvo J, Maciver SK (2022) Acanthamoeba castellanii exhibits intron retention during encystment. Parasitol Res 121:2615–2622. https://doi.org/10.1007/s00436-022-07578-5
de Obeso Fernandez del Valle A, Scheckhuber CQ (2022) Superoxide dismutases in eukaryotic microorganisms: four case studies. Antioxidants 11
de Obeso Fernández del Valle A, Scheckhuber CQ, Chavaro-Pérez DA, et al (2023) mRNA sequencing reveals upregulation of glutathione S-transferase genes during Acanthamoeba encystation. Microorganisms 11
Dobin A, Davis CA, Schlesinger F et al (2013) STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29:15–21. https://doi.org/10.1093/bioinformatics/bts635
Dudley R, Jarroll EL, Khan NA (2009) Carbohydrate analysis of Acanthamoeba castellanii. Exp Parasitol 122:338–343. https://doi.org/10.1016/j.exppara.2009.04.009
Edwards SW, Lloyd D (1977) Changes in oxygen uptake rates, enzyme activities, cytochrome amounts and adenine nucleotide pool levels during growth of Acanthamoeba castellanii in batch culture. J Gen Microbiol 102:135–144. https://doi.org/10.1099/00221287-102-1-135
Forman HJ, Zhang H, Rinna A (2009) Glutathione: overview of its protective roles, measurement, and biosynthesis. Mol Aspects Med 30:1–12. https://doi.org/10.1016/j.mam.2008.08.006
Gómez J, Mota-Martorell N, Jové M, et al (2023) Mitochondrial ROS production, oxidative stress and aging within and between species: evidences and recent advances on this aging effector. Exp Gerontol 174. https://doi.org/10.1016/J.EXGER.2023.112134
Gruber J, Schaffer S, Halliwell B (2008) The mitochondrial free radical theory of ageing–where do we stand? Front Biosci 13:6554–6579. https://doi.org/10.2741/3174
Henriquez FL, McBride J, Campbell SJ et al (2009) Acanthamoeba alternative oxidase genes: identification, characterisation and potential as antimicrobial targets. Int J Parasitol 39:1417–1424. https://doi.org/10.1016/J.IJPARA.2009.04.011
Hirschenson J, Melgar-Bermudez E, Mailloux RJ (2022) The uncoupling proteins: a systematic review on the mechanism used in the prevention of oxidative stress. Antioxidants 11
Hirukawa Y, Nakato H, Izumi S et al (1998) Structure and expression of a cyst specific protein of Acanthamoeba castellanii. Biochim Biophys Acta 1398:47–56. https://doi.org/10.1016/S0167-4781(98)00026-8
Jacob L-M, Jarmila K, Enrique M-C et al (2008) Glycogen phosphorylase in Acanthamoeba spp.: determining the role of the enzyme during the encystment process using RNA interference. Eukaryot Cell 7:509–517. https://doi.org/10.1128/ec.00316-07
Jarmuszkiewicz W, Wagner AM, Wagner MJ, Hryniewiecka L (1997) Immunological identification of the alternative oxidase of Acanthamoeba castellanii mitochondria. FEBS Lett 411:110–114. https://doi.org/10.1016/S0014-5793(97)00676-5
Joseph-Horne T, Hollomon DW, Wood PM (2001) Fungal respiration: a fusion of standard and alternative components. Biochimica et Biophysica Acta (BBA) 1504:179–195. https://doi.org/10.1016/S0005-2728(00)00251-6
Kanehisa M, Goto S (2000) KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res 28:27–30
Kanehisa M, Sato Y, Kawashima M et al (2016) KEGG as a reference resource for gene and protein annotation. Nucleic Acids Res 44:D457–D462. https://doi.org/10.1093/nar/gkv1070
Kersey PJ, Allen JE, Allot A, et al (2017) Ensembl Genomes 2018: an integrated omics infrastructure for non-vertebrate species. Nucleic Acids Res 1–7. https://doi.org/10.1093/nar/gkx1011
Kim S-H, Moon E-K, Hong Y et al (2015) Autophagy protein 12 plays an essential role in Acanthamoeba encystation. Exp Parasitol 159:46–52. https://doi.org/10.1016/j.exppara.2015.08.013
Kumar JB, Hui-Jung J, Incheol S et al (2014) Chloroquine has a cytotoxic effect on Acanthamoeba encystation through modulation of autophagy. Antimicrob Agents Chemother 58:6235–6241. https://doi.org/10.1128/aac.03164-14
Lambers H (1985) Respiration in intact plants and tissues: its regulation and dependence on environmental factors, metabolism and invaded organisms. Higher Plant Cell Respiration 418–473. 10.1007/978-3-642-70101-6_15
Lambowitz AM, Sabourin JR, Bertrand H et al (1989) Immunological identification of the alternative oxidase of Neurospora crassa mitochondria. Mol Cell Biol 9:1362–1364. https://doi.org/10.1128/MCB.9.3.1362-1364.1989
Lazzarotto F, Turchetto-Zolet AC, Margis-Pinheiro M (2015) Revisiting the non-animal peroxidase superfamily. Trends Plant Sci 20:807–813. https://doi.org/10.1016/j.tplants.2015.08.005
Lee JY, Song SM, Moon EK et al (2013) Cysteine protease inhibitor (AcStefin) is required for complete cyst formation of Acanthamoeba. Eukaryot Cell 12:567–574. https://doi.org/10.1128/EC.00308-12
Lee JY, Song SM, Moon EK et al (2013) Cysteine protease inhibitor (AcStefin) is required for complete cyst formation of Acanthamoeba. Eukaryot Cell 12:567–574. https://doi.org/10.1128/EC.00308-12
Leitsch D, Köhsler M, Marchetti-Deschmann M et al (2010) Major role for cysteine proteases during the early phase of Acanthamoeba castellanii encystment. Eukaryot Cell 9:611–618. https://doi.org/10.1128/EC.00300-09
Liao Y, Smyth GK, Shi W (2014) FeatureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30:923–930. https://doi.org/10.1093/bioinformatics/btt656
Lloyd D (2014) Encystment in Acanthamoeba castellanii: a review. Exp Parasitol 145:S20–S27. https://doi.org/10.1016/j.exppara.2014.03.026
Lorenzo-Morales J, Khan NA, Walochnik J (2015) An update on Acanthamoeba keratitis: diagnosis, pathogenesis and treatment. Parasite 22
Lorenzo-Morales J, Kliescikova J, Martinez-Carretero E et al (2008) Glycogen phosphorylase in Acanthamoeba spp.: determining the role of the enzyme during the encystment process using RNA interference. Eukaryot Cell 7:509–517. https://doi.org/10.1128/EC.00316-07
Marciano-Cabral F, Cabral G (2003) Acanthamoeba spp. as agents of disease in humans. Clin Microbiol Rev 16:273–307. https://doi.org/10.1128/CMR.16.2.273-307.2003
Margis R, Dunand C, Teixeira FK, Margis-Pinheiro M (2008) Glutathione peroxidase family – an evolutionary overview. FEBS J 275:3959–3970. https://doi.org/10.1111/j.1742-4658.2008.06542.x
Matthews JL, Zwick MG, Paule MR (1995) Coordinate regulation of ribosomal component synthesis in Acanthamoeba castellanii: 5S RNA transcription is down regulated during encystment by alteration of TFIIIA activity. Mol Cell Biol 15:3327–3335
Maxwell DP, Wang Y, McIntosh L (1999) The alternative oxidase lowers mitochondrial reactive oxygen production in plant cells. Proc Natl Acad Sci U S A 96:8271–8276. https://doi.org/10.1073/PNAS.96.14.8271
Moon EK, Il CD, Hong YC, Kong HH (2007) Differentially expressed genes of Acanthamoeba castellanii during encystation. Korean J Parasitol 45:283–285
Moon EK, Il CD, Hong YC, Kong HH (2009) Autophagy protein 8 mediating autophagosome in encysting Acanthamoeba. Mol Biochem Parasitol 168:43–48. https://doi.org/10.1016/j.molbiopara.2009.06.005
Moon E-K, Hong Y, Chung D-I et al (2014) Down-regulation of cellulose synthase inhibits the formation of endocysts in Acanthamoeba. Korean J Parasitol 52:131–135
Moon EK, Hong Y, Il CD et al (2016) Identification of protein arginine methyltransferase 5 as a regulator for encystation of Acanthamoeba. Korean J Parasitol 54:133–138. https://doi.org/10.3347/kjp.2016.54.2.133
Moon EK, Hong Y, Il CD, Kong HH (2012) Cysteine protease involving in autophagosomal degradation of mitochondria during encystation of Acanthamoeba. Mol Biochem Parasitol 185:121–126. https://doi.org/10.1016/j.molbiopara.2012.07.008
Moon EK, Kong HH (2012) Short-cut pathway to synthesize cellulose of encysting Acanthamoeba. Korean J Parasitol 50:361–364. https://doi.org/10.3347/kjp.2012.50.4.361
Natvig DO, Sylvester K, Dvorachek WH, Baldwin JL (1996) Superoxide dismutases and catalases. Biochem Mol Biol 191–209. https://doi.org/10.1007/978-3-662-10367-8_8
Okada H, Ueda M, Sugaya T et al (1987) Catalase gene of the yeast Candida tropicalis. Eur J Biochem 170:105–110. https://doi.org/10.1111/j.1432-1033.1987.tb13673.x
Palmer JM (2003) The organization and regulation of electron transport in plant mitochondria. Annu Rev Plant Physiol 27:133–157. https://doi.org/10.1146/ANNUREV.PP.27.060176.001025
Purvis AC (1997) Role of the alternative oxidase in limiting superoxide production by plant mitochondria. Physiol Plant 100:165–170. https://doi.org/10.1111/J.1399-3054.1997.TB03468.X
Purvis AC, Shewfelt RL (1993) Does the alternative pathway ameliorate chilling injury in sensitive plant tissues? Physiol Plant 88:712–718. https://doi.org/10.1111/J.1399-3054.1993.TB01393.X
Ritchie ME, Phipson B, Wu D et al (2015) Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43:e47. https://doi.org/10.1093/nar/gkv007
Robinson MD, McCarthy DJ, Smyth GK (2009) edgeR: a bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26:139–140. https://doi.org/10.1093/bioinformatics/btp616
Sakajo S, Minagawa N, Yoshimoto A (1993) Characterization of the alternative oxidase protein in the yeast Hansenula anomala. FEBS Lett 318:310–312. https://doi.org/10.1016/0014-5793(93)80535-3
Smith GA, Lin TH, Sheehan AE et al (2019) Glutathione S-transferase regulates mitochondrial populations in axons through increased glutathione oxidation. Neuron 103:52-65.e6. https://doi.org/10.1016/j.neuron.2019.04.017
Song SM, Han BI, Moon EK et al (2012) Autophagy protein 16-mediated autophagy is required for the encystation of Acanthamoeba castellanii. Mol Biochem Parasitol 183:158–165. https://doi.org/10.1016/j.molbiopara.2012.02.013
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38:3022–3027
Umbach AL, Siedow JN (1993) Covalent and noncovalent dimers of the cyanide-resistant alternative oxidase protein in higher plant mitochondria and their relationship to enzyme activity. Plant Physiol 103:845–854. https://doi.org/10.1104/pp.103.3.845
Vanlerberghe GC (2013) Alternative oxidase: a mitochondrial respiratory pathway to maintain metabolic and signaling homeostasis during abiotic and biotic stress in plants. Int J Mol Sci 14:6805. https://doi.org/10.3390/IJMS14046805
Vanlerberghe GC, Day DA, Wiskich JT et al (1995) Alternative oxidase activity in tobacco leaf mitochondria (dependence on tricarboxylic acid cycle-mediated redox regulation and pyruvate activation). Plant Physiol 109:353–361. https://doi.org/10.1104/PP.109.2.353
Vasanthakumar T, Rubinstein JL (2020) Structure and roles of V-type ATPases. Trends Biochem Sci 45:295–307. https://doi.org/10.1016/j.tibs.2019.12.007
Vickerman K (1960) Structural changes in mitochondria of Acanthamoeba at encystation. Nature 188:248–249. https://doi.org/10.1038/188248a0
Wagner AM (1995) A role for active oxygen species as second messengers in the induction of alternative oxidase gene expression in Petunia hybrida cells. FEBS Lett 368:339–342. https://doi.org/10.1016/0014-5793(95)00688-6
Wagner AM, Moore AL (1997) Structure and function of the plant alternative oxidase: its putative role in the oxygen defence mechanism. Biosci Rep 17:319–333. https://doi.org/10.1023/A:1027388729586
Funding
The study was supported by Mexico’s National Council of Science and Technology (CONACYT) and a University of Edinburgh College of Medicine and Veterinary Medicine PhD Studentship (to AdOFdV). Edinburgh Genomics is partly supported through core grants from NERC (R8/H10/56), MRC (MR/K001744/1), and BBSRC (BB/J004243/1).
Author information
Authors and Affiliations
Contributions
Conceptualization: CQS, SKM, and AdOFdV; methodology: CQS, RDM, and AdOFdV; formal analysis and investigation: CQS, RDM, JGM, and AdOFdV; writing—original draft preparation: CQS, JGM, and AdOFdV; writing—review and editing: CQS, SKM, and AdOFdV; funding acquisition: AdOFdV; resources: SKM and AdOFdV; supervision: SKM and AdOFdV.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
Sutherland K. Maciver is part of the editorial board of Parasitology Research.
Additional information
Handling Editor: Julia Walochnik
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Scheckhuber, C.Q., Damián Ferrara, R., Gómez-Montalvo, J. et al. Oxidase enzyme genes are differentially expressed during Acanthamoeba castellanii encystment. Parasitol Res 123, 116 (2024). https://doi.org/10.1007/s00436-024-08138-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00436-024-08138-9