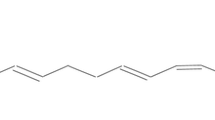
Abstract
Encystment is crucial for defense and reproduction in Cryptocaryon irritans. Therefore, understanding the encystment-related events in the protomont stage can help prevent and control C. irritans. Autophagy promotes protozoan parasite encystation. However, 3MA can inhibit autophagy. In this study, the effects of autophagy inhibition on encystation, survival rate, ultrastructural features, and metabolomic profiles of C. irritans, were evaluated using protomonts treated with 3MA (20 mM). The treatment with 3MA for about 4 h significantly lowered survival and encystation rates of protomonts to about 86.44% and 76.08%, respectively. Microstructural observations showed that the 3MA-treated protomonts showed deformed cell membranes and the cytoplasmic content spill. Furthermore, observation of the ultrastructure of 3MA-treated protomonts showed the destruction of organelles (Golgi bodies and mucocyst) and a lack of autophagosomes. However, no abnormality was observed in the control experiments. Furthermore, the metabolic analysis revealed suppression of metabolites, such as lipids, amino acids, and carbohydrates. These results demonstrate that 3MA can inhibit autophagy in C. irritans, thus hindering encystation, suppressing the metabolism of metabolites, and altering morphological ultrastructure in these parasites.
Graphical Abstract






Similar content being viewed by others
Data availability
The experimental materials and data are true and reliable, and have not been disclosed to other journals except this journal.
References
Aslan E et al (2017) A comparative in-silico analysis of autophagy proteins in ciliates. PeerJ 5:e2878. https://doi.org/10.7717/peerj.2878
Bess AS et al (2012) Mitochondrial dynamics and autophagy aid in removal of persistent mitochondrial DNA damage in Caenorhabditis elegans. Nucleic Acids Res 40:7916–7931. https://doi.org/10.1093/nar/gks532
Bursch W et al (2008) Cell death and autophagy: cytokines, drugs, and nutritional factors. Toxicol 254:147–157. https://doi.org/10.1016/j.tox.2008.07.048
Cardenas-Zuniga R et al (2017) Ubiquitin-like Atg8 protein is expressed during autophagy and the encystation process in Naegleria gruberi. Parasitol Res 116:303–312. https://doi.org/10.1007/s00436-016-5293-x
Cervera L et al (2022) Severe natural outbreak of Cryptocaryon irritans in gilthead seabream produces leukocyte mobilization and innate immunity at the gill tissue. Int J Mol Sci 23:937. https://doi.org/10.3390/ijms23020937
Colorni A, Burgess P (1997) Cryptocaryon irritans Brown 1951, the cause of ‘white spot disease’ in marine fish: an update. Aquar Sci Conserv 1:217–238. https://doi.org/10.1023/A:1018360323287
Colorni A, Diamant A (1993) Ultrastructural features of Cryptocaryon irritans, a ciliate parasite of marine fish. Eur J Protistol 29:425–434. https://doi.org/10.1016/s0932-4739(11)80405-0
Diao C et al (2019) Aged kidneys are refractory to autophagy activation in a rat model of renal ischemia-reperfusion injury. Clin Interv Aging 14:525–534. https://doi.org/10.2147/cia.S197444
Duszenko M et al (2011) Autophagy in protists. Autophagy 7:127–158. https://doi.org/10.4161/auto.7.2.13310
Ekman DR et al (2015) Metabolite profiling of fish skin mucus: a novel approach for minimally-invasive environmental exposure monitoring and surveillance. Environ Sci Technol 49:3091–3100. https://doi.org/10.1021/es505054f
Gutierrez JC et al (2001) Ciliate cryptobiosis: a microbial strategy against environmental starvation. Int Microbiol 4:151–157. https://doi.org/10.1007/s10123-001-0030-3
Huang KY et al (2014) Adaptive responses to glucose restriction enhance cell survival, antioxidant capability, and autophagy of the protozoan parasite Trichomonas vaginalis. Biochim Biophysic Acta 1840:53–64. https://doi.org/10.1016/j.bbagen.2013.08.008
Jeelani G et al (2012) Metabolic profiling of the protozoan parasite Entamoeba invadens revealed activation of unpredicted pathway during encystation. PLoS ONE 7:37740. https://doi.org/10.1371/journal.pone.0037740
Jiang B et al (2018) The development of Cryptocaryon irritans in a less susceptible host rabbitfish, Siganus oramin. Parasitol Res 117:3835–3842. https://doi.org/10.1007/s00436-018-6088-z
Levine B, Klionsky DJ (2004) Development by self-digestion: molecular mechanisms and biological functions of autophagy. Devp Cell 6:463–477. https://doi.org/10.1016/s1534-5807(04)00099-1
Li Yw et al (2021) Cryptocaryon irritans (Brown, 1951) is a serious threat to aquaculture of marine fish. https://doi.org/10.1111/raq.12594
Lokanathan Y et al (2010) Transcriptome analysis of the Cryptocaryon irritans tomont stage identifies potential genes for the detection and control of cryptocaryonosis. BMC Genom 11:76. https://doi.org/10.1186/1471-2164-11-76
Ma R et al (2017) Ultrastructural features of the tomont of Cryptocaryon irritans (Ciliophora: Prostomatea), a parasitic ciliate of marine fishes. Parasitol 144:720–729. https://doi.org/10.1017/s0031182016002651
Ma R et al (2016) Ultrastructure observation on the cells at different life history stages of Cryptocaryon irritans (Ciliophora: Prostomatea), a parasitic ciliate of marine fishes. Parasitol 143:1479–1489. https://doi.org/10.1017/s0031182016001074
Moon EK et al (2012) Cysteine protease involving in autophagosomal degradation of mitochondria during encystation of Acanthamoeba. Mol Biochem Parasitol 185:121–126. https://doi.org/10.1016/j.molbiopara.2012.07.008
Morais P et al (2009) Resveratrol induces mitochondrial alterations, autophagy and a cryptobiosis-like state in scuticociliates. Protist 160:552–564. https://doi.org/10.1016/j.protis.2009.04.004
O’Donnell VB et al (2019) LIPID MAPS: Serving the next generation of lipid researchers with tools, resources, data, and training. Sci Signal 12:eaww2964. https://doi.org/10.1126/scisignal.aaw2964
Picazarri K et al (2008) Autophagy during proliferation and encystation in the protozoan parasite Entamoeba invadens. Infect Immun 76:278–288. https://doi.org/10.1128/iai.00636-07
Seglen PO, Gordon PB (1982) 3-Methyladenine: specific inhibitor of autophagic/lysosomal protein degradation in isolated rat hepatocytes. Proc Nat Acad Sci USA 79:1889–1892. https://doi.org/10.1073/pnas.79.6.1889
Seglen PO et al (1980) Amino acid control of protein synthesis and degradation in isolated rat hepatocytes. Ann NY Acad Sci 349:1–17. https://doi.org/10.1111/j.1749-6632.1980.tb29510.x
Sheng Y et al (2013) 3-Methyladenine induces cell death and its interaction with chemotherapeutic drugs is independent of autophagy. Biochem Biophysic Res Commun 432:5–9. https://doi.org/10.1016/j.bbrc.2013.01.106
Thevenot EA et al (2015) Analysis of the human adult urinary metabolome variations with age, body mass index, and gender by implementing a comprehensive workflow for univariate and OPLS statistical analyses. J Proteom Res 14:3322–3335
Tourancheau AB et al (1999) Messenger RNA in dormant cells of Sterkiella histriomuscorum (Oxytrichiade): indentification of putative regulatory gene transcripts. Protist 150:137–147. https://doi.org/10.1016/s1434-4610(99)70017-9
Want EJ et al (2013) Global metabolic profiling of animal and human tissues via UPLC-MS. Nat Protoc 8:17–32. https://doi.org/10.1038/nprot.2012.135
Wishart DS et al (2018) HMDB 4.0: the human metabolome database for 2018. Nucleic Acids Res 46:608–617. https://doi.org/10.1093/nar/gkx1089
Wu YT et al (2010) Dual role of 3-methyladenine in modulation of autophagy via different temporal patterns of inhibition on class I and III phosphoinositide 3-kinase. J Biol Chem 285:10850–10861. https://doi.org/10.1074/jbc.M109.080796
Xie X et al (2021) Integration of metabolomic and transcriptomic analyses to characterize the influence of the gill metabolism of Nibea albiflora on the response to Cryptocaryon irritans infection. Vet Parasitol 298:109533. https://doi.org/10.1016/j.vetpar.2021.109533
Xu L, Shao X (2004) Methods of chemometrics. Sci-ence publishing company, Beijing, p 258
Yin F et al (2015) Stress, antioxidant defence and mucosal immune responses of the large yellow croaker Pseudosciaena crocea challenged with Cryptocaryon irritans. Fish Shellfish Immunol 47:344–351. https://doi.org/10.1016/j.fsi.2015.09.013
Zhong Z-H et al (2019) Antiparasitic efficacy of honokiol against Cryptocaryon irritans in pompano, Trachinotus ovatus. Aquac 500:398–406. https://doi.org/10.1016/j.aquaculture.2018.10.037
Zhou L et al (2022) pH regulates the formation and hatching of Cryptocaryon irritans tomonts, which affects Cryptocaryoniasis occurrence in Larimichthys crocea Aquaculture. Appl Environ Microbiol 88:e0005822. https://doi.org/10.1128/aem.00058-22
Acknowledgements
The completion of this manuscript could not have been possible without the support and guidance of my all lab teachers and colleagues Liyao Zhou, Ruiling Zhou, and Lingling Wang. They had helped me for purchasing experimental tools. Their contributions are sincerely appreciated and gratefully acknowledged. Also, we deeply acknowledge funding support.
Funding
This work was supported by the Key program of Science and Technology of Zhejiang Province (grant No.2021C02024), the Ningbo Public Welfare Project (grant No.2022S159; 202002N3042),the Shanghai Natural Science Foundation (grant No. 17ZR1439600), the Shanghai Natural Science Foundation (grant No. 17ZR1439600), and the National Key Research and Development Project of China (grant No.2019YFD0900102).
Author information
Authors and Affiliations
Contributions
Bushra, Ivon F. Maha, Xiao Xie, and Fei Yin conceived and designed the study. Ivon F. Maha, Bushra, Youbin Yu, and Linhua Jiang performed animal feeding and laboratory experiments, analyzed data, interpreted the data, and prepared figures. Bushra and Ivon F. Maha wrote the manuscript and contributed equally to the study. Fei Yin, Summing Zhou, and Xiao Xie interpret the data and revise the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All animal and parasite experiments were performed according to local and central government regulations. The Institutional Animal Care and Use Committee of the Ningbo University approved all experiments. Informed consent was obtained from all individual participants included in the study.
Consent for publication
The participant has consented to the submission of the case report to the journal.
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Una Ryan
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bushra, Maha, I.F., Yu, Y. et al. Effects of autophagy inhibition by 3-methyladenine on encystation, morphology, and metabolites of Cryptocaryon irritans. Parasitol Res 122, 509–517 (2023). https://doi.org/10.1007/s00436-022-07751-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-022-07751-w