Abstract
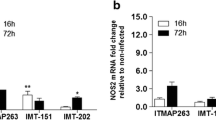
Visceral leishmaniasis is a life-threatening disease characterized by intense parasitism of the spleen, liver, and bone marrow. Antimonials have served as front-line antileishmanial therapeutics for decades, but the increasing failure rates under antimonial treatment have challenged the continued use of these drugs. Pentavalent antimonials are known to reinforce the killing mechanisms of macrophages, although the associated mechanism remains unclear. Here, for the first time, we determined whether Leishmania infantum strains isolated from patients refractory to antimony treatment (relapse cases) were cross-resistant to antimonials, liposomal amphotericin B, and/or nitric oxide, and also whether these strains modulate macrophage infection. We selected four clinical isolates from relapse cases and two clinical isolates from antimony-responsive patients (control group) for the present study. The L. infantum promastigotes from all four relapse cases were resistant to trivalent antimonial treatment and nitric oxide, while only one isolate was resistant to liposomal amphotericin B. We evaluated whether the resistant strains from relapse cases showed enhanced infectivity and amastigote survival in macrophages, or macrophage-killing mechanisms in macrophages activated by lipopolysaccharide plus interferon gamma. Infection indexes calculated using macrophages infected with isolates from relapse were higher than those observed with control strains that were stimulated independently. Macrophage infection was higher with L. infantum isolates from relapse cases and correlated with enhanced interleukin 1-β production but showed similar nitrite production. Our results demonstrate that L. infantum field isolates from relapse cases were resistant to antimonials and nitric oxide and that these parasites stimulated inflammatory cytokines and were resistant to macrophage-killing mechanisms, factors that may contribute to disease severity.




Similar content being viewed by others
References
Ait-Oudhia K, Gazanion E, Vergnes B, Oury B, Sereno D (2011) Leishmania antimony resistance: what we know what we can learn from the field. Parasitol Res 109:1225–1232. doi:10.1007/s00436-011-2555-5
Assreuy J, Cunha FQ, Epperlein M, Noronha-Dutra A, O’Donnell CA, Liew FY, Moncada S (1994) Production of nitric oxide and superoxide by activated macrophages and killing of Leishmania major. Eur J Immunol 24:672–676. doi:10.1002/eji.1830240328
Badaro R, Falcoff E, Badaro FS, Carvalho EM, Pedral-Sampaio D, Barral A, Carvalho JS, Barral-Netto M, Brandely M, Silva L et al (1990) Treatment of visceral leishmaniasis with pentavalent antimony and interferon gamma. N Engl J Med 322:16–21. doi:10.1056/NEJM199001043220104
Bagher Khadem Erfan M, Mohebali M, Kazemi-Rad E, Hajjaran H, Edrissian G, Mamishi S, Saffari M, Raoofian R, Heidari M (2013) Downregulation of calcineurin gene is associated with glucantime ((R)) resiatance in leishmania infantum. Iran J Parasitol 8:359–366
Carrio J, Portus M (2002) In vitro susceptibility to pentavalent antimony in Leishmania infantum strains is not modified during in vitro or in vivo passages but is modified after host treatment with meglumine antimoniate. BMC Pharmacol 2:11
Carrio J, Riera C, Gallego M, Portus M (2001) In vitro activity of pentavalent antimony derivatives on promastigotes and intracellular amastigotes of Leishmania infantum strains from humans and dogs in Spain. Acta Trop 79:179–183
Carter KC, Sundar S, Spickett C, Pereira OC, Mullen AB (2003) The in vivo susceptibility of Leishmania donovani to sodium stibogluconate is drug specific and can be reversed by inhibiting glutathione biosynthesis. Antimicrob Agents Chemother 47:1529–1535
Carter KC, Hutchison S, Henriquez FL, Legare D, Ouellette M, Roberts CW, Mullen AB (2006) Resistance of Leishmania donovani to sodium stibogluconate is related to the expression of host and parasite gamma-glutamylcysteine synthetase. Antimicrob Agents Chemother 50:88–95. doi:10.1128/AAC.50.1.88-95.2006
Chandra D, Naik S (2008) Leishmania donovani infection down-regulates TLR2-stimulated IL-12p40 and activates IL-10 in cells of macrophage/monocytic lineage by modulating MAPK pathways through a contact-dependent mechanism. Clin Exp Immunol 154:224–234. doi:10.1111/j.1365-2249.2008.03741.x
Cohen ML (1992) Epidemiology of drug resistance: implications for a post-antimicrobial era. Science 257:1050–1055
Croft SL, Sundar S, Fairlamb AH (2006) Drug resistance in leishmaniasis. Clin Microbiol Rev 19:111–126. doi:10.1128/CMR.19.1.111-126.2006
de Azevedo AF, Dutra JL, Santos ML, Santos Dde A, Alves PB, de Moura TR, de Almeida RP, Fernandes MF, Scher R, Fernandes RP (2014) Fatty acid profiles in Leishmania spp. isolates with natural resistance to nitric oxide and trivalent antimony. Parasitol Res 113:19–27. doi:10.1007/s00436-013-3621-y
de Melo EC, Fortaleza CM (2013) Challenges in the therapy of visceral leishmaniasis in Brazil: a public health perspective. J Trop Med 2013:319234. doi:10.1155/2013/319234
Decuypere S, Rijal S, Yardley V, De Doncker S, Laurent T, Khanal B, Chappuis F, Dujardin JC (2005) Gene expression analysis of the mechanism of natural Sb (V) resistance in Leishmania donovani isolates from Nepal. Antimicrob Agents Chemother 49:4616–4621
Decuypere S, Vanaerschot M, Brunker K, Imamura H, Muller S, Khanal B, Rijal S, Dujardin JC, Coombs GH (2012) Molecular mechanisms of drug resistance in natural Leishmania populations vary with genetic background. PLoS Negl Trop Dis 6:e1514
Di Giorgio C, Faraut-Gambarelli F, Imbert A, Minodier P, Gasquet M, Dumon H (1999) Flow cytometric assessment of amphotericin B susceptibility in Leishmania infantum isolates from patients with visceral leishmaniasis. J Antimicrob Chemother 44:71–76
Escobar P, Matu S, Marques C, Croft SL (2002) Sensitivities of Leishmania species to hexadecylphosphocholine (miltefosine), ET-18-OCH (3) (edelfosine) and amphotericin B. Acta Trop 81:151–157
Faraut-Gambarelli F, Piarroux R, Deniau M, Giusiano B, Marty P, Michel G, Faugere B, Dumon H (1997) In vitro and in vivo resistance of Leishmania infantum to meglumine antimoniate: a study of 37 strains collected from patients with visceral leishmaniasis. Antimicrob Agents Chemother 41:827–830
Feng GJ, Goodridge HS, Harnett MM, Wei XQ, Nikolaev AV, Higson AP, Liew FY (1999) Extracellular signal-related kinase (ERK) and p38 mitogen-activated protein (MAP) kinases differentially regulate the lipopolysaccharide-mediated induction of inducible nitric oxide synthase and IL-12 in macrophages: Leishmania phosphoglycans subvert macrophage IL-12 production by targeting ERK MAP kinase. J Immunol 163:6403–6412
Genestra M, Guedes-Silva D, Souza WJ, Cysne-Finkelstein L, Soares-Bezerra RJ, Monteiro FP, Leon LL (2006) Nitric oxide synthase (NOS) characterization in Leishmania amazonensis axenic amastigotes. Arch Med Res 37:328–333
Giudice A, Camada I, Leopoldo PT, Pereira JM, Riley LW, Wilson ME, Ho JL, de Jesus AR, Carvalho EM, Almeida RP (2007) Resistance of Leishmania (Leishmania) amazonensis and Leishmania (Viannia) braziliensis to nitric oxide correlates with disease severity in tegumentary leishmaniasis. BMC Infect Dis 7:7
Goyeneche-Patino DA, Valderrama L, Walker J, Saravia NG (2008) Antimony resistance and trypanothione in experimentally selected and clinical strains of Leishmania panamensis. Antimicrob Agents Chemother 52:4503–4506. doi:10.1128/AAC.01075-08
Gramiccia M, Gradoni L, Orsini S (1992) Decreased sensitivity to meglumine antimoniate (Glucantime) of Leishmania infantum isolated from dogs after several courses of drug treatment. Ann Trop Med Parasitol 86:613–620
Holzmuller P, Sereno D, Lemesre JL (2005) Lower nitric oxide susceptibility of trivalent antimony-resistant amastigotes of Leishmania infantum. Antimicrob Agents Chemother 49:4406–4409
Jeddi F, Mary C, Aoun K, Harrat Z, Bouratbine A, Faraut F, Benikhlef R, Pomares C, Pratlong F, Marty P, Piarroux R (2014) Heterogeneity of molecular resistance patterns in antimony-resistant field isolates of Leishmania species from the Western Mediterranean area. Antimicrob Agents Chemother 58:4866–4874. doi:10.1128/AAC.02521-13
Kothari H, Kumar P, Sundar S, Singh N (2007) Possibility of membrane modification as a mechanism of antimony resistance in Leishmania donovani. Parasitol Int 56:77–80. doi:10.1016/j.parint.2006.10.005
Lachaud L, Marchergui-Hammami S, Chabbert E, Dereure J, Dedet JP, Bastien P (2002) Comparison of six PCR methods using peripheral blood for detection of canine visceral leishmaniasis. J Clin Microbiol 40:210–215
Lima-Junior DS, Costa DL, Carregaro V, Cunha LD, Silva AL, Mineo TW, Gutierrez FR, Bellio M, Bortoluci KR, Flavell RA, Bozza MT, Silva JS, Zamboni DS (2013) Inflammasome-derived IL-1beta production induces nitric oxide-mediated resistance to Leishmania. Nat Med 19:909–915
Lonardoni MV, Russo M, Jancar S (2000) Essential role of platelet-activating factor in control of Leishmania (Leishmania) amazonensis infection. Infect Immun 68:6355–6361
Mauel J, Buchmuller-Rouiller Y (1987) Effect of lipopolysaccharide on intracellular killing of Leishmania enriettii and correlation with macrophage oxidative metabolism. Eur J Immunol 17:203–208. doi:10.1002/eji.1830170209
Mikus J, Steverding D (2000) A simple colorimetric method to screen drug cytotoxicity against Leishmania using the dye Alamar Blue. Parasitol Int 48:265–269
Mishra M, Biswas UK, Jha DN, Khan AB (1992) Amphotericin versus pentamidine in antimony-unresponsive kala-azar. Lancet 340:1256–1257
Mittal MK, Rai S, Ashutosh R, Gupta S, Sundar S, Goyal N (2007) Characterization of natural antimony resistance in Leishmania donovani isolates. Am J Trop Med Hyg 76:681–688
Mookerjee Basu J, Mookerjee A, Sen P, Bhaumik S, Sen P, Banerjee S, Naskar K, Choudhuri SK, Saha B, Raha S, Roy S (2006) Sodium antimony gluconate induces generation of reactive oxygen species and nitric oxide via phosphoinositide 3-kinase and mitogen-activated protein kinase activation in Leishmania donovani-infected macrophages. Antimicrob Agents Chemother 50:1788–1797. doi:10.1128/AAC.50.5.1788-1797.2006
Mukherjee B, Mukhopadhyay R, Bannerjee B, Chowdhury S, Mukherjee S, Naskar K, Allam US, Chakravortty D, Sundar S, Dujardin JC, Roy S (2013) Antimony-resistant but not antimony-sensitive Leishmania donovani up-regulates host IL-10 to overexpress multidrug-resistant protein 1. Proc Natl Acad Sci U S A 110:E575–E582. doi:10.1073/pnas.1213839110
Ouellette M, Drummelsmith J, Papadopoulou B (2004) Leishmaniasis: drugs in the clinic, resistance and new developments. Drug Resist Updat 7:257–266. doi:10.1016/j.drup.2004.07.002
Purkait B, Kumar A, Nandi N, Sardar AH, Das S, Kumar S, Pandey K, Ravidas V, Kumar M, De T, Singh D, Das P (2012) Mechanism of amphotericin B resistance in clinical isolates of Leishmania donovani. Antimicrob Agents Chemother 56:1031–1041. doi:10.1128/AAC.00030-11
Ralph P, Nakoinz I (1975) Phagocytosis and cytolysis by a macrophage tumour and its cloned cell line. Nature 257:393–394
Romero GA, Boelaert M (2010) Control of visceral leishmaniasis in latin america-a systematic review. PLoS Negl Trop Dis 4:e584. doi:10.1371/journal.pntd.0000584
Santos PL, Costa RV, Braz JM, Santos LF, Batista AC, Vasconcelos CR, Rangel MR, Ribeiro de Jesus A, de Moura TR, Leopoldo PT, Almeida RP (2012) Leishmania chagasi naturally resistant to nitric oxide isolated from humans and dogs with visceral leishmaniasis in Brazil. Nitric Oxide 27:67–71. doi:10.1016/j.niox.2012.04.004
Souza AS, Giudice A, Pereira JM, Guimaraes LH, de Jesus AR, de Moura TR, Wilson ME, Carvalho EM, Almeida RP (2010) Resistance of Leishmania (Viannia) braziliensis to nitric oxide: correlation with antimony therapy and TNF-alpha production. BMC Infect Dis 10:209. doi:10.1186/1471-2334-10-209
Sundar S, Chakravarty J (2013) Leishmaniasis: an update of current pharmacotherapy. Expert Opin Pharmacother 14:53–63. doi:10.1517/14656566.2013.755515
Sundar S, More DK, Singh MK, Singh VP, Sharma S, Makharia A, Kumar PC, Murray HW (2000) Failure of pentavalent antimony in visceral leishmaniasis in India: report from the center of the Indian epidemic. Clin Infect Dis 31:1104–1107. doi:10.1086/318121
Sundar S, Chakravarty J, Rai VK, Agrawal N, Singh SP, Chauhan V, Murray HW (2007) Amphotericin B treatment for Indian visceral leishmaniasis: response to 15 daily versus alternate-day infusions. Clin Infect Dis 45:556–561. doi:10.1086/520665
Teixeira AC, Paes MG, Guerra Jde O, Prata A, Silva-Vergara ML (2008) Failure of both azithromycin and antimony to treat cutaneous leishmaniasis in Manaus, AM, Brazil. Rev Inst Med Trop Sao Paulo 50:157–160
t’Kindt R, Scheltema RA, Jankevics A, Brunker K, Rijal S, Dujardin JC, Breitling R, Watson DG, Coombs GH, Decuypere S (2010) Metabolomics to unveil and understand phenotypic diversity between pathogen populations. PLoS Negl Trop Dis 4:e904. doi:10.1371/journal.pntd.0000904
Vanaerschot M, De Doncker S, Rijal S, Maes L, Dujardin JC, Decuypere S (2011) Antimonial resistance in Leishmania donovani is associated with increased in vivo parasite burden. PLoS ONE 6:e23120. doi:10.1371/journal.pone.0023120
Zauli-Nascimento RC, Miguel DC, Yokoyama-Yasunaka JK, Pereira LI, Pelli de Oliveira MA, Ribeiro-Dias F, Dorta ML, Uliana SR (2010) In vitro sensitivity of Leishmania (Viannia) braziliensis and Leishmania (Leishmania) amazonensis Brazilian isolates to meglumine antimoniate and amphotericin B. Trop Med Int Health 15:68–76. doi:10.1111/j.1365-3156.2009.02414.x
Acknowledgments
We thank members of the Infectious Disease Clinic Group from the University Hospital, Universidade Federal de Sergipe.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Ethical approval was obtained from the Hospital Universitário from Universidade Federal de Sergipe, CAAE 0151.0.107.000-07.
Conflict of interest
The authors declare that they have no competing interests.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
de Moura, T.R., Santos, M.L.B., Braz, J.M. et al. Cross-resistance of Leishmania infantum isolates to nitric oxide from patients refractory to antimony treatment, and greater tolerance to antileishmanial responses by macrophages. Parasitol Res 115, 713–721 (2016). https://doi.org/10.1007/s00436-015-4793-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-015-4793-4