Abstract
Purpose
Lung cancer is the leading cause of cancer-related death worldwide. The early detection of lung cancer is crucial for the diagnosis of this disease. Therefore, an effective and noninvasive method for the early diagnosis of lung cancer is urgently needed.
Methods
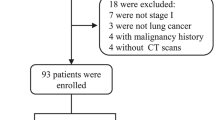
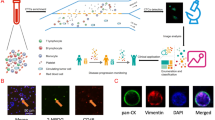
To evaluate the diagnostic performance of circulating genetically abnormal cells (CACs) in early lung cancer, a total of 63 participants who completed CAC detection by Zhuhai SanMed Biotech Inc. and obtained pathological results from January to December 2020 were included in our study; 50 patients had lung cancer and 13 patients had benign lung disease. The levels of lung cancer-related markers in peripheral blood and the chest computed tomography (CT) imaging characteristics of these patients were collected before pathological acquisition.
Results
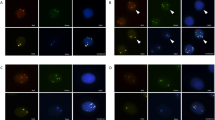
The positive rate of CAC was 90.0% in the lung cancer group and 23.1% in the benign lung disease group, and the difference was statistically significant (P < 0.01). The area under the receiver operating characteristic (ROC) curve of CAC was 0.837, the sensitivity was 90%, and the specificity was 76.9%. The area under the ROC curve and sensitivity were both higher than those of the combined or single serum tumor marker test.
Conclusions
This study preliminarily concludes that the CAC test, as a noninvasive test, has high sensitivity and specificity for the early diagnosis of lung cancer. This test is expected to help with the early detection of disease in lung cancer patients.








Similar content being viewed by others
Availability of data and material
The data and materials are true and valid.
References
Aberle DR, Adams AM, Berg CD, Black WC, Clapp JD, Fagerstrom RM, Gareen IF, Gatsonis C, Marcus PM, Sicks JD (2011) Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 365:395–409. https://doi.org/10.1056/NEJMoa1102873
Bade BC, Dela Cruz CS (2020) Lung cancer 2020: epidemiology, etiology, and prevention. Clin Chest Med 41:1–24. https://doi.org/10.1016/j.ccm.2019.10.001
Blackhall F, Frese KK, Simpson K, Kilgour E, Brady G, Dive C (2018) Will liquid biopsies improve outcomes for patients with small-cell lung cancer? Lancet Oncol 19:e470–e481. https://doi.org/10.1016/s1470-2045(18)30455-8
Brodersen J, Voss T, Martiny F, Siersma V, Barratt A, Heleno B (2020) Overdiagnosis of lung cancer with low-dose computed tomography screening: meta-analysis of the randomised clinical trials. Breathe (Sheff) 16:200013. https://doi.org/10.1183/20734735.0013-2020
Cao M, Chen W (2019) Epidemiology of lung cancer in China. Thorac Cancer 10:3–7. https://doi.org/10.1111/1759-7714.12916
Carter BW, Lichtenberger JP 3rd, Benveniste MK, de Groot PM, Wu CC, Erasmus JJ, Truong MT (2018) Revisions to the TNM staging of lung cancer: rationale, significance, and clinical application. Radiographics 38:374–391. https://doi.org/10.1148/rg.2018170081
Cohen JD, Li L, Wang Y et al (2018) Detection and localization of surgically resectable cancers with a multi-analyte blood test. Science 359:926–930. https://doi.org/10.1126/science.aar3247
Detterbeck FC, Mazzone PJ, Naidich DP, Bach PB (2013) Screening for lung cancer: diagnosis and management of lung cancer, 3rd ed: American college of chest physicians evidence-based clinical practice guidelines. Chest 143:e78S-e92S. https://doi.org/10.1378/chest.12-2350
Ferreira MM, Ramani VC, Jeffrey SS (2016) Circulating tumor cell technologies. Mol Oncol 10:374–394. https://doi.org/10.1016/j.molonc.2016.01.007
Gareen IF, Duan F, Greco EM, Snyder BS, Boiselle PM, Park ER, Fryback D, Gatsonis C (2014) Impact of lung cancer screening results on participant health-related quality of life and state anxiety in the National lung screening trial. Cancer 120:3401–3409. https://doi.org/10.1002/cncr.28833
Gould MK, Tang T, Liu IL, Lee J, Zheng C, Danforth KN, Kosco AE, Di Fiore JL, Suh DE (2015) Recent trends in the identification of incidental pulmonary nodules. Am J Respir Crit Care Med 192:1208–1214. https://doi.org/10.1164/rccm.201505-0990OC
Hirsch FR, Scagliotti GV, Mulshine JL, Kwon R, Curran WJ, Jr., Wu YL, Paz-Ares L (2017) Lung cancer: current therapies and new targeted treatments. Lancet 389:299–311. https://doi.org/10.1016/s0140-6736(16)30958-8. https://www.cancer.org/treatment/understanding-your-diagnosis/staging
IARC (2020) Latest global cancer data: cancer burden rises to 19.3 million new cases and 10.0 million cancer deaths in 2020. https://www.iarc.who.int/fr/news-events/latest-global-cancer-data-cancer-burden-rises-to-19-3-million-new-cases-and-10-0-million-cancer-deaths-in-2020/
Karachaliou N, Mayo-de-Las-Casas C, Molina-Vila MA, Rosell R (2015) Real-time liquid biopsies become a reality in cancer treatment. Ann Transl Med 3:36. https://doi.org/10.3978/j.issn.2305-5839.2015.01.16
Katz RL, He W, Khanna A et al (2010) Genetically abnormal circulating cells in lung cancer patients: an antigen-independent fluorescence in situ hybridization-based case-control study. Clin Cancer Res 16:3976–3987. https://doi.org/10.1158/1078-0432.ccr-09-3358
Katz RL, Zaidi TM, Pujara D, Shanbhag ND, Truong D, Patil S, Mehran RJ, El-Zein RA, Shete SS, Kuban JD (2020) Identification of circulating tumor cells using 4-color fluorescence in situ hybridization: validation of a noninvasive aid for ruling out lung cancer in patients with low-dose computed tomography-detected lung nodules. Cancer Cytopathol 128:553–562. https://doi.org/10.1002/cncy.22278
Li Y, Tian X, Gao L, Jiang X, Fu R, Zhang T, Ren T, Hu P, Wu Y, Zhao P, Yang D (2019) Clinical significance of circulating tumor cells and tumor markers in the diagnosis of lung cancer. Cancer Med 8:3782–3792. https://doi.org/10.1002/cam4.2286
Liu WR, Zhang B, Chen C et al (2020) Detection of circulating genetically abnormal cells in peripheral blood for early diagnosis of non-small cell lung cancer. Thorac Cancer 11:3234–3242. https://doi.org/10.1111/1759-7714.13654
Lung Cancer Research Group of Respiratory Society of Chinese Medical Association and Expert Group of China Lung Cancer Prevention and Control Alliance, Chinese experts consensus on the diagnosis and treatment of pulmonary nodules (2018) Chinese J Tuberculosis Respir Dis 41(10)
McWilliams A, Tammemagi MC, Mayo JR et al (2013) Probability of cancer in pulmonary nodules detected on first screening CT. N Engl J Med 369:910–919. https://doi.org/10.1056/NEJMoa1214726
Mohan S, Chemi F, Brady G (2017) Challenges and unanswered questions for the next decade of circulating tumour cell research in lung cancer. Transl Lung Cancer Res 6:454–472. https://doi.org/10.21037/tlcr.2017.06.04
Nicholson AG, Chansky K, Crowley J, Beyruti R, Kubota K, Turrisi A, Eberhardt WE, van Meerbeeck J, Rami-Porta R (2016) The International association for the study of lung cancer lung cancer staging project: proposals for the revision of the clinical and pathologic staging of small cell lung cancer in the forthcoming eighth edition of the TNM classification for lung cancer. J Thorac Oncol 11:300–311
Poulet G, Massias J, Taly V (2019) Liquid biopsy: general concepts. Acta Cytol 63:449–455. https://doi.org/10.1159/000499337
Shin H, Oh S, Hong S et al (2020) Early-stage lung cancer diagnosis by deep learning-based spectroscopic analysis of circulating exosomes. ACS Nano 14:5435–5444. https://doi.org/10.1021/acsnano.9b09119
Sihong C, Jing Q, Xing J, Baiying L, Tianfu W, Dong N, Jie-Zhi C (2017) Automatic scoring of multiple semantic attributes with multi-task feature leverage: a study on pulmonary nodules in CT images. IEEE Trans Med Imaging 36:802–814. https://doi.org/10.1109/tmi.2016.2629462
Siravegna G, Marsoni S, Siena S, Bardelli A (2017) Integrating liquid biopsies into the management of cancer. Nat Rev Clin Oncol 14:531–548. https://doi.org/10.1038/nrclinonc.2017.14
Smith RA, Manassaram-Baptiste D, Brooks D, Cokkinides V, Doroshenk M, Saslow D, Wender RC, Brawley OW (2014) Cancer screening in the United States, 2014: a review of current American cancer society guidelines and current issues in cancer screening. CA Cancer J Clin 64:30–51. https://doi.org/10.3322/caac.21212
Tanoue LT, Tanner NT, Gould MK, Silvestri GA (2015) Lung cancer screening. Am J Respir Crit Care Med 191:19–33. https://doi.org/10.1164/rccm.201410-1777CI
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65:87–108. https://doi.org/10.3322/caac.21262
Travis WD, Brambilla E, Nicholson AG et al (2015) The 2015 world health organization classification of lung tumors: impact of genetic, clinical and radiologic advances since the 2004 classification. J Thorac Oncol 10:1243–1260. https://doi.org/10.1097/jto.0000000000000630
van Iersel CA, de Koning HJ, Draisma G, Mali WP, Scholten ET, Nackaerts K, Prokop M, Habbema JD, Oudkerk M, van Klaveren RJ (2007) Risk-based selection from the general population in a screening trial: selection criteria, recruitment and power for the Dutch-Belgian randomised lung cancer multi-slice CT screening trial (NELSON). Int J Cancer 120:868–874. https://doi.org/10.1002/ijc.22134
Wender R, Fontham ET, Barrera E Jr et al (2013) American cancer society lung cancer screening guidelines. CA Cancer J Clin 63:107–117. https://doi.org/10.3322/caac.21172
Woodard GA, Jones KD, Jablons DM (2016) Lung cancer staging and prognosis. Cancer Treat Res 170:47–75. https://doi.org/10.1007/978-3-319-40389-2_3
Yang Q, Zhang P, Wu R, Lu K, Zhou H (2018) Identifying the best marker combination in CEA, CA125, CY211, NSE, and SCC for lung cancer screening by combining ROC curve and logistic regression analyses: is it feasible? Dis Markers 2018:2082840. https://doi.org/10.1155/2018/2082840
Yousefi M, Ghaffari P, Nosrati R, Dehghani S, Salmaninejad A, Abarghan YJ, Ghaffari SH (2020) Prognostic and therapeutic significance of circulating tumor cells in patients with lung cancer. Cell Oncol (Dordr) 43:31–49. https://doi.org/10.1007/s13402-019-00470-y
Yu Z, Zhang G, Yang M, Zhang S, Zhao B, Shen G, Chai Y (2017) Systematic review of CYFRA 21–1 as a prognostic indicator and its predictive correlation with clinicopathological features in non-small cell lung cancer: a meta-analysis. Oncotarget 8:4043–4050. https://doi.org/10.18632/oncotarget.14022
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
There is no conflict of interest involved in this study.
Ethics approval
The study was approved by the ethics committee of Shandong First Medical University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Qiu, X., Zhang, H., Zhao, Y. et al. Application of circulating genetically abnormal cells in the diagnosis of early-stage lung cancer. J Cancer Res Clin Oncol 148, 685–695 (2022). https://doi.org/10.1007/s00432-021-03648-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-021-03648-w