Abstract
Purpose
To evaluate the efficacy of 177Lu-DOTA0-Tyr3-octreotate (177Lu-DOTATATE) radionuclide therapy in patients with inoperable or metastatic neuroendocrine tumours (NETs), (PROSPERO ID CRD42019130755).
Methods
All published clinical studies of NETs treated with 177Lu-DOTATATE were identified based on systematic searches in the PubMed, EMBASE, Cochrane Library, Web of Science and ClinicalTrials.gov databases up to January 2019. Among these studies, only the reports evaluated with the “Response Evaluation Criteria in Solid Tumours (RECIST)” or “Southwest Oncology Group (SWOG)” criteria or both were included. We analysed the disease response rate (DRR) and disease control rate (DCR) of each group to evaluate the efficacy of 177Lu-DOTATATE.
Results
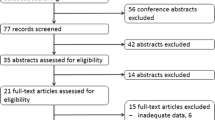
Fifteen studies were selected from 715 references. The pooled effect in the RECIST group (13 studies) was 27.58% (95% confidence interval (CI) 21.03–35.27%) for the DRR and 79.14% (95% CI 75.83–82.1%) for the DCR. In the SWOG criteria group (7 studies), the pooled effect was 20.59% (95% CI 10.89–35.51%) for the DRR and 78.28% (95% CI 74.39–81.72%) for the DCR. Therefore, the RECIST and SWOG groups showed similar DRRs and DCRs after177Lu-DOTATATE treatment, indicating that 177Lu-DOTATATE treatment has excellent efficacy with a control rate of approximately 78–79%. Moreover, adverse effects of 177Lu-DOTATATE were minimal, including fatigue, nausea, vomiting and hormonal disorders.
Conclusions
For patients with inoperable or metastatic NETs, 177Lu-DOTATATE is an effective treatment with minimal side effects.






Similar content being viewed by others
References
Albain KS, Crowley JJ, Turrisi AT, Gandara DR, Farrar WB, Clark JI, Beasley KR, Livingston RB (2002) Concurrent cisplatin, etoposide, and chest radiotherapy in pathologic stage IIIB non-small-cell lung cancer: a Southwest oncology group phase II study, SWOG 9019. J Clin Oncol 20:3454–3460. https://doi.org/10.1200/jco.2002.03.055
Bodei L, Cremonesi M, Grana CM, Fazio N, Iodice S, Baio SM, Bartolomei M, Lombardo D, Ferrari ME, Sansovini M, Chinol M, Paganelli G (2011) Peptide receptor radionuclide therapy with 177Lu-DOTATATE: the IEO phase I-II study. Eur J Nucl Med Mol Imaging 38:2125–2135. https://doi.org/10.1007/s00259-011-1902-1
Bodei L, Kidd M, Modlin IM, Severi S, Drozdov I, Nicolini S, Kwekkeboom DJ, Krenning EP, Baum RP, Paganelli G (2016) Measurement of circulating transcripts and gene cluster analysis predicts and defines therapeutic efficacy of peptide receptor radionuclide therapy (PRRT) in neuroendocrine tumors. Eur J Nucl Med Mol Imaging 43:839–851. https://doi.org/10.1007/s00259-015-3250-z
Dasari A, Shen C, Halperin D, Zhao B, Zhou S, Xu Y, Shih T, Yao JC (2017) Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States. JAMA Oncol 3:1335–1342. https://doi.org/10.1001/jamaoncol.2017.0589
Delpassand ES, Samarghandi A, Zamanian S et al (2014) Peptide receptor radionuclide therapy with 177Lu-DOTATATE for patients with somatostatin receptor-expressing neuroendocrine tumors: the first US phase 2 experience. Pancreas 43:518–525. https://doi.org/10.1097/mpa.0000000000000113
del Prete M, Buteau FA, Beauregard JM (2017) Personalized 177Lu-octreotate peptide receptor radionuclide therapy of neuroendocrine tumours: a simulation study. Eur J Nucl Med Mol Imaging 44:1490–1500. https://doi.org/10.1007/s00259-017-3688-2
del Prete M, Buteau FA, Arsenault F, Saighi N, Bouchard LO, Beaulieu A, Beauregard JM (2018) Personalized 177Lu-octreotate peptide receptor radionuclide therapy of neuroendocrine tumours: initial results from the P-PRRT trial. Eur J Nucl Med Mol Imaging 46:728–742. https://doi.org/10.1007/s00259-018-4209-7
Development Core Team R (2011) R: a language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria
Ezziddin S, Khalaf F, Vanezi M, Haslerud T, Mayer K, Zreiqat A, Willinek W, Biersack HJ, Sabet A (2014) Outcome of peptide receptor radionuclide therapy with 177Lu-octreotate in advanced grade 1/2 pancreatic neuroendocrine tumours. Eur J Nucl Med Mol Imaging 41:925–933. https://doi.org/10.1007/s00259-013-2677-3
Hallet J, Law CH, Cukier M, Saskin R, Liu N, Singh S (2015) Exploring the rising incidence of neuroendocrine tumors: a population-based analysis of epidemiology, metastatic presentation, and outcomes. Cancer 121:589–597. https://doi.org/10.1002/cncr.29099
Hamiditabar M, Ali M, Roys J, Wolin EM, O'Dorisio TM, Ranganathan D, Tworowska I, Strosberg JR, Delpassand ES (2017) Peptide receptor radionuclide therapy with 177Lu-octreotate in patients with somatostatin receptor expressing neuroendocrine tumors: six years' assessment. Clin Nucl Med 42:436–443. https://doi.org/10.1097/rlu.0000000000001629
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558. https://doi.org/10.1002/sim.1186
Hodi FS, Hwu WJ, Kefford R et al (2016) Evaluation of immune-related response criteria and RECIST v1.1 in patients with advanced melanoma treated with pembrolizumab. J Clin Oncol 34:1510–1517. https://doi.org/10.1200/jco.2015.64.0391
Kalshetty A, Ramaswamy A, Ostwal V, Basu S (2018) Resistant functioning and/or progressive symptomatic metastatic gastroenteropancreatic neuroendocrine tumors: efficacy of 177Lu-DOTATATE peptide receptor radionuclide therapy in this setting. Nucl Med Commun 39:1143–1149. https://doi.org/10.1097/mnm.0000000000000926
Kam BL, Teunissen JJ, Krenning EP, de Herder WW, Khan S, van Vliet EI, Kwekkeboom DJ (2012) Lutetium-labelled peptides for therapy of neuroendocrine tumours. Eur J Nucl Med Mol Imaging 39:S103–S112. https://doi.org/10.1007/s00259-011-2039-y
Kim SJ, Pak K, Koo PJ, Kwak JJ, Chang S (2015) The efficacy of 177Lu-labelled peptide receptor radionuclide therapy in patients with neuroendocrine tumours: a meta-analysis. Eur J Nucl Med Mol Imaging 42:1964–1970. https://doi.org/10.1007/s00259-015-3155-x
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol 62:e1–e34. https://doi.org/10.1016/j.jclinepi.2009.06.006
Mujica-Mota R, Varley-Campbell J, Tikhonova I et al (2018) Everolimus, lutetium-177 DOTATATE and sunitinib for advanced, unresectable or metastatic neuroendocrine tumours with disease progression: a systematic review and cost-effectiveness analysis. Health Technol Assess 22:1–326. https://doi.org/10.3310/hta22490
Oberg K, Jelic S, Esmo Guidelines Working Group (2009) Neuroendocrine gastroenteropancreatic tumors: ESMO clinical recommendation for diagnosis, treatment and follow-up. Ann Oncol 20(150):153. https://doi.org/10.1093/annonc/mdp158
Paganelli G, Sansovini M, Ambrosetti A, Severi S, Monti M, Scarpi E, Donati C, Ianniello A, Matteucci F, Amadori D (2014) 177 Lu-Dota-octreotate radionuclide therapy of advanced gastrointestinal neuroendocrine tumors: results from a phase II study. Eur J Nucl Med Mol Imaging 41:1845–1851. https://doi.org/10.1007/s00259-014-2735-5
Sabet A, Dautzenberg K, Haslerud T, Aouf A, Sabet A, Simon B, Mayer K, Biersack HJ, Ezziddin S (2015) Specific efficacy of peptide receptor radionuclide therapy with 177Lu-octreotate in advanced neuroendocrine tumours of the small intestine. Eur J Nucl Med Mol Imaging 42:1238–1246. https://doi.org/10.1007/s00259-015-3041-6
Sansovini M, Severi S, Ambrosetti A, Monti M, Nanni O, Sarnelli A, Bodei L, Garaboldi L, Bartolomei M, Paganelli G (2013) treatment with the radiolabelled somatostatin analog Lu-DOTATATE for advanced pancreatic neuroendocrine tumors. Neuroendocrinology 97:347–354. https://doi.org/10.1159/000348394
Schwarzer G (2012) Meta: meta‐analysis with R. R package version 2.0‐0. https://CRAN.R-project.org/package=meta. Accessed 23 June 2017
Severi S, Grassi I, Nicolini S, Sansovini M, Bongiovanni A, Paganelli G (2017) Peptide receptor radionuclide therapy in the management of gastrointestinal neuroendocrine tumors: efficacy profile, safety, and quality of life. Onco Targets Ther 10:551–557. https://doi.org/10.2147/OTT.S97584
Slim K, Nini E, Forestier D, Kwiatkowski F, Panis Y, Chipponi J (2003) Methodological index for non-randomized studies (minors): development and validation of a new instrument. ANZ J Surg 73:712–716. https://doi.org/10.1046/j.1445-2197.2003.02748.x
Soydal C, Peker A, Ozkan E, Kucuk ON, Kir MK (2016) The role of baseline Ga-68 DOTATATE positron emission tomography/computed tomography in the prediction of response to fixed-dose peptide receptor radionuclide therapy with Lu-177 DOTATATE. Turk J Med Sci 46:409–413. https://doi.org/10.3906/sag-1412-11
Strosberg J, El-Haddad G, Wolin E et al (2017) Phase 3 trial of 177Lu-DOTATATE for midgut neuroendocrine tumors. New Engl J Med 376:125–135. https://doi.org/10.1056/NEJMoa1607427
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, Moher D, Becker BJ, Sipe TA, Thacker SB (2000) Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis of observational studies in epidemiology (MOOSE) group. JAMA 283:2008–2012. https://doi.org/10.1001/jama.283.15.2008
Sward C, Bernhardt P, Ahlman H, Wangberg B, Forssell-Aronsson E, Larsson M, Svensson J, Rossi-Norrlund R, Kolby L (2010) [177Lu-DOTA 0-Tyr 3]-octreotate treatment in patients with disseminated gastroenteropancreatic neuroendocrine tumors: the value of measuring absorbed dose to the kidney. World J Surg 34:1368–1372. https://doi.org/10.1007/s00268-009-0387-6
Trotti A, Colevas AD, Setser A, Rusch V, Jaques D, Budach V, Langer C, Murphy B, Cumberlin R, Coleman CN, Rubin P (2003) CTCAE v3.0: development of a comprehensive grading system for the adverse effects of cancer treatment. Semin Radiat Oncol 13:176–181. https://doi.org/10.1016/s1053-4296(03)00031-6
Valkema R, Pauwels SA, Kvols LK, Kwekkeboom DJ, Jamar F, de Jong M, Barone R et al (2005) Long-term follow-up of renal function after peptide receptor radiation therapy with (90)Y-Dota(0),Tyr(3)-octreotide and (177)Lu-Dota(0), Tyr(3)-octreotate. J Nucl Med 46:83s–91s. https://jnm.snmjournals.org/content/46/1_suppl/83S.
Valkema R, Pauwels S, Kvols LK, Barone R, Jamar F, Bakker WH, Kwekkeboom DJ, Bouterfa H, Krenning EP (2006) Survival and response after peptide receptor radionuclide therapy with [90Y-DOTA0, Tyr3]octreotide in patients with advanced gastroenteropancreatic neuroendocrine tumors. Semin Nucl Med 36:147–156. https://doi.org/10.1053/j.semnuclmed.2006.01.001
van Binnebeek S, Vanbilloen B, Baete K et al (2016) Comparison of diagnostic accuracy of (111)in-pentetreotide SPECT and (68)Ga-DOTATOC PET/CT: a lesion-by-lesion analysis in patients with metastatic neuroendocrine tumours. Eur Radiol 26:900–909. https://doi.org/10.1007/s00330-015-3882-1
van Essen M, Krenning EP, Kam BL, de Herder WW, Feelders RA, Kwekkeboom DJ (2010) Salvage therapy with 177Lu-octreotate in patients with bronchial and gastroenteropancreatic neuroendocrine tumors. J Nucl Med 51:383–390. https://doi.org/10.2967/jnumed.109.068957
van Vliet EI, Krenning EP, Teunissen JJ, Bergsma H, Kam BL, Kwekkeboom DJ (2013) Comparison of response evaluation in patients with gastroenteropancreatic and thoracic neuroendocrine tumors after treatment with [177Lu-DOTA0, Tyr3]octreotate. J Nucl Med 54:1689–1696. https://doi.org/10.2967/jnumed.112.117408
Velikyan I, Xu H, Nair M, Hall H (2012) Robust labeling and comparative preclinical characterization of DOTA-TOC and DOTA-TATE. Nucl Med Biol 39:628–639. https://doi.org/10.1016/j.nucmedbio.2011.12.010
Werner RA, Bluemel C, Allen-Auerbach MS, Higuchi T, Herrmann K (2015) 68Gallium- and 90Yttrium-/ 177Lutetium: "theranostic twins" for diagnosis and treatment of NETs. Ann Nucl Med 29:1–7. https://doi.org/10.1007/s12149-014-0898-6
Funding
This study was funded by the Science and Technology Department of Sichuan Province (Grant Number 2018JPT0023).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study, formal consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, J., Song, Q., Cai, L. et al. The efficacy of 177Lu-DOTATATE peptide receptor radionuclide therapy (PRRT) in patients with metastatic neuroendocrine tumours: a systematic review and meta-analysis. J Cancer Res Clin Oncol 146, 1533–1543 (2020). https://doi.org/10.1007/s00432-020-03181-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03181-2