Abstract
Purpose
Telomere dysfunction is believed to be a significant factor in carcinogenesis. To elucidate the carcinogenesis mechanism in gastric cancer, the expression of telomeric proteins and changes in telomere length were investigated during multistage carcinogenesis of gastric cancer.
Methods
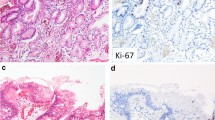
Tissue samples were obtained during surgical operations from the normal gastric mucosa of 10 patients, the precancerous lesions of 15 patients, the gastric cancer tissues (GC) of 20 patients, and of tumors due to gastric cancer with lymph node metastasis (GCLM) from 5 patients. The expression of TRF1, TRF2, and TIN2 proteins was measured by Western blotting, while the expression of TERT, KU70, and BRCA1 proteins was detected using the immunohistochemical method. The mean telomere length was determined by Southern blotting.
Results
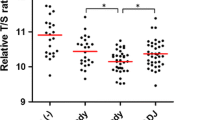
Compared with normal gastric mucosa tissues, the expression of TRF1, TRF2, and TIN2 proteins was significantly higher in precancerous lesions, GC, and GCLM (P < 0.01). The expression of TRF1, TRF2, and TIN2 proteins was significantly higher in GC and GCLM than in precancerous lesions (P < 0.01). The expression of TERT and Ku70 proteins in precancerous lesions and GC tissues was significantly higher than that in normal gastric mucosa tissues (P < 0.01). The expression of TERT and Ku70 proteins in GC tissues was significantly higher than in precancerous lesions (P < 0.01). In normal gastric mucosa, the BRCA1 protein was primarily located in the cell nucleus. In precancerous lesions and GC, the expression of the BRCA1 protein was apparent in the cell cytoplasm. The mean telomere length in precancerous lesions, GC, and GCLM was significantly shorter than that in normal gastric mucosa tissues (P < 0.05). The mean telomere length in GC and GCLM was significantly shorter than that in precancerous lesions (P < 0.05). The mean telomere length in all tissue samples was inversely correlated with the level of TRF1, TRF2, TIN2, TERT, and Ku70 proteins.
Conclusions
Our results suggest that the over-expression of telomeric proteins, TRF1, TRF2, TIN2, TERT, and Ku70, and the transposition of the BRCA1 protein may work together to reduce the telomere length in precancerous lesions and gastric cancer, and could contribute to the multistage carcinogenesis of gastric cancer. These findings offer new insight into the mechanism of carcinogenesis in gastric cancer.




Similar content being viewed by others
References
Autexier C, Lue NF (2006) The structure and function of telomerase reverse transcriptase. Annu Rev Biochem 75:493–517. doi:10.1146/annurev.biochem.75.103004.142412
Bellon M, Datta A, Brown M et al (2006) Increased expression of telomere length regulating factors TRF1, TRF2 and TIN2 in patients with adult T-cell leukemia. Int J Cancer 119(9):2090–2097. doi:10.1002/ijc.22026
Blackburn EH (1991) Structure and function of telomeres. Nature 350(6319):569–573. doi:10.1038/350569a0
Blasco MA (2005) Telomeres and human disease: ageing, cancer and beyond. Nat Rev Genet 6(8):611–622. doi:10.1038/nrg1656
Broccoli D, Smogorzewska A, Chong L et al (1997) Human telomeres contain two distinct Myb-related proteins, TRF1 and TRF2. Nat Genet 17(2):231–235. doi:10.1038/ng1097-231
Celli GB, Denchi EL, de Lange T et al (2006) Ku70 stimulates fusion of dysfunctional telomeres yet protects chromosome ends from homologous recombination. Nat Cell Biol 8(8):885–890. doi:10.1038/ncb1444
Chai W, Ford LP, Lenertz L et al (2002) Human Ku70/80 associates physically with telomerase through interaction with Htert. J Biol Chem 277(49):47242–47247. doi:10.1074/jbc.M208542200
Chen J, Rocken C, Malfertheiner P et al (2004) Recent advances in molecular diagnosis and therapy of gastric cancer. Dig Dis 22:380–385. doi:10.1159/000083602
Chen LY, Liu D, Songyang Z et al (2007) Telomere Maintenance through Spatial Control of Telomeric Proteins. Mol Cell Biol 27(16):5898–5909. doi:10.1128/MCB.00603-07
Cook BD, Dynek JN, Chang W et al (2002) Role for the related poly(ADP-ribose) polymerases tankyrase 1 and 2 at human telomeres. Mol Cell Biol 22:332–342. doi:10.1128/MCB.22.1.332-342.2002
Cornelis RS, Neuhausen SL, Johannson O et al (1995) High allele loss rates at 17q12–q21 in breast and ovarian tumors from BRCA1-linked families. the Breast Cancer Linkage Consortium. Genes Chromosom Cancer 13:203–210. doi:10.1002/gcc.2870130310
Dahse R, Fiedler W, Junker K et al (1999) Telomerase activity and telomere lengths: alterations in renal cell carcinomas. Kidney Int 56(4):1289–1290. doi:10.1046/j.1523-1755.1999.00688.x
de Lange T (2005) Shelterin: the protein complex that shapes and safeguards human telomeres. Genes Dev 19(18):2100–2110. doi:10.1101/gad.1346005
Garcia-Aranda C, de Juan C, Diaz-Lopez A et al (2006) Correlations of telomere length, telomerase activity, and telomeric-repeat binding factor 1 expression in colorectal carcinoma. Cancer 106(3):541–551. doi:10.1002/cncr.21625
Hahn WC (2003) Role of telomeres and telomerase in the pathogenesis of human cancer. J Clin Oncol 21(10):2034–2043. doi:10.1200/JCO.2003.06.018
Hsu HL, Gilley D, Blackburn EH et al (1999) Ku is associated with the telomere in mammals. Proc Natl Acad Sci USA 96(22):12454–12458. doi:10.1073/pnas.96.22.12454
Hu J, Sun L, Zhang C et al (2006) Expression of telomeric repeat binding factor 1 in non-small cell lung cancer. J Surg Oncol 93(1):62–67. doi:10.1002/jso.20421
Kim SH, Kaminker P, Campisi J et al (1999) TIN2, a new regulator of telomere length in human cells. Nat Genet 23(4):405–412. doi:10.1038/70508
Kim HR, Kim YJ, Kim HJ et al (2002) Telomere length changes in colorectal cancers and polyps. J Korean Med Sci 17(3):360–365
Li B, Oestreich S, de Lange T et al (2000) Identification of human Rap1: implications for telomere evolution. Cell 101:471–483. doi:10.1016/S0092-8674(00)80858-2
Li H, Lee TH, Avraham H et al (2002) A novel tricomplex of BRCA1, Nmi, and c-Myc inhibits c-Myc-induced human telomerase reverse transcriptase gene (hTERT) promoter activity in breast cancer. J Biol Chem 277(23):20965–20973. doi:10.1074/jbc.M112231200
Miyachi K, Fujita M, Tanaka N et al (2002) Correlation between telomerase activity and telomeric-repeat binding factors in gastric cancer. J Exp Clin Cancer Res 21(2):269–275
Neuhausen SL, Marshall CJ (1994) Loss of heterozygosity in familial tumors from three BRCA1-linked kindreds. Cancer Res 54:6069–6072
Oh BK, Kim YJ, Park C et al (2005) Up-regulation of telomere-binding proteins, TRF1, TRF2, and TIN2 is related to telomere shortening during human multistep hepatocarcinogenesis. Am J Pathol 166(1):73–80
Poncet D, Belleville A, t’kint de Roodenbeke C et al (2008) Changes in the expression of telomere maintenance genes suggest global telomere dysfunction in B-chronic lymphocytic leukemia. Blood 111(4):2388–2391. doi:10.1182/blood-2007-09-111245
Reddel RR, Bryan TM, Colgin LM et al (2001) Alternative lengthening of telomeres in human cells. Radiat Res 155(1 Pt 2):194–200. doi:10.1667/0033-7587(2001)155[0194:ALOTIH]2.0.CO;2
Smith S, de Lange T (1997) TRF1, a mammalian telomeric protein. Trends Genet 13(1):21–26. doi:10.1016/S0168-9525(96)10052-4
Smogorzewska A, van Steensel B, Bianchi A et al (2000) Control of human telomere length by TRF1 and TRF2. Mol Cell Biol 20(5):1659–1668. doi:10.1128/MCB.20.5.1659-1668.2000
Song K, Jung D, Jung Y et al (2000) Interaction of human Ku70 with TRF2. FEBS Lett 481(1):81–85. doi:10.1016/S0014-5793(00)01958-X
Stansel RM, de Lange T, Griffith JD (2001) T-loop assembly in vitro involves binding of TRF2 near the 3′ telomeric overhang. EMBO J 20:5532–5540. doi:10.1093/emboj/20.19.5532
van Steensel Band de Lange T (1997) Control of telomere length by the human telomeric protein TRF1. Nature 385(6618):740–743. doi:10.1038/385740a0
Wainwright LJ, Middleton PG, Rees JL et al (1995) Changes in mean telomere length in basal cell carcinomas of the skin. Genes Chromosom Cancer 12(1):45–49. doi:10.1002/gcc.2870120108
Xiong J, Fan S, Meng Q et al (2003) BRCA1 inhibition of telomerase activity in cultured cells. Mol Cell Biol 23(23):8668–8690. doi:10.1128/MCB.23.23.8668-8690.2003
Yamada M, Tsuji N, Nakamura M et al (2002) Down-regulation of TRF1, TRF2 and TIN2 genes is important to maintain telomeric DNA for gastric cancers. Anticancer Res 22(6A):3303–3307
Ye JZ, Donigian JR, van Overbeek M et al (2004) TIN2 binds TRF1 and TRF2 simultaneously and stabilizes the TRF2 complex on telomeres. J Biol Chem 279(45):47264–47271. doi:10.1074/jbc.M409047200
Zhou XZ, Lu KP (2001) The Pin2/TRF1 interacting protein PinX1 is a potent telomerase inhibitor. Cell 107:347–359. doi:10.1016/S0092-8674(01)00538-4
Zhu XD, Kuster B, Mann M et al (2000) Cellcycle-regulated association of Rad50/MRE11/NBS1 with TRF2 and human telomeres. Nat Genet 25:347–352. doi:10.1038/77139
Acknowledgments
This work was supported by the construct program of the key discipline in Hunan Province.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hu, H., Zhang, Y., Zou, M. et al. Expression of TRF1, TRF2, TIN2, TERT, KU70, and BRCA1 proteins is associated with telomere shortening and may contribute to multistage carcinogenesis of gastric cancer. J Cancer Res Clin Oncol 136, 1407–1414 (2010). https://doi.org/10.1007/s00432-010-0795-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-010-0795-x