Abstract
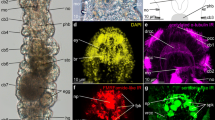
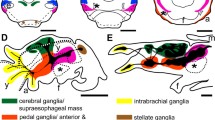
Coleoid cephalopods have a high intelligence, complex structures, and large brain. The cephalopod brain is divided into supraesophageal mass, subesophageal mass and optic lobe. Although much is known about the structural organization and connections of various lobes of octopus brain, there are few studies on the brain of cephalopod at the molecular level. In this study, we demonstrated the structure of an adult Octopus minor brain by histomorphological analyses. Through visualization of neuronal and proliferation markers, we found that adult neurogenesis occurred in the vL and posterior svL. We also obtained specific 1015 genes by transcriptome of O. minor brain and selected OLFM3, NPY, GnRH, and GDF8 genes. The expression of genes in the central brain showed the possibility of using NPY and GDF8 as molecular marker of compartmentation in the central brain. This study will provide useful information for establishing a molecular atlas of cephalopod brain.




Similar content being viewed by others
Data availability
The RNA-seq raw data used in this study have been deposited in the Zenodo database under accession code https://doi.org/10.5281/zenodo.7701881. Related sequencing data have been uploaded to NCBI’s GenBank (BTG1, OP751951; SerT, OP751952; OLFM3, OP751948; NPY, OP751944; GnRH, OP751945; GDF8, OP751949). Other data supporting the findings of the present study are available from the corresponding author upon reasonable request.
References
Bahl A, Engert F (2020) Neural circuits for evidence accumulation and decision making in larval zebrafish. Nat Neurosci 23(1):94–102. https://doi.org/10.1038/s41593-019-0534-9
Bertapelle C, Polese G, Di Cosmo A (2017) Enriched environment increases PCNA and PARP1 levels in Octopus vulgaris central nervous system: first evidence of adult neurogenesis in Lophotrochozoa. J Exp Zool B Mol Dev Evol 328(4):347–359. https://doi.org/10.1002/jez.b.22735
Bogoraze D, Cazal P (1946) Remarques sur le système stomatogastrique du Poulpe (Octopus vulgaris Lamarck): le complexe retro-buccal. Arch Zool Exp Gén 84:115–131
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30(15):2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Boycott BB, Young JZ (1955) A memory system in Octopus vulgaris Lamarck. Proc R Soc Lond Ser B-Biol Sci 143(913):449–480. https://doi.org/10.1098/rspb.1955.0024
Boyle P (1991) The UFAW handbook on the care and management of cephalopods in the laboratory. Universities Federation for Animal Welfare
Burgus R, Butcher M, Amoss M, Ling N, Monahan M, Rivier J, Fellows R, Blackwell R, Vale W, Guillemin R (1972) Primary structure of the ovine hypothalamic luteinizing hormone-releasing factor (LRF)(LH-hypothalamus-LRF-gas chromatography-mass spectrometry-decapeptide-Edman degradation). Proc Natl Acad Sci 69(1):278–282. https://doi.org/10.1073/pnas.69.1.278
Butler AB (2000) Chordate evolution and the origin of craniates: an old brain in a new head. Anatomical Record 261(3):111–125. https://doi.org/10.1002/1097-0185(20000615)261:3%3c111::AID-AR6%3e3.0.CO;2-F
Christie BR, Cameron HA (2006) Neurogenesis in the adult hippocampus. Hippocampus 16(3):199–207. https://doi.org/10.1002/hipo.20151
Di Cosmo A, Bertapelle C, Porcellini A, Polese G (2018) Magnitude assessment of adult neurogenesis in the Octopus vulgaris brain using a flow cytometry-based technique. Front Physiol. https://doi.org/10.3389/fphys.2019.00076
Dickel L, Chichery MP, Chichery R (2001) Increase of learning abilities and maturation of the vertical lobe complex during postembryonic development in the cuttlefish, Sepia. Develop Psychobiol 39(2):92–98. https://doi.org/10.1002/dev.1033
Erö C, Gewaltig M-O, Keller D, Markram H (2018) A cell atlas for the mouse brain. Front Neuroinform 12:84. https://doi.org/10.3389/fninf.2018.00084
Farioli-Vecchioli S, Micheli L, Saraulli D, Ceccarelli M, Cannas S, Scardigli R, Leonardi L, Cinà I, Costanzi M, Ciotti MT (2012) Btg1 is required to maintain the pool of stem and progenitor cells of the dentate gyrus and subventricular zone. Front Neurosci 6:124. https://doi.org/10.3389/fnins.2012.00124
Ferreira FF, Ribeiro FF, Rodrigues RS, Sebastião AM, Xapelli S (2018) Brain-derived neurotrophic factor (BDNF) role in cannabinoid-mediated neurogenesis. Front Cellular Neurosci. https://doi.org/10.3389/fncel.2018.00441
Fiorito G, Scotto P (1992) Observational learning in Octopus vulgaris. Science 256(5056):545–547. https://doi.org/10.1126/science.256.5056.545
Gandal MJ, Zhang P, Hadjimichael E, Walker RL, Chen C, Liu S, Won H, Van Bakel H, Varghese M, Wang Y (2018) Transcriptome-wide isoform-level dysregulation in ASD, schizophrenia, and bipolar disorder. Science 362(6420):eaat8127. https://doi.org/10.1126/science.aat8127
Goff L, Trapnell C, Kelley D Cummerbund: analysis, exploration, manipulation and visualization of Cufflinks high-throughput sequencing data. 2012 R package versoin 2.8. 2.
Hansen JY, Shafiei G, Markello RD, Smart K, Cox SM, Wu Y, Gallezot J-D, Aumont É, Servaes S, Scala SG (2021) Mapping neurotransmitter systems to the structural and functional organization of the human neocortex. Biorxiv. https://doi.org/10.1101/2021.10.28.466336
Hochner B, Shomrat T, Fiorito G (2006) The octopus: a model for a comparative analysis of the evolution of learning and memory mechanisms. Biol Bull 210(3):308–317. https://doi.org/10.2307/4134567
Iwakoshi-Ukena E, Ukena K, Takuwa-Kuroda K, Kanda A, Tsutsui K, Minakata H (2004) Expression and distribution of octopus gonadotropin-releasing hormone in the central nervous system and peripheral organs of the octopus (Octopus vulgaris) by in situ hybridization and immunohistochemistry. J Comparat Neurol 477(3):310–323. https://doi.org/10.1002/cne.20260
Jaremko WJ, Huang Z, Wen W, Wu A, Karl N, Niu L (2017) One aptamer, two functions: the full-length aptamer inhibits AMPA receptors, while the short one inhibits both AMPA and kainate receptors. RNA Dis (Houston, Tex) 4(2):e1560
Jereb P, Roper C, Norman M, Finn J (2014) Cephalopods of the world. An annotated and illustrated catalogue of cephalopod species known to date. Volume 3. Octopods and Vampire Squids. FAO species catalogue for fishery purposes 4(3):370
Jung S-H, Song HY, Hyun YS, Kim Y-C, Whang I, Choi T-Y, Jo S (2018) A brain atlas of the long arm octopus, octopus minor. Exp Neurobiol 27(4):257. https://doi.org/10.5607/en.2018.27.4.257
Kanda A, Satake H, Kawada T, Minakata H (2005) Novel evolutionary lineages of the invertebrate oxytocin/vasopressin superfamily peptides and their receptors in the common octopus (Octopus vulgaris). Biochem J 387(1):85–91. https://doi.org/10.1042/BJ20041230
Kanda A, Takahashi T, Satake H, Minakata H (2006) Molecular and functional characterization of a novel gonadotropin-releasing-hormone receptor isolated from the common octopus (Octopus vulgaris). Biochem J 395(1):125–135. https://doi.org/10.1042/BJ20051615
Kee N, Sivalingam S, Boonstra R, Wojtowicz J (2002) The utility of Ki-67 and BrdU as proliferative markers of adult neurogenesis. J Neurosci Methods 115(1):97–105. https://doi.org/10.1016/S0165-0270(02)00007-9
Kenning M, Harzsch S (2013) Brain anatomy of the marine isopod Saduria entomon Linnaeus, 1758 (Valvifera, Isopoda) with special emphasis on the olfactory pathway. Front Neuroanat 7:32. https://doi.org/10.3389/fnana.2013.00032
Kim B-M, Kang S, Ahn D-H, Jung S-H, Rhee H, Yoo JS, Lee J-E, Lee S, Han Y-H, Ryu K-B (2018) The genome of common long-arm octopus Octopus minor. Gigascience 7(11):giy119. https://doi.org/10.1093/gigascience/giy119
Klempin F, Beis D, Mosienko V, Kempermann G, Bader M, Alenina N (2013) Serotonin is required for exercise-induced adult hippocampal neurogenesis. J Neurosci 33(19):8270–8275. https://doi.org/10.1523/JNEUROSCI.5855-12.2013
Matsuo H, Baba Y, Nair RG, Arimura A, Schally A (1971) Structure of the porcine LH-and FSH-releasing hormone. I. The proposed amino acid sequence. Biochem Biophys Res Commun 43(6):1334–1339. https://doi.org/10.1016/S0006-291X(71)80019-0
Miyata S (2017) Advances in understanding of structural reorganization in the hypothalamic neurosecretory system. Front Endocrinol 8:275. https://doi.org/10.3389/fendo.2017.00275
Murphy D, Konopacka A, Hindmarch C, Paton JF, Sweedler JV, Gillette MU, Ueta Y, Grinevich V, Lozic M, Japundzic-Zigon N (2012) The hypothalamic-neurohypophyseal system: from genome to physiology. J Neuroendocrinol 24(4):539–553. https://doi.org/10.1111/j.1365-2826.2011.02241.x
Nakaya N, Sultana A, Munashinghe J, Cheng A, Mattson MP, Tomarev SI (2013) Deletion in the N-terminal half of olfactomedin 1 modifies its interaction with synaptic proteins and causes brain dystrophy and abnormal behavior in mice. Exp Neurol 250:205–218. https://doi.org/10.1016/j.expneurol.2013.09.019
Nakazawa M, Cebrià F, Mineta K, Ikeo K, Agata K, Gojobori T (2003) Search for the evolutionary origin of a brain: planarian brain characterized by microarray. Mol Biol Evol 20(5):784–791. https://doi.org/10.1093/molbev/msg086
Newth D (1972) The anatomy of the nervous system of Octopus vulgaris. J Neurol Neurosurg Psychiatry 35(3):421
Nixon M, Young JZ, Young JZ (2003) The brains and lives of cephalopods. Oxford University Press, Oxford
Ortiz C, Navarro JF, Jurek A, Märtin A, Lundeberg J, Meletis K (2020) Molecular atlas of the adult mouse brain. Sci Adv 6(26):eabb3446. https://doi.org/10.1126/sciadv.abb3446
Pertea M, Kim D, Pertea GM, Leek JT, Salzberg SL (2016) Transcript-level expression analysis of RNA-seq experiments with HISAT, StringTie and Ballgown. Nat Protocols 11(9):1650–1667. https://doi.org/10.1038/nprot.2016.095
Raffo-Romero A, Arab T, Van Camp C, Lemaire Q, Wisztorski M, Franck J, Aboulouard S, Marrec-Croq L, Sautiere P-E, Vizioli J (2019) ALK4/5-dependent TGF-β signaling contributes to the crosstalk between neurons and microglia following axonal lesion. Sci Rep 9(1):1–17. https://doi.org/10.1038/s41598-019-43328-x
Roth G (2015) Convergent evolution of complex brains and high intelligence. Philos Transact R Soc b 370(1684):20150049. https://doi.org/10.1098/rstb.2015.0049
Sakaue Y, Bellier J-P, Kimura S, D’Este L, Takeuchi Y, Kimura H (2014) Immunohistochemical localization of two types of choline acetyltransferase in neurons and sensory cells of the octopus arm. Brain Struct Funct 219:323–341. https://doi.org/10.1007/s00429-012-0502-6
Schmidt H, Gour A, Straehle J, Boergens KM, Brecht M, Helmstaedter M (2017) Axonal synapse sorting in medial entorhinal cortex. Nature 549(7673):469–475. https://doi.org/10.1038/nature24005
Schnell AK, Amodio P, Boeckle M, Clayton NS (2021) How intelligent is a cephalopod? Lessons from comparative cognition. Biol Rev 96(1):162–178. https://doi.org/10.1111/brv.12651
Schwenk J, Harmel N, Brechet A, Zolles G, Berkefeld H, Müller CS, Bildl W, Baehrens D, Hüber B, Kulik A (2012) High-resolution proteomics unravel architecture and molecular diversity of native AMPA receptor complexes. Neuron 74(4):621–633. https://doi.org/10.1016/j.neuron.2012.03.034
Segi-Nishida E (2017) The effect of serotonin-targeting antidepressants on neurogenesis and neuronal maturation of the hippocampus mediated via 5-HT1A and 5-HT4 receptors. Front Cell Neurosci 11:142. https://doi.org/10.3389/fncel.2017.00142
Shanks NF, Savas JN, Maruo T, Cais O, Hirao A, Oe S, Ghosh A, Noda Y, Greger IH, Yates Iii JR (2012) Differences in AMPA and kainate receptor interactomes facilitate identification of AMPA receptor auxiliary subunit GSG1L. Cell Rep 1(6):590–598. https://doi.org/10.1016/j.celrep.2012.05.004
Shao Y, Wang X-B, Zhang J-J, Li M-L, Wu S-S, Ma X-Y, Wang X, Zhao H-F, Li Y, Zhu HH (2020) Genome and single-cell RNA-sequencing of the earthworm Eisenia andrei identifies cellular mechanisms underlying regeneration. Nat Commun 11(1):1–15. https://doi.org/10.1038/s41467-020-16454-8
Shigeno S, Ragsdale CW (2015) The gyri of the octopus vertical lobe have distinct neurochemical identities. J Comparat Neurol 523(9):1297–1317. https://doi.org/10.1002/cne.23755
Shigeno S, Yamamoto M (2005) Embryonic brain development of Loliginids: axonal scaffold and neuropil formation related to early life styles. Phuket Mar Biol Cent Res Bull 66:155–165
Shigeno S, Kidokoro H, Tsuchiya K, Segawa S, Yamamoto M (2001) Development of the brain in the oegopsid squid, Todarodes pacificus: an atlas from hatchling to juvenile. Zoolog Sci 18(8):1081–1096. https://doi.org/10.2108/zsj.18.1081
Shigeno S, Andrews PL, Ponte G, Fiorito G (2018) Cephalopod brains: an overview of current knowledge to facilitate comparison with vertebrates. Front Physiol. https://doi.org/10.3389/fphys.2018.00952
Somoza GM, Miranda LA, Strobl-Mazzulla P, Guilgur LG (2002) Gonadotropin-releasing hormone (GnRH): from fish to mammalian brains. Cell Mol Neurobiol 22(5):589–609. https://doi.org/10.1023/a:1021888420271
Strausfeld NJ (2009) Brain organization and the origin of insects: an assessment. Proc R Soc b 276(1664):1929–1937. https://doi.org/10.1098/rspb.2008.1471
Strugnell J, Nishiguchi MK (2007) Molecular phylogeny of coleoid cephalopods (Mollusca: Cephalopoda) inferred from three mitochondrial and six nuclear loci: a comparison of alignment, implied alignment and analysis methods. J Mollusc Studies 73(4):399–410. https://doi.org/10.1093/mollus/eym038
Su C, Huang DP, Liu JW, Liu WY, Cao YO (2019) miR-27a-3p regulates proliferation and apoptosis of colon cancer cells by potentially targeting BTG1. Oncol Lett 18(3):2825–2834. https://doi.org/10.3892/ol.2019.10629
Sultana A, Nakaya N, Dong L, Abu-Asab M, Qian H, Tomarev SI (2014) Deletion of olfactomedin 2 induces changes in the AMPA receptor complex and impairs visual, olfactory, and motor functions in mice. Exp Neurol 261:802–811. https://doi.org/10.1016/j.expneurol.2014.09.002
Swanson L (2000) A history of neuroanatomical mapping. In: Toga AW, Mazziota JC (eds) Brain mapping: the systems. Elsevier. pp 77–109. https://doi.org/10.1016/B978-012692545-6/50005-2
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, Van Baren MJ, Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28(5):511–515. https://doi.org/10.1038/nbt.1621
Trapnell C, Hendrickson DG, Sauvageau M, Goff L, Rinn JL, Pachter L (2013) Differential analysis of gene regulation at transcript resolution with RNA-seq. Nat Biotechnol 31(1):46–53. https://doi.org/10.1038/nbt.2450
Trejo JL, Carro E, Torres-Aleman I (2001) Circulating insulin-like growth factor I mediates exercise-induced increases in the number of new neurons in the adult hippocampus. J Neurosci 21(5):1628–1634. https://doi.org/10.1523/JNEUROSCI.21-05-01628.2001
Uribe JE, Zardoya R (2017) Revisiting the phylogeny of Cephalopoda using complete mitochondrial genomes. J Mollusc Studies 83(2):133–144. https://doi.org/10.1093/mollus/eyw052
Wells M, Wells J (1969) Pituitary analogue in the octopus. Nature 222(5190):293–294. https://doi.org/10.1038/222293a0
Widagdo J, Guntupalli S, Jang SE, Anggono V (2017) Regulation of AMPA receptor trafficking by protein ubiquitination. Front Mol Neurosci 10:347. https://doi.org/10.3389/fnmol.2017.00347
Wild E, Wollesen T, Haszprunar G, Heß M (2015) Comparative 3D microanatomy and histology of the eyes and central nervous systems in coleoid cephalopod hatchlings. Org Divers Evol 15(1):37–64. https://doi.org/10.1007/s13127-014-0184-4
Winters GC, Polese G, Di Cosmo A, Moroz LL (2020) Mapping of neuropeptide Y expression in Octopus brains. J Morphol 281(7):790–801. https://doi.org/10.1002/jmor.21141
Young J (1963) The number and sizes of nerve cells in Octopus. In: Proceedings of the Zoological Society of London,. vol 2. Wiley Online Library, pp 229–254. https://doi.org/10.1111/j.1469-7998.1963.tb01862.x
Young JZ (1970) Neurovenous tissues in cephalopods. Philos Transact R Soc Lond b 257(815):309–321. https://doi.org/10.1098/rstb.1970.0027
Young J (1991) Computation in the learning system of cephalopods. Biol Bull 180(2):200–208. https://doi.org/10.2307/1542389
Zeng H (2018) Mesoscale connectomics. Curr Opin Neurobiol 50:154–162. https://doi.org/10.1016/j.conb.2018.03.003
Zeng L-C, Han Z-G, Ma W-J (2005) Elucidation of subfamily segregation and intramolecular coevolution of the olfactomedin-like proteins by comprehensive phylogenetic analysis and gene expression pattern assessment. FEBS Lett 579(25):5443–5453. https://doi.org/10.1016/j.febslet.2005.08.064
Zhao C, Deng W, Gage FH (2008) Mechanisms and functional implications of adult neurogenesis. Cell 132(4):645–660. https://doi.org/10.1016/j.cell.2008.01.033
Zilles K, Amunts K (2010) Centenary of Brodmann’s map—conception and fate. Nat Rev Neurosci 11(2):139–145. https://doi.org/10.1038/nrn2776
Funding
This work was supported by the research grant of the Chungbuk National University in 2021.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. C-JL, H-YL, Y-CG, SS, and S-JC contributed to experiments, material preparation, data collection, and analysis. Y-SY DEG contributed to analysis. HL and KK contributed to western blotting. C-JL, K-BR contributed to experiments and prepared the figures. First draft of the manuscript was written by C-JL, H-YL, Y-CG, SS and S-JC, S-JC supervised the project. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial and non-financial interests to disclose.
Ethical approval
Based on ‘The regulation of IACUC in CBNU (Chungbuk national University)’ and ‘The related laws (animal welfare act, laboratory animal act)’, the IACUC in CBNU require an animal use protocol for experiments involving vertebrate animals. However, it does not require animal use protocol for experiments using invertebrate species (e.g., brine shrimp, fruit flies, nematodes, Cephalopoda, etc.). Therefore, the review of the animal use protocol by IACUC is not mandatory for this research. We have uploaded a specific waiver letter-stamped by the ethics committee (CBNU) as Supplementary Material.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, CJ., Lee, HY., Yu, YS. et al. Brain compartmentalization based on transcriptome analyses and its gene expression in Octopus minor. Brain Struct Funct 228, 1283–1294 (2023). https://doi.org/10.1007/s00429-023-02647-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-023-02647-6