Abstract
Main conclusion
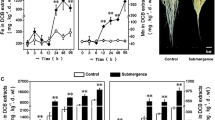
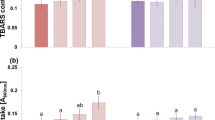
In this study, we report that peroxynitrite is necessary for ethylene-mediated aerenchyma formation in rice roots under waterlogging conditions.
Abstract
Plants under waterlogging stress face anoxygenic conditions which reduce their metabolism and induce several adaptations. The formation of aerenchyma is of paramount importance for the survival of plants under waterlogging conditions. Though some studies have shown the involvement of ethylene in aerenchyma formation under waterlogging conditions, the implication of peroxynitrite (ONOO−) in such a developmental process remains elusive. Here, we report an increase in aerenchyma formation in rice roots exposed to waterlogging conditions under which the number of aerenchyma cells and their size was further enhanced in response to exogenous ethephon (a donor of ethylene) or SNP (a donor of nitric oxide) treatment. Application of epicatechin (a peroxynitrite scavenger) to waterlogged plants inhibited the aerenchyma formation, signifying that ONOO− might have a role in aerenchyma formation. Interestingly, epicatechin and ethephon co-treated waterlogged plants were unable to form aerenchyma, indicating the necessity of ONOO− in ethylene-mediated aerenchyma formation under waterlogging conditions. Taken together, our results highlight the role of ONOO− in ethylene-mediated aerenchyma formation in rice and could be used in the future to develop waterlogging stress-tolerant varieties of rice.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
Abbreviations
- Epi:
-
Epicatechin
- Et:
-
Ethephon
- Fv/Fm:
-
Maximum photochemical efficiency of photosystem II
- GSNOR:
-
S-Nitrosoglutathione reductase
- NO:
-
Nitric oxide
- NOS-like activity:
-
Nitric oxide synthase-like activity
- ONOO− :
-
Peroxynitrite
- GSNO:
-
S-Nitrosoglutathione
- SNP:
-
Sodium nitroprusside
- WL:
-
Waterlogging conditions
References
Ahammed GJ, Li X (2022) Elevated carbon dioxide-induced regulation of ethylene in plants. Environ Exp Bot 202:105025
Ahammed GJ, Wu Y, Wang Y, Guo T, Shamsy R, Li X (2023) Epigallocatechin-3-Gallate (EGCG): a unique secondary metabolite with diverse roles in plant-environment interaction. Environ Exp Bot 209:105299
Alamillo JM, García-Olmedo F (2001) Effects of urate, a natural inhibitor of peroxynitrite-mediated toxicity, in the response of Arabidopsis thaliana to the bacterial pathogen Pseudomonas syringae. Plant J 25(5):529–540
Alpuerto JB, Hussain RMF, Fukao T (2016) The key regulator of submergence tolerance, SUB1A, promotes photosynthetic and metabolic recovery from submergence damage in rice leaves. Plant Cell Environ 39(3):672–684
Alvarez B, Radi R (2003) Peroxynitrite reactivity with amino acids and proteins. Amino Acids 25:295–311
Bailey-Serres J, Fukao T, Gibbs DJ, Holdsworth MJ, Lee SC, Licausi F, Perata P, Voesenek LA, van Dongen JT (2012a) Making sense of low oxygen sensing. Trends Plant Sci 17(3):129–138
Bailey-Serres J, Lee SC, Brinton E (2012b) Waterproofing crops: effective flooding survival strategies. Plant Physiol 160(4):1698–1709
Baxter-Burrell A, Yang Z, Springer PS, Bailey-Serres J (2002) RopGAP4-dependent Rop GTPase rheostat control of Arabidopsis oxygen deprivation tolerance. Science 296(5575):2026–2028
Ding J, Liang P, Wu P, Zhu M, Li C, Zhu X, Gao D, Chen Y, Guo W (2020) Effects of waterlogged on grain yield and associated traits of historic wheat cultivars in the middle and lower reaches of the Yangtze River. China Field Crops Res 246:107695
Ferrer-Sueta G, Radi R (2009) Chemical biology of peroxynitrite: kinetics, diffusion, and radicals. ACS Chem Biol 4(3):161–177
Frungillo L, De Oliveira JFP, Saviani EE, Oliveira HC, Martínez MC, Salgado I (2013) Modulation of mitochondrial activity by S-nitrosoglutathione reductase in Arabidopsis thaliana transgenic cell lines. Biochim Biophys Acta-Bioenerg 1827(3):239–247
Gaupels F, Spiazzi-Vandelle E, Yang D, Delledonne M (2011) Detection of peroxynitrite accumulation in Arabidopsis thaliana during the hypersensitive defense response. Nitric Oxide 25(2):222–228
Horchani F, Aschi-Smiti S, Brouquisse R (2010) Involvement of nitrate reduction in the tolerance of tomato (Solanum lycopersicum L.) plants to prolonged root hypoxia. Acta Physiol Plant 32(6):1113–1123
Kuai J, Liu Z, Wang Y, Meng Y, Chen B, Zhao W, Zhou Z, Oosterhuis DM (2014) Waterlogging during flowering and boll forming stages affects sucrose metabolism in the leaves subtending the cotton ball and its relationship with boll weight. Plant Sci 223:79–98
Kuroha T, Nagai K, Gamuyao R, Wang DR, Furuta T, Nakamori M, Kitaoka T, Adachi K, Minami A, Mori Y, Mashiguchi K (2018) Ethylene-gibberellin signaling underlies adaptation of rice to periodic flooding. Science 361(6398):181–186
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382
Mignolli F, Barone JO, Vidoz ML (2021) Root submergence enhances respiration and sugar accumulation in the stem of flooded tomato plants. Plant Cell Environ 44(11):3643–3654
Mondal D (2020) Floodplain alteration of the Bagmari–Bansloi–Pagla river system. In: Das BC, Ghosh S, Islam A, Roy S (eds) Anthropogeomorphology of bhagirathi-hooghly river system in India. CRC Press, pp 123–133
Oliveira HC, Salgado I, Sodek L (2013) Involvement of nitrite in the nitrate-mediated modulation of fermentative metabolism and nitric oxide production of soybean roots during hypoxia. Planta 237:255–264
Pan J, Sharif R, Xu X, Chen X (2021) Mechanisms of waterlogging tolerance in plants: research progress and prospects. Front Plant Sci 11:627331
Qi X, Li Q, Ma X, Qian C, Wang H, Ren N, Shen C, Huang S, Xu X, Xu Q, Chen X (2019) Waterlogging-induced adventitious root formation in cucumber is regulated by ethylene and auxin through reactive oxygen species signalling. Plant Cell Environ 42(5):1458–1470
Rauf M, Arif M, Fisahn J, Xue GP, Balazadeh S, Mueller-Roeber B (2013) NAC transcription factor speedy hyponastic growth regulates flooding-induced leaf movement in Arabidopsis. Plant Cell 25(12):4941–4955
Rodríguez-Ruiz M, Mioto P, Palma JM, Corpas FJ (2017) S-nitrosoglutathione reductase (GSNOR) activity is down-regulated during pepper (Capsicum annuum L.) fruit ripening. Nitric Oxide 68:51–55
Saito S, Yamamoto-Katou A, Yoshioka H, Doke N, Kawakita K (2006) Peroxynitrite generation and tyrosine nitration in defense responses in tobacco BY-2 cells. Plant Cell Physiol 47(6):689–697
Sakamoto A, Ueda M, Morikawa H (2002) Arabidopsis glutathione-dependent formaldehyde dehydrogenase is an S-nitrosoglutathione reductase. FEBS Lett 515(1–3):20–24
Singh S, Husain T, Kushwaha BK, Suhel M, Fatima A, Mishra V, Singh SK, Bhatt JA, Rai M, Prasad SM, Dubey NK et al (2021) Regulation of ascorbate-glutathione cycle by exogenous nitric oxide and hydrogen peroxide in soybean roots under arsenate stress. J Hazard Mater 409:123686
Song J, Shi G, Gao B, Fan H, Wang B (2011) Waterlogging and salinity effects on two Suaeda salsa populations. Physiol Plant 141(4):343–351
Sun C, Liu L, Lu L, Jin C, Lin X (2018) Nitric oxide acts downstream of hydrogen peroxide in regulating aluminum-induced antioxidant defense that enhances aluminum resistance in wheat seedlings. Environ Exp Bot 145:95–103
Timilsina A, Dong W, Hasanuzzaman M, Liu B, Hu C (2022) Nitrate–nitrite–nitric oxide pathway: a mechanism of hypoxia and anoxia tolerance in plants. Int J Mol Sci 23(19):11522
Wany A, Gupta KJ (2018) Reactive oxygen species, nitric oxide production and antioxidant gene expression during development of aerenchyma formation in wheat. Plant Signal Behav 13(2):3002–3017
Wany A, Kumari A, Gupta KJ (2017) Nitric oxide is essential for the development of aerenchyma in wheat roots under hypoxic stress. Plant Cell Environ 40(12):3002–3017
Xie Y, Mao Y, Lai D, Zhang W, Zheng T, Shen W (2013) Roles of NIA/NR/NOA1-dependent nitric oxide production and HY1 expression in the modulation of Arabidopsis salt tolerance. J Exp Bot 64(10):3045–3060
Xuewen X, Huihui W, Xiaohua Q, Qiang X, Xuehao C (2014) Waterlogging-induced increase in fermentation and related gene expression in the root of cucumber (Cucumis sativus L.). Sci Hortic 179:388–395
Yamauchi T, Shimamura S, Nakazono M, Mochizuki T (2013) Aerenchyma formation in crop species: a review. Field Crops Res 152:8–16
Yamauchi T, Watanabe K, Fukazawa A, Mori H, Abe F, Kawaguchi K, Oyanagi A, Nakazono M (2014) Ethylene and reactive oxygen species are involved in root aerenchyma formation and adaptation of wheat seedlings to oxygen-deficient conditions. J Exp Bot 65(1):261–273
Yamauchi T, Yoshioka M, Fukazawa A, Mori H, Nishizawa NK, Tsutsumi N, Yoshioka H, Nakazono M (2017) An NADPH oxidase RBOH functions in rice roots during lysigenous aerenchyma formation under oxygen-deficient conditions. Plant Cell 29(4):775–790
Yamauchi T, Colmer TD, Pedersen O, Nakazono M (2018) Regulation of root traits for internal aeration and tolerance to soil waterlogging-flooding stress. Plant Physiol 176(2):1118–1130
Yan K, Zhao S, Cui M, Han G, Wen P (2018) Vulnerability of photosynthesis and photosystem I in Jerusalem artichoke (Helianthus tuberosus L.) exposed to waterlogging. Plant Physiol Biochem 125:239–246
Zhang P, Lyu D, Jia L, He J, Qin S (2017) Physiological and de novo transcriptome analysis of the fermentation mechanism of Cerasus sachalinensis roots in response to short-term waterlogging. BMC Genom 18:1–14
Zhou W, Chen F, Meng Y, Chandrasekaran U, Luo X, Yang W, Shu K (2020) Plant waterlogging/flooding stress responses: from seed germination to maturation. Plant Physiol Biochem 148:228–236
Acknowledgements
The authors are thankful to the UGC for financial assistance to carry out this work.
Funding
The authors are thankful to the UGC for financial assistance to carry out this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest. The authors declare that no human and/or animal material, data, or cell lines were used in this study.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, P., Jaiswal, S., Kushwaha, A. et al. Peroxynitrite is essential for aerenchyma formation in rice roots under waterlogging conditions. Planta 258, 2 (2023). https://doi.org/10.1007/s00425-023-04148-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-023-04148-6