Abstract
Main conclusion
Expression of TaSnRK2.7 promoter is strongly induced under abiotic stress and could be used as a valuable tool for improving plant stress resistance via transgenic techniques.
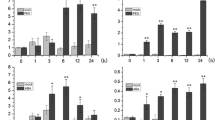
The sucrose non-fermenting 1-related protein kinase 2 (SnRK2) gene family plays pivotal roles in response to abiotic stresses (drought, salinity and cold). Here, we studied the expression of five wheat TaSnRK2.7 promoter-5′-deletion constructs (− 2547, − 1621, − 806, − 599, and − 254) fused to beta-glucuronidase (GUS) in Arabidopsis. Tissue-expression analysis revealed that the − 254 to ATG fragment was sufficient for inducing GUS expression in hypocotyls. Additionally, the − 806 to − 599 and − 2547 to − 1621 fragments contained leaf- and root-specific elements, respectively. Deletion analysis showed that these fragments were unresponsive to ABA treatment, suggesting that TaSnRK2.7 participates in an ABA-independent signaling pathway. Assays examining stress responses of constructs demonstrated that the − 599 to − 254 and − 806 to − 599 fragments contained elements responsive to abiotic and osmotic stress, respectively. The TaSnRK2.7 promoter contained enhancers from − 806 to − 254 and − 2547 to − 1621, while the − 1621 to − 806 fragment contained negative regulatory elements that restrict root and leaf gene expression in response to abiotic stress. Furthermore, under drought and salt stress, the TaSnRK2.7 promoter conferred greater gene expression in leaves than the rd29A promoter, even though both were induced by abiotic stress. These findings enhance our understanding of the molecular mechanisms behind TaSnRK2.7 action, which should prove useful in transgenic studies investigating stress-induced gene expression.




Similar content being viewed by others
References
Behnam B, Kikuchi A, Fevziye CT, Yamanaka S, Kasuga M, Yamaguchi-Shinozaki K, Watanabe KN (2006) The Arabidopsis DREB1A gene driven by the stress-inducible rd29A promoter increases salt-stress tolerance in proportion to its copy number in tetrasomic tetraploid potato (Solanum tuberosum). Plant Tissue Cultc Letters 23:169–177
Boudsocq M, Barbier-Brygoo H, Lauriere C (2004) Identification of nine sucrose nonfermenting 1-related protein kinases 2 activated by hyperosmotic and saline stresses in Arabidopsis thaliana. Biol Chem 279:41758–41766
Boudsocq M, Droillard MJ, Barbier-Brygoo H, Lauriere C (2007) Different phosphorylation mechanisms are involved in the activation of sucrose non-fermenting 1 related protein kinases 2 by osmotic stresses and abscisic acid. Plant Mol Biol 63:491–503
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Checker VG, Chhibbar AK, Khurana P (2012) Stress-inducible expression of barley Hva1 gene in transgenic mulberry displays enhanced tolerance against drought, salinity and cold stress. Transgenic Res 21:939–957
Christensen AH, Sharrock RA, Quail PH (1992) Maize polyubiquitin genes: structure, thermal perturbation of expression and transcript splicing, and promoter activity following transfer to protoplasts by electroporation. Plant Mol Biol 18:675–689
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16:735–743
Dubcovsky J, Dvorak J (2007) Genome plasticity a key factor in the success of polyploid wheat under domestication. Science 316:1862–1866
Dulai S, Molnar I, Pronay J, Csernak A, Tarnai R, Molnár-Láng M (2006) Effects of drought on photosynthetic parameters and heat stability of PSII in wheat and in Aegilops species originating from dry habitats. Acta Biol Szeged 50:11–17
Fujita Y, Nakashima K, Yoshida T, Katagiri T, Kidokoro S, Kanamori N, Umezawa T, Fujita M, Maruyama K, Ishiyama K (2009) Three SnRK2 protein kinases are the main positive regulators of abscisic acid signaling in response to water stress in Arabidopsis. Plant Cell Physiol 50:2123–2132
Gill BS, Friebe B, Raupp WJ, Wilson DL, Stan CT, Sears RG, BrownGuedira GL, Fritz AK (2006) Wheat genetics resource center: the first 25 years. Adv Agron 89:73–136
Gu YQ, Coleman-Derr D, Kong XY, Anderson OD (2004) Rapid genome evolution revealed by comparative sequence analysis of orthologous regions from four Triticeae genomes. Plant Physiol 135:459–470
Halford NG, Hardie DG (1998) SNF1-related protein kinases: global regulators of carbon metabolism in plants? Plant Mol Biol 37:735–748
Hou J, Jiang P, Qi S, Zhang K, He Q, Xu C, Ding Z, Zhang K, Li K (2016) Isolation and functional validation of salinity and osmotic stress inducible promoter from the maize type-II H + -pyrophosphatase gene by deletion analysis in transgenic tobacco plants. PLoS One 5:e16041
Hsieh TH, Lee JT, Charng YY, Chan MT (2002) Tomato plants ectopically expressing Arabidopsis CBF1 show enhanced resistance to water deficit stress. Plant Physiol 130:618–626
Huai J, Wang M, He J, Zheng J, Dong Z, Lv H, Zhao J, Wang G (2008) Cloning and characterization of the SnRK2 gene family from Zea mays. Plant Cell Rep 27:1861–1868
Jefferson RA (1987) Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol Biol Rep 5:387–405
Kasuga M, Miura S, Shinozaki K, Yamaguchi-Shinozaki K (2004) A combination of the Arabidopsis DREB1A gene and stress inducible rd29A promoter improved drought- and lowtemperature stress tolerance in tobacco by gene transfer. Plant Cell Physiol 45:346–350
Kelemen Z, SebastianA XuW, Grain D, Salsac F, Avon A, Berger N, Tran J, Dubreucq B, Lurin C, Lepiniec L, Contreras-Moreira B, Dubos C (2015) Analysis of the DNA-binding activities of the Arabidopsis R2R3-MYB transcription factor family by one-hybrid experiments in yeast. PLoS One 10:e0141044
Kobayashi Y, Yamamoto S, Minami H, Kagaya Y, Hattori T (2004) Differential activation of the rice sucrose nonfermenting1-related protein kinase2 family by hyperosmotic stress and abscisic acid. Plant Cell 16:1163–1177
Kobayashi Y, Murata M, Minami H, Yamamoto S, Kagaya Y, Hobo T, Yamamoto A, Hattori T (2005) Abscisic acid-activated SNRK2 protein kinases function in the gene-regulation pathway of ABA signal transduction by phosphorylating ABA response element-binding factors. Plant J 44:939–949
Lee SC, Luan S (2012) ABA signal transduction at the crossroad of biotic and abiotic stress responses. Plant Cell Environ 35:53–60
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2–∆∆CT method. Methods 25:402–408
Mao XG, Zhang HY, Tian SJ, Chang XP, Jing RL (2009) TaSnRK2.4, an SNF1-type serine/threonine protein kinase of wheat (Triticum aestivum L.), confers enhanced multistress tolerance in Arabidopsis. J Exp Bot 61:683–696
McElroy D, Zhang W, Cao J, Wu R (1990) Isolation of an efficient actin promoter for use in rice transformation. Plant Cell 2:163–171
Melcher K, Ng LM, Zhou XE, Soon FF, Xu Y, Suino-Powell KM, Park SY, Weiner JJ, Fujii H, Chinnusamy V, Korte A, Li J, Wang YH, Li JY, Peterson FC, Jensen DR, Yong EL, Volkman BF, Cutler SR, Zhu JK, Xu HE (2009) A gate-latch-lock mechanism for hormone signaling by abscisic acid receptors. Nature 462:602–608
Mizoguchi M, Umezawa T, Nakashima K, Kidokoro S, Takasaki H, Fujita Y, Yamaguchi-Shinozaki K, Shinozaki K (2010) Two closely related subclass II SnRK2 protein kinases cooperatively regulate drought-inducible gene expression. Plant Cell Physiol 51:842–847
Mustilli A, Merlot S, Vavasseur A, Fenzi F, Giraudat J (2002) Arabidopsis OST1 protein kinase mediates the regulation of stomatal aperture by abscisic acid and acts upstream of reactive oxygen species production. Plant Cell 14:3089
Nishimura N, Hitomi K, Arvai AS, Rambo RP, Hitomi C, Cutler SR, Schroeder JI, Getzoff ED (2009) Structural mechanism of abscisic acid binding and signaling by dimeric PYR1. Science 326(5958):1373–1379
Odell JT, Nagy F, Chua NH (1985) Identification of DNA sequences required for activity of the cauliflower mosaic virus 35S promoter. Nature 313(6005):810–812
Pellegrineschi A, Reynolds M, Pacheco M, Brito RM, Almeraya R, Yamaguchi-Shinozaki K, Hoisington D (2004) Stress-induced expression in wheat of the Arabidopsis thaliana DREB1A gene delays water stress symptoms under greenhouse conditions. Genome 47:493–500
Raghavendra AS, Gonugunta VK, Christmann A, Grill E (2010) ABA perception and signaling. Trends Plant Sci 15:395–401
Rai M, He C, Wu R (2009) Comparative functional analysis of three abiotic stress-inducible promoters in transgenic rice. Transgenic Res 18:787–799
Rampino P, Pataleo S, Gerardi C, Mita G, Perrotta C (2006) Drought stress response in wheat: physiological and molecular analysis of resistant and sensitive genotypes. Plant Cell Environ 29:2143–2152
Rerksiri W, Zhang X, Xiong H, Chen X (2013) Expression and promoter analysis of six heat stress-inducible genes in rice. Sci World J 20:57–69
Sheard LB, Zheng N (2009) Plant biology: signal advance for abscisic acid. Nature 462:575–576
Tian SJ, Mao XG, Zhang HY, Chen SS, Zhai CC, Yang SM, Jing RL (2013) Cloning and characterization of TaSnRK2.3, a novel SnRK2 gene in common wheat. J Exp Bot 64:2063–2080
Yamaguchi-Shinozaki K, Mino M, Mundy J, Chua NH (1990) Analysis of an ABA-responsive rice gene promoter in transgenic tobacco. Plant Mol Biol 15:905–912
Zavallo D, Bilbao ML, Hopp HE, Heinz R (2010) Isolation and functional characterization of two novel seedspecific promoters from sunflower (Helianthus annuus L.). Plant Cell Rep 29:239–248
Zhang HY, Mao XG, Wang CS, Jing RL (2010) Overexpression of a common wheat gene TaSnRK2.8 enhances tolerance to drought, salt and low temperature in Arabidopsis. PLoS One 5:e16041
Zhang HY, Mao XG, Jing RL, Xie HM (2011) Characterization of a common wheat (Triticum aestivum L.) TaSnRK2.7 gene involved in abiotic stress responses. J Exp Bot 62:975–988
Zhang HY, Wei LI, Mao XG, Jing RL (2015) Characterization of genomic sequence of a drought-resistant gene TaSnRK2.7 in wheat species. J Genet 94:1–6
Zhang HY, Li WY, Mao XG, Jing RL, Jia HF (2016) Differential activation of the wheat SnRK2 family by abiotic stresses. Front Plant Sci 7:420
Zhang HY, Mao XG, Jing RL (2017) Functional Characterization of TaSnRK2.8 promoter in response to abiotic stresses by deletion analysis in transgenic Arabidopsis. Front Plant Sci 8:1198
Acknowledgements
This work was supported by the National Science Foundation of China (NSF31401371).
Author information
Authors and Affiliations
Contributions
Author Contributions
JAW performed the deletion analysis experiments. ZPS and SY participated to the data analysis. HFJ performed the qRT-PCR experiments, HYZ projected design and supervision.
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, J., Song, Z., Jia, H. et al. Characterization of wheat TaSnRK2.7 promoter in Arabidopsis. Planta 248, 1393–1401 (2018). https://doi.org/10.1007/s00425-018-2984-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-018-2984-9