Abstract
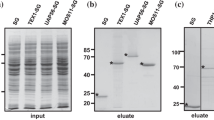
The exon junction complex (EJC) plays an important role in post-transcriptional control of gene expression. Mago nashi (Mago) and Y14 are core EJC proteins that operate as a functional unit in animal cells, and the Mago-Y14 heterodimer interacts with other EJC core and peripheral proteins. Little is known about the biochemical and cellular characteristics of the EJC and its orthologs in plants. Here, we demonstrate that Arabidopsis Mago and Y14 form a ternary complex with PYM, an RNA-binding protein that was previously shown to interact with the Mago-Y14 heterodimer in Drosophila. Fluorescence microscopy indicated that Arabidopsis Mago and Y14 are localized primarily in the nucleus, whereas PYM is mostly cytoplasmic. In vitro pull-down assays using recombinant proteins showed that the amino-terminal region of the Arabidopsis PYM interacts with the Mago-Y14 heterodimer, a similar observation to that previously reported for the animal versions of these proteins. However, we demonstrated also that Arabidopsis PYM has the ability to interact with monomeric Mago and monomeric Y14. Immunoprecipitation and tandem affinity purification from whole cell extracts detected a subtle interaction between the Arabidopsis Mago-Y14 heterodimer and PYM in flowers, indicating that the ternary complex is not abundant in plant cells. The regions of the polypeptide responsible for nuclear import and export were defined using protein truncations and site-directed mutagenesis. This study identifies unique characteristics of Arabidopsis Mago, Y14 and PYM compared to those observed in animal cells. These are predicted to have important functional implications associated with post-transcriptional regulation of gene expression in plant cells.






Similar content being viewed by others
Abbreviations
- EJC:
-
Exon junction complex
- NMD:
-
Nonsense-mediated decay
- At:
-
Arabidopsis thaliana
- TAP tag:
-
Tandem-affinity purification tag
- GFP:
-
Green fluorescent protein
- RFP:
-
Red fluorescent protein
References
Bono F, Ebert J, Unterholzner L, Guttler T, Izaurralde E, Conti E (2004) Molecular insights into the interaction of PYM with the Mago-Y14 core of the exon junction complex. EMBO Rep 5:304–310
Boswell RE, Prout ME, Steichen JC (1991) Mutations in a newly identified Drosophila melanogaster gene, mago nashi, disrupt germ cell formation and result in the formation of mirror-image symmetrical double abdomen embryos. Development 113:373–384
Chuong SD, Mullen RT, Muench DG (2005) The peroxisomal multifunctional protein interacts with cortical microtubules in plant cells. BMC Cell Biol 6:40
Clough S, Bent A (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Conti E, Izaurralde E (2005) Nonsense-mediated mRNA decay: molecular insights and mechanistic variations across species. Curr Opin Cell Biol 17:316–325
Dickey LF, Nguyen TT, Allen GC, Thompson WF (1994) Light modulation of ferredoxin mRNA abundance requires an open reading frame. Plant Cell 6:1171–1176
Dreyfuss G, Kim VN, Kataoka N (2002) Messenger-RNA-binding proteins and the messages they carry. Nat Rev Mol Cell Biol 3:195–205
Forler D, Kocher T, Rode M, Gentzel M, Izaurralde E, Wilm M (2003) An efficient protein complex purification method for functional proteomics in higher eukaryotes. Nat Biotechnol 21:89–92
Fribourg S, Gatfield D, Izaurralde E, Conti E (2003) A novel mode of RBD-protein recognition in the Y14-Mago complex. Nat Struct Biol 10:433–439
Gehring NH, Kunz JB, Neu-Yilik G, Breit S, Viegas MH, Hentze MW, Kulozik AE (2005) Exon-junction complex components specify distinct routes of nonsense-mediated mRNA decay with differential cofactor requirements. Mol Cell 20:65–75
Gonzalez CI, Wang W, Peltz SW (2001) Nonsense-mediated mRNA decay in Saccharomyces cerevisiae: a quality control mechanism that degrades transcripts harboring premature termination codons. Cold Spring Harb Symp Quant Biol 66:321–328
Hachet O, Ephrussi A (2001) Drosophila Y14 shuttles to the posterior of the oocyte and is required for oskar mRNA transport. Curr Biol 11:1666–1674
Hachet O, Ephrussi A (2004) Splicing of oskar RNA in the nucleus is coupled to its cytoplasmic localization. Nature 428:959–963
van Hoof A, Green PJ (1996) Premature nonsense codons decrease the stability of phytohemagglutinin mRNA in a position-dependent manner. Plant J 10:415–424
Jofuku KD, Schipper RD, Goldberg RB (1989) A frameshift mutation prevents Kunitz trypsin inhibitor mRNA accumulation in soybean embryos. Plant Cell 1:567
Johnson MA, von Besser K, Zhou Q, Smith E, Aux G, Patton D, Levin JZ, Preuss D (2004) Arabidopsis hapless mutations define essential gametophytic functions. Genetics 168:971–982
Kataoka N, Yong J, Kim VN, Velazquez F, Perkinson RA, Wang F, Dreyfuss G (2000) Pre-mRNA splicing imprints mRNA in the nucleus with a novel RNA-binding protein that persists in the cytoplasm. Mol Cell 6:673–682
Lau CK, Diem MD, Dreyfuss G, Van Duyne GD (2003) Structure of the Y14-Magoh core of the exon junction complex. Curr Biol 13:933–941
Le Hir H, Izaurralde E, Maquat LE, Moore MJ (2000a) The spliceosome deposits multiple proteins 20–24 nucleotides upstream of mRNA exon-exon junctions. Embo J 19:6860–6869
Le Hir H, Moore MJ, Maquat LE (2000b) Pre-mRNA splicing alters mRNP composition: evidence for stable association of proteins at exon-exon junctions. Genes Dev 14:1098–1108
Lejeune F, Maquat LE (2005) Mechanistic links between nonsense-mediated mRNA decay and pre-mRNA splicing in mammalian cells. Curr Opin Cell Biol 17:309–315
Lorkovic ZJ, Hilscher J, Barta A (2004) Use of fluorescent protein tags to study nuclear organization of the spliceosomal machinery in transiently transformed living plant cells. Mol Biol Cell 15:3233–3243
Lorkovic ZJ, Barta A (2004) Compartmentalization of the splicing machinery in plant cell nuclei. Trends Plant Sci 9:565–568
McCartney AW, Greenwood JS, Fabian MR, White KA, Mullen RT (2005) Localization of the tomato bushy stunt virus replication protein p33 reveals a peroxisome-to-endoplasmic reticulum sorting pathway. Plant Cell 17:3513–3531
Mohr SE, Dillon ST, Boswell RE (2001) The RNA-binding protein Tsunagi interacts with Mago nashi to establish polarity and localize oskar mRNA during Drosophila oogenesis. Genes Dev 15:2886–2899
Muench DG, Park NI (2006) Messages on the move: the role of the cytoskeleton in mRNA localization and translation in plant cells. Can J Bot 84:572–580
Newmark PA, Boswell RE (1994) The mago nashi locus encodes an essential product required for germ plasm assembly in Drosophila. Development 120:1303–1313
Nott A, Le Hir H, Moore MJ (2004) Splicing enhances translation in mammalian cells: an additional function of the exon junction complex. Genes Dev 18:210–222
Orphanides G, Reinberg D (2002) A unified theory of gene expression. Cell 108:439–451
Palacios IM, Gatfield D, St Johnston D, Izaurralde E (2004) An eIF4AIII-containing complex required for mRNA localization and nonsense-mediated mRNA decay. Nature 427:753–757
Pendle AF, Clark GP, Boon R, Lewandowska D, Lam YW, Andersen J, Mann M, Lamond AI, Brown JW, Shaw PJ (2005) Proteomic analysis of the Arabidopsis nucleolus suggests novel nucleolar functions. Mol Biol Cell 16:260–269
Petracek ME, Nuygen T, Thompson WF, Dickey LF (2000) Premature termination codons destabilize ferredoxin-1 mRNA when ferredoxin-1 is translated. Plant J 21:563–569
Rigaut G, Shevchenko A, Rutz B, Wilm M, Mann M, Seraphin B (1999) A generic protein purification method for protein complex characterization and proteome exploration. Nat Biotechnol 17:1030–1032
Rohila JS, Chen M, Cerny R, Fromm ME (2004) Improved tandem affinity purification tag and methods for isolation of protein heterocomplexes from plants. Plant J 38:172–181
Scott A, Wyatt S, Tsou PL, Robertson D, Allen NS (1999) Model system for plant cell biology: GFP imaging in living onion epidermal cells. Biotechniques 26:1125, 1128–1132
Seki M, Carninci P, Nishiyama Y, Hayashizaki Y, Shinozaki K (1998) High-efficiency cloning of Arabidopsis full-length cDNA by biotinylated CAP trapper. Plant J 15:707–720
Seki M, Narusaka M, Kamiya A, Ishida J, Satou M, Sakurai T, Nakajima M, Enju A, Akiyama K, Oono Y, Muramatsu M, Hayashizaki Y, Kawai J, Carninci P, Itoh M, Ishii Y, Arakawa T, Shibata K, Shinagawa A, Shinozaki K (2002) Functional annotation of a full-length Arabidopsis cDNA collection. Science 296:141–145
Shi H, Xu RM (2003) Crystal structure of the Drosophila Mago nashi-Y14 complex. Genes Dev 17:971–976
Strom AC, Weis K (2001) Importin-beta-like nuclear transport receptors. Genome Biol 2: REVIEWS3008
Swidzinski J, Zaplachinski S, Chuong SDX, Wong J, Muench DG (2001) Molecular characterization and expression analysis of a highly conserved rice mago nashi homolog. Genome 44:394–400
Tange TO, Shibuya T, Jurica MS, Moore MJ (2005) Biochemical analysis of the EJC reveals two new factors and a stable tetrameric protein core. Rna 11:1869–1883
Voelker TA, Moreno J, Chrispeels MJ (1990) Expression analysis of a pseudogene in transgenic tobacco: a frameshift mutation prevents mRNA accumulation. Plant Cell 2:255–261
Zhao X-F, Nowak N, Shows T, Aplan P (2000) MAGOH interacts with a novel RNA-binding protein. Genomics 63:145–148
Zimmermann P, Hirsch-Hoffmann M, Hennig L, Gruissem W (2004) GENEVESTIGATOR. Arabidopsis microarray database and analysis toolbox. Plant Physiol 136:2621–2632
Acknowledgments
We thank Michael Fromm (University of Nebraska) for providing us with the TAP-Tag expression cassette and TAP-GUS control seeds. We are grateful to Andrea Barta (Vienna Biocenter, Institute of Medical Biochemistry) for providing the SR-RFP and RFP-PRH75 clones, and Robert Mullen (University of Guelph) for providing the pRTL2ΔNS/GFP and pRTL2ΔNS/RFP clones. Mark Johnson (Brown University) and Catherine Smith (University of Calgary) provided valuable comments and assisted in editing the manuscript. We also thank Marie Fraser (University of Calgary) for assistance in the interpretation of the AtPYM binding results, and Raymond J. Turner (University of Calgary) for advice on recombinant protein expression. The Arabidopsis full-length cDNA clones were provided by the RIKEN BioResource Center. Financial support for this research was provided by a Natural Sciences and Engineering Research Council of Canada (NSERC) award to DGM, and a Bettina Bahlsen Memorial Graduate Scholarship to NP.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Park, Ni., Muench, D.G. Biochemical and cellular characterization of the plant ortholog of PYM, a protein that interacts with the exon junction complex core proteins Mago and Y14. Planta 225, 625–639 (2007). https://doi.org/10.1007/s00425-006-0385-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-006-0385-y