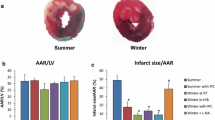
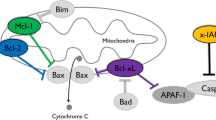
Abstract
Hibernation allows animals to enter an energy conserving state to survive severe drops in external temperatures and a shortage of food. It has been observed that the hearts of mammalian hibernators exhibit intrinsic protection against ischemia-reperfusion (I/R) injury and cardiac arrhythmias in the winter whether they are hibernating or not. However, the molecular and ionic mechanisms for cardioprotection in mammalian hibernators remain elusive. Recent studies in woodchucks (Marmota monax) have suggested that cardiac adaptation occurs at different levels and mediates an intrinsic cardioprotection prior to/in the winter. The molecular/cellular remodeling in the winter (with or without hibernation) includes (1) an upregulation of transcriptional factor, anti-apoptotic factor, nitric oxide synthase, protein kinase C-ε, and phosphatidylinositol-4,5-bisphosphate 3-kinase; (2) an upregulation of antioxidant enzymes (e.g. superoxide dismutase and catalase); (3) a reduction in the oxidation level of Ca2+/calmodulin-dependent protein kinase II (CaMKII); and (4) alterations in the expression and activity of multiple ion channels/transporters. Therefore, the cardioprotection against I/R injury in the winter is most likely mediated by enhancement in signaling pathways that are shared by preconditioning, reduced cell apoptosis, and increased detoxification of reactive oxygen species (ROS). The resistance to cardiac arrhythmias and sudden cardiac death in the winter is closely associated with an upregulation of the antioxidant catalase and a downregulation of CaMKII activation. This remodeling of the heart is associated with a reduction in the incidence of afterdepolarizations and triggered activities. In this short review article, we will discuss the seasonal changes in gene and protein expression profiles as well as alterations in the function of key proteins that are associated with the occurrence of cardioprotection against myocardial damage from ischemic events and fatal arrhythmias in a mammalian hibernator. Understanding the intrinsic cardiac adaptive mechanisms that confer cardioprotection in hibernators may offer new strategies to protect non-hibernating animals, especially humans, from I/R injury and ischemia-induced fatal cardiac arrhythmias.



Similar content being viewed by others
References
Alekseev AE, Markevich NI, Korystova AF, Terzic A, Kokoz YM (1996) Comparative analysis of the kinetic characteristics of L-type calcium channels in cardiac cells of hibernators. Biophys J 70:786–797. https://doi.org/10.1016/S0006-3495(96)79618-2
Andrews MT (2007) Advances in molecular biology of hibernation in mammals. Bioessays 29:431–440
Andrews MT (2019) Molecular interactions underpinning the phenotype of hibernation in mammals. J Exp Biol 222:jeb160606. https://doi.org/10.1242/jeb.160606
Aquilano K, Baldelli S, Pagliei B, Cannata SM, Rotilio G, Ciriolo MR (2013) p53 orchestrates the PGC-1alpha-mediated antioxidant response upon mild redox and metabolic imbalance. Antioxid Redox Signal 18:386–399. https://doi.org/10.1089/ars.2012.4615
Baba Y, Higa JK, Shimada BK, Horiuchi KM, Suhara T, Kobayashi M, Woo JD, Aoyagi H, Marh KS, Kitaoka H, Matsui T (2018) Protective effects of the mechanistic target of rapamycin against excess iron and ferroptosis in cardiomyocytes. Am J Physiol Heart Circ Physiol 314:H659–H668. https://doi.org/10.1152/ajpheart.00452.2017
Becker LB (2004) New concepts in reactive oxygen species and cardiovascular reperfusion physiology. Cardiovasc Res 61:461–470. https://doi.org/10.1016/j.cardiores.2003.10.025
Belke DD, Wang LC, Lopaschuk GD (1998) Acetyl-CoA carboxylase control of fatty acid oxidation in hearts from hibernating Richardson's ground squirrels. Biochim Biophys Acta 1391:25–36. https://doi.org/10.1016/s0005-2760(97)00199-9
Berridge MJ (2012) Calcium signalling remodelling and disease. Biochem Soc Trans 40:297–309. https://doi.org/10.1042/BST20110766
Brauch KM, Dhruv ND, Hanse EA, Andrews MT (2005) Digital transcriptome analysis indicates adaptive mechanisms in the heart of a hibernating mammal. Physiol Genomics 23:227–234
Buck MJ, Squire TL, Andrews MT (2002) Coordinate expression of the PDK4 gene: a means of regulating fuel selection in a hibernating mammal. Physiol Genomics 8:5–13. https://doi.org/10.1152/physiolgenomics.00076.2001
Carey HV, Andrews MT, Martin SL (2003) Mammalian hibernation: cellular and molecular responses to depressed metabolism and low temperature. Physiol Rev 83:1153–1181
Chen J, Zhang Z, Cai L (2014) Diabetic cardiomyopathy and its prevention by nrf2: current status. Diabetes Metab J 38:337–345. https://doi.org/10.4093/dmj.2014.38.5.337
Couchonnal LF, Anderson ME (2008) The role of calmodulin kinase II in myocardial physiology and disease. Physiology (Bethesda) 23:151–159
Dawe AR, Morrison PR (1955) Characteristics of the hibernating heart. Am Heart J 49:367–384. https://doi.org/10.1016/0002-8703(55)90031-4
De Cesare D, Fimia GM, Sassone-Corsi P (1999) Signaling routes to CREM and CREB: plasticity in transcriptional activation. Trends Biochem Sci 24:281–285. https://doi.org/10.1016/s0968-0004(99)01414-0
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, Patel DN, Bauer AJ, Cantley AM, Yang WS, Morrison B 3rd, Stockwell BR (2012) Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149:1060–1072. https://doi.org/10.1016/j.cell.2012.03.042
Dreger H, Westphal K, Weller A, Baumann G, Stangl V, Meiners S, Stangl K (2009) Nrf2-dependent upregulation of antioxidative enzymes: a novel pathway for proteasome inhibitor-mediated cardioprotection. Cardiovasc Res 83:354–361. https://doi.org/10.1093/cvr/cvp107
Drew KL, Buck CL, Barnes BM, Christian SL, Rasley BT, Harris MB (2007) Central nervous system regulation of mammalian hibernation: implications for metabolic suppression and ischemia tolerance. J Neurochem 102:1713–1726. https://doi.org/10.1111/j.1471-4159.2007.04675.x
Dugbartey GJ, Bouma HR, Strijkstra AM, Boerema AS, Henning RH (2015) Induction of a torpor-like state by 5'-AMP does not depend on H2S production. PLoS One 10:e0136113. https://doi.org/10.1371/journal.pone.0136113
Erickson JR, Joiner ML, Guan X, Kutschke W, Yang J, Oddis CV, Bartlett RK, Lowe JS, O'Donnell SE, Aykin-Burns N, Zimmerman MC, Zimmerman K, Ham AJ, Weiss RM, Spitz DR, Shea MA, Colbran RJ, Mohler PJ, Anderson ME (2008) A dynamic pathway for calcium-independent activation of CaMKII by methionine oxidation. Cell 133:462–474
Fang X, Wang H, Han D, Xie E, Yang X, Wei J, Gu S, Gao F, Zhu N, Yin X, Cheng Q, Zhang P, Dai W, Chen J, Yang F, Yang HT, Linkermann A, Gu W, Min J, Wang F (2019) Ferroptosis as a target for protection against cardiomyopathy. Proc Natl Acad Sci U S A 116:2672–2680. https://doi.org/10.1073/pnas.1821022116
Fang X, Cai Z, Wang H, Han D, Cheng Q, Zhang P, Gao F, Yu Y, Song Z, Wu Q, An P, Huang S, Pan J, Chen HZ, Chen J, Linkermann A, Min J, Wang F (2020) Loss of cardiac ferritin H facilitates cardiomyopathy via Slc7a11-mediated ferroptosis. Circ Res 127:486–501. https://doi.org/10.1161/CIRCRESAHA.120.316509
Fedorov VV, Li L, Glukhov A, Shishkina I, Aliev RR, Mikheeva T, Nikolski VP, Rosenshtraukh LV, Efimov IR (2005) Hibernator Citellus undulatus maintains safe cardiac conduction and is protected against tachyarrhythmias during extreme hypothermia: possible role of Cx43 and Cx45 up-regulation. Heart Rhythm 2:966–975
Frerichs KU, Smith CB, Brenner M, DeGracia DJ, Krause GS, Marrone L, Dever TE, Hallenbeck JM (1998) Suppression of protein synthesis in brain during hibernation involves inhibition of protein initiation and elongation. Proc Natl Acad Sci U S A 95:14511–14516. https://doi.org/10.1073/pnas.95.24.14511
Gao TL, Huang YZ, Wang J (1996) The resistance to ischemia-reperfusion injury of the isolated heart from hibernator Citellus dauricus. Acta Sci Nat Univ Pekinensis 32:527–533
Hampton M, Melvin RG, Kendall AH, Kirkpatrick BR, Peterson N, Andrews MT (2011) Deep sequencing the transcriptome reveals seasonal adaptive mechanisms in a hibernating mammal. PLoS One 6:e27021. https://doi.org/10.1371/journal.pone.0027021
Herzig S, Long F, Jhala US, Hedrick S, Quinn R, Bauer A, Rudolph D, Schutz G, Yoon C, Puigserver P, Spiegelman B, Montminy M (2001) CREB regulates hepatic gluconeogenesis through the coactivator PGC-1. Nature 413:179–183. https://doi.org/10.1038/35093131
Heusch G (2015) Molecular basis of cardioprotection: signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res 116:674–699. https://doi.org/10.1161/CIRCRESAHA.116.305348
Jinka TR, Toien O, Drew KL (2011) Season primes the brain in an arctic hibernator to facilitate entrance into torpor mediated by adenosine a(1) receptors. J Neurosci 31:10752–10758. https://doi.org/10.1523/JNEUROSCI.1240-11.2011
Johansson BW (1985) Ventricular repolarization and fibrillation threshold in hibernating species. Eur Heart J 6(Suppl D):53–62
Johansson BW (1996) The hibernator heart--nature's model of resistance to ventricular fibrillation. Cardiovasc Res 31:826–832
Kim HP, Roe JH, Chock PB, Yim MB (1999) Transcriptional activation of the human manganese superoxide dismutase gene mediated by tetradecanoylphorbol acetate. J Biol Chem 274:37455–37460. https://doi.org/10.1074/jbc.274.52.37455
Kinugawa S, Tsutsui H, Hayashidani S, Ide T, Suematsu N, Satoh S, Utsumi H, Takeshita A (2000) Treatment with dimethylthiourea prevents left ventricular remodeling and failure after experimental myocardial infarction in mice: role of oxidative stress. Circ Res 87:392–398
Kronke G, Bochkov VN, Huber J, Gruber F, Bluml S, Furnkranz A, Kadl A, Binder BR, Leitinger N (2003) Oxidized phospholipids induce expression of human heme oxygenase-1 involving activation of cAMP-responsive element-binding protein. J Biol Chem 278:51006–51014. https://doi.org/10.1074/jbc.M304103200
Lakatta EG, Sollott SJ (2002) The "heartbreak" of older age. Mol Interv 2:431–446
Li H, Liu T, Chen W, Jain MR, Vatner DE, Vatner SF, Kudej RK, Yan L (2013) Proteomic mechanisms of cardioprotection during mammalian hibernation in woodchucks, Marmota monax. J Proteome Res 12:4221–4229. https://doi.org/10.1021/pr400580f
Luczak ED, Anderson ME (2014) CaMKII oxidative activation and the pathogenesis of cardiac disease. J Mol Cell Cardiol 73:112–116. https://doi.org/10.1016/j.yjmcc.2014.02.004
Marczin N, El-Habashi N, Hoare GS, Bundy RE, Yacoub M (2003) Antioxidants in myocardial ischemia-reperfusion injury: therapeutic potential and basic mechanisms. Arch Biochem Biophys 420:222–236
Morin P Jr, Storey KB (2007) Antioxidant defense in hibernation: cloning and expression of peroxiredoxins from hibernating ground squirrels, Spermophilus tridecemlineatus. Arch Biochem Biophys 461:59–65
Morin P Jr, Ni Z, McMullen DC, Storey KB (2008) Expression of Nrf2 and its downstream gene targets in hibernating 13-lined ground squirrels, Spermophilus tridecemlineatus. Mol Cell Biochem 312:121–129. https://doi.org/10.1007/s11010-008-9727-3
Nardone RM (1955) Electrocardiogram of the arctic ground squirrel during hibernation and hypothermia. Am J Phys 182:364–368. https://doi.org/10.1152/ajplegacy.1955.182.2.364
Nelson OL, Robbins CT (2015) Cardiovascular function in large to small hibernators: bears to ground squirrels. J Comp Physiol B 185:265–279. https://doi.org/10.1007/s00360-014-0881-5
Neuman RB, Bloom HL, Shukrullah I, Darrow LA, Kleinbaum D, Jones DP, Dudley SC Jr (2007) Oxidative stress markers are associated with persistent atrial fibrillation. Clin Chem 53:1652–1657
Nordberg J, Arner ES (2001) Reactive oxygen species, antioxidants, and the mammalian thioredoxin system. Free Radic Biol Med 31:1287–1312
Ohta H, Okamoto I, Hanaya T, Arai S, Ohta T, Fukuda S (2006) Enhanced antioxidant defense due to extracellular catalase activity in Syrian hamster during arousal from hibernation. Comp Biochem Physiol C Toxicol Pharmacol 143:484–491
Oka SI, Sabry AD, Cawley KM, Warren JS (2020) Multiple levels of PGC-1alpha Dysregulation in heart failure. Front Cardiovasc Med 7:2. https://doi.org/10.3389/fcvm.2020.00002
Olson JM, Jinka TR, Larson LK, Danielson JJ, Moore JT, Carpluck J, Drew KL (2013) Circannual rhythm in body temperature, torpor, and sensitivity to a(1) adenosine receptor agonist in arctic ground squirrels. J Biol Rhythm 28:201–207. https://doi.org/10.1177/0748730413490667
Piktel JS, Jeyaraj D, Said TH, Rosenbaum DS, Wilson LD (2011) Enhanced dispersion of repolarization explains increased arrhythmogenesis in severe versus therapeutic hypothermia. Circ Arrhythm Electrophysiol 4:79–86. https://doi.org/10.1161/CIRCEP.110.958355
Rankin AC, Rae AP (1984) Cardiac arrhythmias during rewarming of patients with accidental hypothermia. Br Med J 289:874–877. https://doi.org/10.1136/bmj.289.6449.874
Rodrigo R, Cereceda M, Castillo R, Asenjo R, Zamorano J, Araya J, Castillo-Koch R, Espinoza J, Larrain E (2008) Prevention of atrial fibrillation following cardiac surgery: basis for a novel therapeutic strategy based on non-hypoxic myocardial preconditioning. Pharmacol Ther 118:104–127
Rubart M, Zipes DP (2005) Mechanisms of sudden cardiac death. J Clin Invest 115:2305–2315
Slezak J, Tribulova N, Pristacova J, Uhrik B, Thomas T, Khaper N, Kaul N, Singal PK (1995) Hydrogen peroxide changes in ischemic and reperfused heart. Cytochemistry and biochemical and X-ray microanalysis. Am J Pathol 147:772–781
Song Y, Shryock JC, Wagner S, Maier LS, Belardinelli L (2006) Blocking late sodium current reduces hydrogen peroxide-induced arrhythmogenic activity and contractile dysfunction. J Pharmacol Exp Ther 318:214–222
Steven A, Leisz S, Wickenhauser C, Schulz K, Mougiakakos D, Kiessling R, Denkert C, Seliger B (2017) Linking CREB function with altered metabolism in murine fibroblast-based model cell lines. Oncotarget 8:97439–97463. https://doi.org/10.18632/oncotarget.22135
St-Pierre J, Drori S, Uldry M, Silvaggi JM, Rhee J, Jager S, Handschin C, Zheng K, Lin J, Yang W, Simon DK, Bachoo R, Spiegelman BM (2006) Suppression of reactive oxygen species and neurodegeneration by the PGC-1 transcriptional coactivators. Cell 127:397–408. https://doi.org/10.1016/j.cell.2006.09.024
Vermillion KL, Jagtap P, Johnson JE, Griffin TJ, Andrews MT (2015) Characterizing cardiac molecular mechanisms of mammalian hibernation via quantitative Proteogenomics. J Proteome Res 14:4792–4804. https://doi.org/10.1021/acs.jproteome.5b00575
Wagner S, Dybkova N, Rasenack EC, Jacobshagen C, Fabritz L, Kirchhof P, Maier SK, Zhang T, Hasenfuss G, Brown JH, Bers DM, Maier LS (2006) Ca2+/calmodulin-dependent protein kinase II regulates cardiac Na+ channels. J Clin Invest 116:3127–3138
Wang SQ, Lakatta EG, Cheng H, Zhou ZQ (2002) Adaptive mechanisms of intracellular calcium homeostasis in mammalian hibernators. J Exp Biol 205:2957–2962
Ward CA, Giles WR (1997) Ionic mechanism of the effects of hydrogen peroxide in rat ventricular myocytes. J Physiol 500(Pt 3):631–642
Xie LH, Chen F, Karagueuzian HS, Weiss JN (2009) Oxidative-stress-induced afterdepolarizations and calmodulin kinase II signaling. Circ Res 104:79–86. https://doi.org/10.1161/CIRCRESAHA.108.183475
Xie LH, Wen H, Zhao Z, Fefelova N, Allen C (2012) Calcium handling properties in a hibernating animal: insights into antiarrhythmic mechanisms. Biophys J 102:102
Yan L, Kudej RK, Vatner DE, Vatner SF (2015) Myocardial ischemic protection in natural mammalian hibernation. Basic Res Cardiol 110:9. https://doi.org/10.1007/s00395-015-0462-0
Yatani A, Kim SJ, Kudej RK, Wang Q, Depre C, Irie K, Kranias EG, Vatner SF, Vatner DE (2004) Insights into cardioprotection obtained from study of cellular Ca2+ handling in myocardium of true hibernating mammals. Am J Physiol Heart Circ Physiol 286:H2219–H2228
Zakharova NM, Tarahovsky YS, Fadeeva IS, Komelina NP, Khrenov MO, Glushkova OV, Prokhorov DA, Kutyshenko VP, Kovtun AL (2019) A pharmacological composition for induction of a reversible torpor-like state and hypothermia in rats. Life Sci 219:190–198. https://doi.org/10.1016/j.lfs.2019.01.023
Zhao Z, Fefelova N, Shanmugam M, Bishara P, Babu GJ, Xie LH (2011) Angiotensin II induces afterdepolarizations via reactive oxygen species and calmodulin kinase II signaling. J Mol Cell Cardiol 50:128–136. https://doi.org/10.1016/j.yjmcc.2010.11.001
Zhao Z, Wen H, Fefelova N, Allen C, Baba A, Matsuda T, Xie LH (2012) Revisiting the ionic mechanisms of early afterdepolarizations in cardiomyocytes: predominant by Ca waves or Ca currents? Am J Physiol Heart Circ Physiol 302:H1636–H1644. https://doi.org/10.1152/ajpheart.00742.2011
Zhao Z, Babu GJ, Wen H, Fefelova N, Gordan R, Sui X, Yan L, Vatner DE, Vatner SF, Xie LH (2015) Overexpression of adenylyl cyclase type 5 (AC5) confers a proarrhythmic substrate to the heart. Am J Physiol Heart Circ Physiol 308:H240–H249. https://doi.org/10.1152/ajpheart.00630.2014
Zhao Z, Kudej RK, Wen H, Fefelova N, Yan L, Vatner DE, Vatner SF, Xie LH (2018) Antioxidant defense and protection against cardiac arrhythmias: lessons from a mammalian hibernator (the woodchuck). FASEB J 32:4229–4240. https://doi.org/10.1096/fj.201701516R
Zipes DP, Wellens HJ (1998) Sudden cardiac death. Circulation 98:2334–2351
Acknowledgments
This work was partially supported by the National Institute of Health (R01s HL97979 and HL133294), and the American Heart Association (19TPA34900003) to LHX. The authors thank Qinyu Guo and Mengqing Liu for their work on reference collection.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the special issue on Calcium Signal Dynamics in Cardiac Myocytes and Fibroblasts: Mechanisms in Pflügers Archiv—European Journal of Physiology
Rights and permissions
About this article
Cite this article
Xie, LH., Gwathmey, J.K. & Zhao, Z. Cardiac adaptation and cardioprotection against arrhythmias and ischemia-reperfusion injury in mammalian hibernators. Pflugers Arch - Eur J Physiol 473, 407–416 (2021). https://doi.org/10.1007/s00424-020-02511-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-020-02511-0