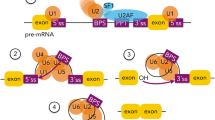
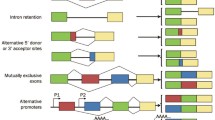
Abstract
Alternative splicing (AS) of protein-coding messenger RNAs is an essential regulatory mechanism in eukaryotic gene expression that controls the proper function of proteins. It is also implicated in the physiological regulation of mitochondria and various ion channels. Considering that mis-splicing can result in various human diseases by modifying or abrogating important physiological protein functions, a fine-tuned balance of AS is essential for human health. Accumulated data highlight the importance of alternatively spliced isoforms in various diseases, including neurodegenerative disorders, cancer, immune and infectious diseases, cardiovascular diseases, and metabolic conditions. However, basic understanding of disease mechanisms and development of clinical applications still require the integration and interpretation of physiological roles of AS. This review discusses the roles of AS in health and various diseases, while highlighting potential AS-targeting therapeutic applications.






Similar content being viewed by others
References
Alieva A, Shadrina MI, Filatova EV, Karabanov AV, Illarioshkin SN, Limborska SA, Slominsky PA (2014) Involvement of endocytosis and alternative splicing in the formation of the pathological process in the early stages of Parkinson's disease. Biomed Res Int 2014:718732. https://doi.org/10.1155/2014/718732
Araki S, Dairiki R, Nakayama Y, Murai A, Miyashita R, Iwatani M, Nomura T, Nakanishi O (2015) Inhibitors of CLK protein kinases suppress cell growth and induce apoptosis by modulating pre-mRNA splicing. PLoS One 10:e0116929. https://doi.org/10.1371/journal.pone.0116929
Arimura T, Inagaki N, Hayashi T, Shichi D, Sato A, Hinohara K, Vatta M, Towbin JA, Chikamori T, Yamashina A, Kimura A (2009) Impaired binding of ZASP/Cypher with phosphoglucomutase 1 is associated with dilated cardiomyopathy. Cardiovasc Res 83:80–88. https://doi.org/10.1093/cvr/cvp119
Athyros VG, Kakafika AI, Tziomalos K, Karagiannis A, Mikhailidis DP (2008) Antisense technology for the prevention or the treatment of cardiovascular disease: the next blockbuster? Expert Opin Investig Drugs 17:969–972. https://doi.org/10.1517/13543784.17.7.969
Ayoubi TA, Van De Ven WJ (1996) Regulation of gene expression by alternative promoters. FASEB J 10:453–460
Baralle FE, Giudice J (2017) Alternative splicing as a regulator of development and tissue identity. Nat Rev Mol Cell Biol 18:437–451. https://doi.org/10.1038/nrm.2017.27
Brauch KM, Karst ML, Herron KJ, de Andrade M, Pellikka PA, Rodeheffer RJ, Michels VV, Olson TM (2009) Mutations in ribonucleic acid binding protein gene cause familial dilated cardiomyopathy. J Am Coll Cardiol 54:930–941. https://doi.org/10.1016/j.jacc.2009.05.038
Caceres JF, Kornblihtt AR (2002) Alternative splicing: multiple control mechanisms and involvement in human disease. Trends Genet 18:186–193
Carmeliet P, Ng YS, Nuyens D, Theilmeier G, Brusselmans K, Cornelissen I, Ehler E, Kakkar VV, Stalmans I, Mattot V, Perriard JC, Dewerchin M, Flameng W, Nagy A, Lupu F, Moons L, Collen D, D'Amore PA, Shima DT (1999) Impaired myocardial angiogenesis and ischemic cardiomyopathy in mice lacking the vascular endothelial growth factor isoforms VEGF164 and VEGF188. Nat Med 5:495–502. https://doi.org/10.1038/8379
Castanotto D, Stein CA (2014) Antisense oligonucleotides in cancer. Curr Opin Oncol 26:584–589. https://doi.org/10.1097/CCO.0000000000000127
Chang JS, Fernand V, Zhang Y, Shin J, Jun HJ, Joshi Y, Gettys TW (2012) NT-PGC-1alpha protein is sufficient to link beta3-adrenergic receptor activation to transcriptional and physiological components of adaptive thermogenesis. J Biol Chem 287:9100–9111. https://doi.org/10.1074/jbc.M111.320200
Chang JS, Ha K (2017) An unexpected role for the transcriptional coactivator isoform NT-PGC-1alpha in the regulation of mitochondrial respiration in brown adipocytes. J Biol Chem 292:9958–9966. https://doi.org/10.1074/jbc.M117.778373
Chang YF, Imam JS, Wilkinson MF (2007) The nonsense-mediated decay RNA surveillance pathway. Annu Rev Biochem 76:51–74. https://doi.org/10.1146/annurev.biochem.76.050106.093909
Chen L, Zhao M, Li J, Wang Y, Bao Q, Wu S, Deng X, Tang X, Wu W, Liu X (2017) Critical role of X-box binding protein 1 in NADPH oxidase 4-triggered cardiac hypertrophy is mediated by receptor interacting protein kinase 1. Cell Cycle 16:348–359. https://doi.org/10.1080/15384101.2016.1260210
Chiocco MJ, Zhu X, Walther D, Pletnikova O, Troncoso JC, Uhl GR, Liu QR (2010) Fine mapping of calcineurin (PPP3CA) gene reveals novel alternative splicing patterns, association of 5'UTR trinucleotide repeat with addiction vulnerability, and differential isoform expression in Alzheimer's disease. Subst Use Misuse 45:1809–1826. https://doi.org/10.3109/10826084.2010.482449
Chow LT, Gelinas RE, Broker TR, Roberts RJ (1977) An amazing sequence arrangement at the 5′ ends of adenovirus 2 messenger RNA. Cell 12:1–8. https://doi.org/10.1016/0092-8674(77)90180-5
Cieply B, Carstens RP (2015) Functional roles of alternative splicing factors in human disease. Wiley Interdiscip Rev RNA 6:311–326. https://doi.org/10.1002/wrna.1276
Cooper TA (2005) Alternative splicing regulation impacts heart development. Cell 120:1–2. https://doi.org/10.1016/j.cell.2004.12.030
Cooper TA, Mattox W (1997) The regulation of splice-site selection, and its role in human disease. Am J Hum Genet 61:259–266. https://doi.org/10.1086/514856
Correia JC, Ferreira DM, Ruas JL (2015) Intercellular: local and systemic actions of skeletal muscle PGC-1s. Trends Endocrinol Metab 26:305–314. https://doi.org/10.1016/j.tem.2015.03.010
Cunha SR, Le Scouarnec S, Schott JJ, Mohler PJ (2008) Exon organization and novel alternative splicing of the human ANK2 gene: implications for cardiac function and human cardiac disease. J Mol Cell Cardiol 45:724–734. https://doi.org/10.1016/j.yjmcc.2008.08.005
Dahal LN, Basu N, Youssef H, Khanolkar RC, Barker RN, Erwig LP, Ward FJ (2016) Immunoregulatory soluble CTLA-4 modifies effector T-cell responses in systemic lupus erythematosus. Arthritis Res Ther 18:180. https://doi.org/10.1186/s13075-016-1075-1
Dally S, Bredoux R, Corvazier E, Andersen JP, Clausen JD, Dode L, Fanchaouy M, Gelebart P, Monceau V, Del Monte F, Gwathmey JK, Hajjar R, Chaabane C, Bobe R, Raies A, Enouf J (2006) Ca2+-ATPases in non-failing and failing heart: evidence for a novel cardiac sarco/endoplasmic reticulum Ca2+-ATPase 2 isoform (SERCA2c). Biochem J 395:249–258. https://doi.org/10.1042/BJ20051427
Daniels L, Bell JR, Delbridge LM, McDonald FJ, Lamberts RR, Erickson JR (2015) The role of CaMKII in diabetic heart dysfunction. Heart Fail Rev 20:589–600. https://doi.org/10.1007/s10741-015-9498-3
Dehghanian F, Hojati Z, Kay M (2014) New insights into VEGF-A alternative splicing: key regulatory switching in the pathological process. Avicenna J Med Biotechnol 6:192–199
Dever SM, Rodriguez M, El-Hage N (2016) Beta-adrenergic receptor gene expression in HIV-associated neurocognitive impairment and encephalitis: implications for MOR-1K subcellular localization. J Neuro-Oncol 22:866–870. https://doi.org/10.1007/s13365-016-0464-1
Ding JH, Xu X, Yang D, Chu PH, Dalton ND, Ye Z, Yeakley JM, Cheng H, Xiao RP, Ross J, Chen J, Fu XD (2004) Dilated cardiomyopathy caused by tissue-specific ablation of SC35 in the heart. EMBO J 23:885–896. https://doi.org/10.1038/sj.emboj.7600054
Dlamini Z, Mokoena F, Hull R (2017) Abnormalities in alternative splicing in diabetes: therapeutic targets. J Mol Endocrinol 59:R93–R107. https://doi.org/10.1530/JME-17-0049
Dredge BK, Polydorides AD, Darnell RB (2001) The splice of life: alternative splicing and neurological disease. Nat Rev Neurosci 2:43–50. https://doi.org/10.1038/35049061
Dunning CJ, McKenzie M, Sugiana C, Lazarou M, Silke J, Connelly A, Fletcher JM, Kirby DM, Thorburn DR, Ryan MT (2007) Human CIA30 is involved in the early assembly of mitochondrial complex I and mutations in its gene cause disease. EMBO J 26:3227–3237. https://doi.org/10.1038/sj.emboj.7601748
Elliott DA, Kim WS, Gorissen S, Halliday GM, Kwok JB (2012) Leucine-rich repeat kinase 2 and alternative splicing in Parkinson's disease. Mov Disord 27:1004–1011. https://doi.org/10.1002/mds.25005
Escribano O, Beneit N, Rubio-Longas C, Lopez-Pastor AR, Gomez-Hernandez A (2017) The role of insulin receptor isoforms in diabetes and its metabolic and vascular complications. J Diabetes Res 2017:1403206. https://doi.org/10.1155/2017/1403206
Eskens FA, Ramos FJ, Burger H, O'Brien JP, Piera A, de Jonge MJ, Mizui Y, Wiemer EA, Carreras MJ, Baselga J, Tabernero J (2013) Phase I pharmacokinetic and pharmacodynamic study of the first-in-class spliceosome inhibitor E7107 in patients with advanced solid tumors. Clin Cancer Res 19:6296–6304. https://doi.org/10.1158/1078-0432.CCR-13-0485
Felkin LE, Narita T, Germack R, Shintani Y, Takahashi K, Sarathchandra P, Lopez-Olaneta MM, Gomez-Salinero JM, Suzuki K, Barton PJ, Rosenthal N, Lara-Pezzi E (2011) Calcineurin splicing variant calcineurin Abeta1 improves cardiac function after myocardial infarction without inducing hypertrophy. Circulation 123:2838–2847. https://doi.org/10.1161/CIRCULATIONAHA.110.012211
Fernandez-Nogales M, Santos-Galindo M, Hernandez IH, Cabrera JR, Lucas JJ (2016) Faulty splicing and cytoskeleton abnormalities in Huntington's disease. Brain Pathol 26:772–778. https://doi.org/10.1111/bpa.12430
Freyermuth F, Rau F, Kokunai Y, Linke T, Sellier C, Nakamori M, Kino Y, Arandel L, Jollet A, Thibault C, Philipps M, Vicaire S, Jost B, Udd B, Day JW, Duboc D, Wahbi K, Matsumura T, Fujimura H, Mochizuki H, Deryckere F, Kimura T, Nukina N, Ishiura S, Lacroix V, Campan-Fournier A, Navratil V, Chautard E, Auboeuf D, Horie M, Imoto K, Lee KY, Swanson MS, Lopez de Munain A, Inada S, Itoh H, Nakazawa K, Ashihara T, Wang E, Zimmer T, Furling D, Takahashi MP, Charlet-Berguerand N (2016) Splicing misregulation of SCN5A contributes to cardiac-conduction delay and heart arrhythmia in myotonic dystrophy. Nat Commun 7:11067. https://doi.org/10.1038/ncomms11067
Furihata C (2015) An active alternative splicing isoform of human mitochondrial 8-oxoguanine DNA glycosylase (OGG1). Genes Environ 37:21. https://doi.org/10.1186/s41021-015-0021-9
Gao G, Dudley SC Jr (2013) SCN5A splicing variants and the possibility of predicting heart failure-associated arrhythmia. Expert Rev Cardiovasc Ther 11:117–119. https://doi.org/10.1586/erc.12.180
Gao G, Xie A, Huang SC, Zhou A, Zhang J, Herman AM, Ghassemzadeh S, Jeong EM, Kasturirangan S, Raicu M, Sobieski MA 2nd, Bhat G, Tatooles A, Benz EJ Jr, Kamp TJ, Dudley SC Jr (2011) Role of RBM25/LUC7L3 in abnormal cardiac sodium channel splicing regulation in human heart failure. Circulation 124:1124–1131. https://doi.org/10.1161/CIRCULATIONAHA.111.044495
Garcia-Blanco MA, Baraniak AP, Lasda EL (2004) Alternative splicing in disease and therapy. Nat Biotechnol 22:535–546. https://doi.org/10.1038/nbt964
Gelebart P, Martin V, Enouf J, Papp B (2003) Identification of a new SERCA2 splice variant regulated during monocytic differentiation. Biochem Biophys Res Commun 303:676–684
George CH, Rogers SA, Bertrand BM, Tunwell RE, Thomas NL, Steele DS, Cox EV, Pepper C, Hazeel CJ, Claycomb WC, Lai FA (2007) Alternative splicing of ryanodine receptors modulates cardiomyocyte Ca2+ signaling and susceptibility to apoptosis. Circ Res 100:874–883. https://doi.org/10.1161/01.RES.0000260804.77807.cf
Giudice J, Xia Z, Wang ET, Scavuzzo MA, Ward AJ, Kalsotra A, Wang W, Wehrens XH, Burge CB, Li W, Cooper TA (2014) Alternative splicing regulates vesicular trafficking genes in cardiomyocytes during postnatal heart development. Nat Commun 5:3603. https://doi.org/10.1038/ncomms4603
Glatz DC, Rujescu D, Tang Y, Berendt FJ, Hartmann AM, Faltraco F, Rosenberg C, Hulette C, Jellinger K, Hampel H, Riederer P, Moller HJ, Andreadis A, Henkel K, Stamm S (2006) The alternative splicing of tau exon 10 and its regulatory proteins CLK2 and TRA2-BETA1 changes in sporadic Alzheimer's disease. J Neurochem 96:635–644. https://doi.org/10.1111/j.1471-4159.2005.03552.x
Graveley BR (2001) Alternative splicing: increasing diversity in the proteomic world. Trends Genet 17:100–107
Gray CB, Suetomi T, Xiang S, Mishra S, Blackwood EA, Glembotski CC, Miyamoto S, Westenbrink BD, Brown JH (2017) CaMKIIdelta subtypes differentially regulate infarct formation following ex vivo myocardial ischemia/reperfusion through NF-kappaB and TNF-alpha. J Mol Cell Cardiol 103:48–55. https://doi.org/10.1016/j.yjmcc.2017.01.002
Gray P, Michelsen KS, Sirois CM, Lowe E, Shimada K, Crother TR, Chen S, Brikos C, Bulut Y, Latz E, Underhill D, Arditi M (2010) Identification of a novel human MD-2 splice variant that negatively regulates lipopolysaccharide-induced TLR4 signaling. J Immunol 184:6359–6366. https://doi.org/10.4049/jimmunol.0903543
Greene AL, Lalli MJ, Ji Y, Babu GJ, Grupp I, Sussman M, Periasamy M (2000) Overexpression of SERCA2b in the heart leads to an increase in sarcoplasmic reticulum calcium transport function and increased cardiac contractility. J Biol Chem 275:24722–24727. https://doi.org/10.1074/jbc.M001783200
Gu Q, Jin N, Sheng H, Yin X, Zhu J (2011) Cyclic AMP-dependent protein kinase A regulates the alternative splicing of CaMKIIdelta. PLoS One 6:e25745. https://doi.org/10.1371/journal.pone.0025745
Guantes R, Rastrojo A, Neves R, Lima A, Aguado B, Iborra FJ (2015) Global variability in gene expression and alternative splicing is modulated by mitochondrial content. Genome Res 25:633–644. https://doi.org/10.1101/gr.178426.114
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674. https://doi.org/10.1016/j.cell.2011.02.013
Hartmann T, Bergsdorf C, Sandbrink R, Tienari PJ, Multhaup G, Ida N, Bieger S, Dyrks T, Weidemann A, Masters CL, Beyreuther K (1996) Alzheimer's disease betaA4 protein release and amyloid precursor protein sorting are regulated by alternative splicing. J Biol Chem 271:13208–13214
Hayakawa M, Sakashita E, Ueno E, Tominaga S, Hamamoto T, Kagawa Y, Endo H (2002) Muscle-specific exonic splicing silencer for exon exclusion in human ATP synthase gamma-subunit pre-mRNA. J Biol Chem 277:6974–6984. https://doi.org/10.1074/jbc.M110138200
Heide J, Zhang F, Bigos KL, Mann SA, Carr VJ, Shannon Weickert C, Green MJ, Weinberger DR, Vandenberg JI (2016) Differential response to risperidone in schizophrenia patients by KCNH2 genotype and drug metabolizer status. Am J Psychiatry 173:53–59. https://doi.org/10.1176/appi.ajp.2015.14050653
Heinzen EL, Yoon W, Weale ME, Sen A, Wood NW, Burke JR, Welsh-Bohmer KA, Hulette CM, Sisodiya SM, Goldstein DB (2007) Alternative ion channel splicing in mesial temporal lobe epilepsy and Alzheimer's disease. Genome Biol 8:R32. https://doi.org/10.1186/gb-2007-8-3-r32
Hinrich AJ, Jodelka FM, Chang JL, Brutman D, Bruno AM, Briggs CA, James BD, Stutzmann GE, Bennett DA, Miller SA, Rigo F, Marr RA, Hastings ML (2016) Therapeutic correction of ApoER2 splicing in Alzheimer's disease mice using antisense oligonucleotides. EMBO Mol Med 8:328–345. https://doi.org/10.15252/emmm.201505846
Hong DS, Kurzrock R, Naing A, Wheler JJ, Falchook GS, Schiffman JS, Faulkner N, Pilat MJ, O'Brien J, LoRusso P (2014) A phase I, open-label, single-arm, dose-escalation study of E7107, a precursor messenger ribonucleic acid (pre-mRNA) splicesome inhibitor administered intravenously on days 1 and 8 every 21 days to patients with solid tumors. Investig New Drugs 32:436–444. https://doi.org/10.1007/s10637-013-0046-5
Hsu TY, Simon LM, Neill NJ, Marcotte R, Sayad A, Bland CS, Echeverria GV, Sun T, Kurley SJ, Tyagi S, Karlin KL, Dominguez-Vidana R, Hartman JD, Renwick A, Scorsone K, Bernardi RJ, Skinner SO, Jain A, Orellana M, Lagisetti C, Golding I, Jung SY, Neilson JR, Zhang XH, Cooper TA, Webb TR, Neel BG, Shaw CA, Westbrook TF (2015) The spliceosome is a therapeutic vulnerability in MYC-driven cancer. Nature 525:384–388. https://doi.org/10.1038/nature14985
Hughes T, Hansson L, Sonderby IE, Athanasiu L, Zuber V, Tesli M, Song J, Hultman CM, Bergen SE, Landen M, Melle I, Andreassen OA, Djurovic S (2016) A loss-of-function variant in a minor isoform of ANK3 protects against bipolar disorder and schizophrenia. Biol Psychiatry 80:323–330. https://doi.org/10.1016/j.biopsych.2015.09.021
Hurtado C, Prociuk M, Maddaford TG, Dibrov E, Mesaeli N, Hryshko LV, Pierce GN (2006) Cells expressing unique Na+/Ca2+ exchange (NCX1) splice variants exhibit different susceptibilities to Ca2+ overload. Am J Physiol Heart Circ Physiol 290:H2155–H2162. https://doi.org/10.1152/ajpheart.00958.2005
Ingelsson M, Ramasamy K, Cantuti-Castelvetri I, Skoglund L, Matsui T, Orne J, Kowa H, Raju S, Vanderburg CR, Augustinack JC, de Silva R, Lees AJ, Lannfelt L, Growdon JH, Frosch MP, Standaert DG, Irizarry MC, Hyman BT (2006) No alteration in tau exon 10 alternative splicing in tangle-bearing neurons of the Alzheimer's disease brain. Acta Neuropathol 112:439–449. https://doi.org/10.1007/s00401-006-0095-3
Ishunina TA, Swaab DF (2012) Decreased alternative splicing of estrogen receptor-alpha mRNA in the Alzheimer's disease brain. Neurobiol Aging 33:286–296.e283. https://doi.org/10.1016/j.neurobiolaging.2010.03.010
Jensen DP, Urhammer SA, Eiberg H, Borch-Johnsen K, Jorgensen T, Hansen T, Pedersen O (2006) Variation in CAPN10 in relation to type 2 diabetes, obesity and quantitative metabolic traits: studies in 6018 whites. Mol Genet Metab 89:360–367. https://doi.org/10.1016/j.ymgme.2006.06.003
Juan-Mateu J, Rech TH, Villate O, Lizarraga-Mollinedo E, Wendt A, Turatsinze JV, Brondani LA, Nardelli TR, Nogueira TC, Esguerra JL, Alvelos MI, Marchetti P, Eliasson L, Eizirik DL (2017) Neuron-enriched RNA-binding proteins regulate pancreatic beta cell function and survival. J Biol Chem 292:3466–3480. https://doi.org/10.1074/jbc.M116.748335
Jun HJ, Gettys TW, Chang JS (2012) Transcriptional activity of PGC-1alpha and NT-PGC-1alpha is differentially regulated by Twist-1 in brown fat metabolism. PPAR Res 2012:320454. https://doi.org/10.1155/2012/320454
Kaida D, Motoyoshi H, Tashiro E, Nojima T, Hagiwara M, Ishigami K, Watanabe H, Kitahara T, Yoshida T, Nakajima H, Tani T, Horinouchi S, Yoshida M (2007) Spliceostatin A targets SF3b and inhibits both splicing and nuclear retention of pre-mRNA. Nat Chem Biol 3:576–583. https://doi.org/10.1038/nchembio.2007.18
Kalivendi SV, Yedlapudi D, Hillard CJ, Kalyanaraman B (2010) Oxidants induce alternative splicing of alpha-synuclein: implications for Parkinson's disease. Free Radic Biol Med 48:377–383. https://doi.org/10.1016/j.freeradbiomed.2009.10.045
Kalsotra A, Xiao X, Ward AJ, Castle JC, Johnson JM, Burge CB, Cooper TA (2008) A postnatal switch of CELF and MBNL proteins reprograms alternative splicing in the developing heart. Proc Natl Acad Sci U S A 105:20333–20338. https://doi.org/10.1073/pnas.0809045105
Karambataki M, Malousi A, Tzimagiorgis G, Haitoglou C, Fragou A, Georgiou E, Papadopoulou F, Krassas GE, Kouidou S (2017) Association of two synonymous splicing-associated CpG single nucleotide polymorphisms in calpain 10 and solute carrier family 2 member 2 with type 2 diabetes. Biomed Rep 6:146–158. https://doi.org/10.3892/br.2016.833
Keren H, Lev-Maor G, Ast G (2010) Alternative splicing and evolution: diversification, exon definition and function. Nat Rev Genet 11:345–355. https://doi.org/10.1038/nrg2776
Kim E, Goren A, Ast G (2008) Alternative splicing and disease. RNA Biol 5:17–19
Kim HK, Nilius B, Kim N, Ko KS, Rhee BD, Han J (2016) Cardiac response to oxidative stress induced by mitochondrial dysfunction. Rev Physiol Biochem Pharmacol 170:101–127. https://doi.org/10.1007/112_2015_5004
Kobylecki C, Crossman AR, Ravenscroft P (2013) Alternative splicing of AMPA receptor subunits in the 6-OHDA-lesioned rat model of Parkinson's disease and L-DOPA-induced dyskinesia. Exp Neurol 247:476–484. https://doi.org/10.1016/j.expneurol.2013.01.019
Kong SW, Hu YW, Ho JW, Ikeda S, Polster S, John R, Hall JL, Bisping E, Pieske B, dos Remedios CG, Pu WT (2010) Heart failure-associated changes in RNA splicing of sarcomere genes. Circ Cardiovasc Genet 3:138–146. https://doi.org/10.1161/CIRCGENETICS.109.904698
Kotake Y, Sagane K, Owa T, Mimori-Kiyosue Y, Shimizu H, Uesugi M, Ishihama Y, Iwata M, Mizui Y (2007) Splicing factor SF3b as a target of the antitumor natural product pladienolide. Nat Chem Biol 3:570–575. https://doi.org/10.1038/nchembio.2007.16
Lai MK, Esiri MM, Tan MG (2014) Genome-wide profiling of alternative splicing in Alzheimer's disease. Genom Data 2:290–292. https://doi.org/10.1016/j.gdata.2014.09.002
Lara-Pezzi E, Gomez-Salinero J, Gatto A, Garcia-Pavia P (2013) The alternative heart: impact of alternative splicing in heart disease. J Cardiovasc Transl Res 6:945–955. https://doi.org/10.1007/s12265-013-9482-z
Lee C, Low CY, Francis PT, Attems J, Wong PT, Lai MK, Tan MG (2016) An isoform-specific role of FynT tyrosine kinase in Alzheimer's disease. J Neurochem 136:637–650. https://doi.org/10.1111/jnc.13429
Lee C, Low CY, Wong SY, Lai MK, Tan MG (2017) Selective induction of alternatively spliced FynT isoform by TNF facilitates persistent inflammatory responses in astrocytes. Sci Rep 7:43651. https://doi.org/10.1038/srep43651
Lee SC, Abdel-Wahab O (2016) Therapeutic targeting of splicing in cancer. Nat Med 22:976–986. https://doi.org/10.1038/nm.4165
Li G, Wang J, Liao P, Bartels P, Zhang H, Yu D, Liang MC, Poh KK, Yu CY, Jiang F, Yong TF, Wong YP, Hu Z, Huang H, Zhang G, Galupo MJ, Bian JS, Ponniah S, Trasti SL, See K, Foo R, Hoppe UC, Herzig S, Soong TW (2017) Exclusion of alternative exon 33 of CaV1.2 calcium channels in heart is proarrhythmogenic. Proc Natl Acad Sci U S A 114:E4288–E4295. https://doi.org/10.1073/pnas.1617205114
Liao P, Soong TW (2010) Understanding alternative splicing of Cav1.2 calcium channels for a new approach towards individualized medicine. J Biomed Res 24:181–186. https://doi.org/10.1016/S1674-8301(10)60027-9
Liao P, Yong TF, Liang MC, Yue DT, Soong TW (2005) Splicing for alternative structures of Cav1.2 Ca2+ channels in cardiac and smooth muscles. Cardiovasc Res 68:197–203. https://doi.org/10.1016/j.cardiores.2005.06.024
Lin Y, Tan KT, Liu J, Kong X, Huang Z, Xu XQ (2017) Global profiling of Rbm24 bound RNAs uncovers a multi-tasking RNA binding protein. Int J Biochem Cell Biol 94:10–21. https://doi.org/10.1016/j.biocel.2017.11.002
Lin Z, Xu W, Li C, Wang Y, Yang L, Zou B, Gao S, Yao W, Song Z, Liu G (2017) Beta-8-oxoguanine DNA glycosylase overexpression reduces oxidative stress-induced mitochondrial dysfunction and apoptosis through the JNK signaling pathway in human bronchial epithelial cells. DNA Cell Biol 36:1071–1080. https://doi.org/10.1089/dna.2017.3769
Liu S, Zhao K, Su X, Lu L, Zhao H, Zhang X, Wang Y, Wu C, Chen J, Zhou Y, Hu X, Wang Y, Lu M, Chen X, Pei R (2017) MITA/STING and its alternative splicing isoform MRP restrict hepatitis B virus replication. PLoS One 12:e0169701. https://doi.org/10.1371/journal.pone.0169701
Lopez-Olaneta MM, Villalba M, Gomez-Salinero JM, Jimenez-Borreguero LJ, Breckenridge R, Ortiz-Sanchez P, Garcia-Pavia P, Ibanez B, Lara-Pezzi E (2014) Induction of the calcineurin variant CnAbeta1 after myocardial infarction reduces post-infarction ventricular remodelling by promoting infarct vascularization. Cardiovasc Res 102:396–406. https://doi.org/10.1093/cvr/cvu068
Love JE, Hayden EJ, Rohn TT (2015) Alternative splicing in Alzheimer's disease. J Parkinsons Dis Alzheimers Dis 2. https://doi.org/10.13188/2376-922X.1000010
Luzina IG, Lockatell V, Lavania S, Pickering EM, Kang PH, Bashkatova YN, Andreev SM, Atamas SP (2012) Natural production and functional effects of alternatively spliced interleukin-4 protein in asthma. Cytokine 58:20–26. https://doi.org/10.1016/j.cyto.2011.12.017
Ma J, Lu L, Guo W, Ren J, Yang J (2016) Emerging role for RBM20 and its splicing substrates in cardiac function and heart failure. Curr Pharm Des 22:4744–4751
Makarenko I, Opitz CA, Leake MC, Neagoe C, Kulke M, Gwathmey JK, del Monte F, Hajjar RJ, Linke WA (2004) Passive stiffness changes caused by upregulation of compliant titin isoforms in human dilated cardiomyopathy hearts. Circ Res 95:708–716. https://doi.org/10.1161/01.RES.0000143901.37063.2f
Malakar P, Chartarifsky L, Hija A, Leibowitz G, Glaser B, Dor Y, Karni R (2016) Insulin receptor alternative splicing is regulated by insulin signaling and modulates beta cell survival. Sci Rep 6:31222. https://doi.org/10.1038/srep31222
Maracchioni A, Totaro A, Angelini DF, Di Penta A, Bernardi G, Carri MT, Achsel T (2007) Mitochondrial damage modulates alternative splicing in neuronal cells: implications for neurodegeneration. J Neurochem 100:142–153. https://doi.org/10.1111/j.1471-4159.2006.04204.x
Mardon HJ, Sebastio G, Baralle FE (1987) A role for exon sequences in alternative splicing of the human fibronectin gene. Nucleic Acids Res 15:7725–7733
Martin D, Li Y, Yang J, Wang G, Margariti A, Jiang Z, Yu H, Zampetaki A, Hu Y, Xu Q, Zeng L (2014) Unspliced X-box-binding protein 1 (XBP1) protects endothelial cells from oxidative stress through interaction with histone deacetylase 3. J Biol Chem 289:30625–30634. https://doi.org/10.1074/jbc.M114.571984
Martinez-Redondo V, Jannig PR, Correia JC, Ferreira DM, Cervenka I, Lindvall JM, Sinha I, Izadi M, Pettersson-Klein AT, Agudelo LZ, Gimenez-Cassina A, Brum PC, Dahlman-Wright K, Ruas JL (2016) Peroxisome proliferator-activated receptor gamma coactivator-1 alpha isoforms selectively regulate multiple splicing events on target genes. J Biol Chem 291:15169–15184. https://doi.org/10.1074/jbc.M115.705822
Martinez-Redondo V, Pettersson AT, Ruas JL (2015) The hitchhiker's guide to PGC-1alpha isoform structure and biological functions. Diabetologia 58:1969–1977. https://doi.org/10.1007/s00125-015-3671-z
Massone S, Vassallo I, Fiorino G, Castelnuovo M, Barbieri F, Borghi R, Tabaton M, Robello M, Gatta E, Russo C, Florio T, Dieci G, Cancedda R, Pagano A (2011) 17A, a novel non-coding RNA, regulates GABA B alternative splicing and signaling in response to inflammatory stimuli and in Alzheimer disease. Neurobiol Dis 41:308–317. https://doi.org/10.1016/j.nbd.2010.09.019
McFadden JP, Baker BS, Powles AV, Fry L (2010) Psoriasis and extra domain A fibronectin loops. Br J Dermatol 163:5–11. https://doi.org/10.1111/j.1365-2133.2010.09737.x
Michel LY, Verkaart S, Koopman WJ, Willems PH, Hoenderop JG, Bindels RJ (2014) Function and regulation of the Na+-Ca2+ exchanger NCX3 splice variants in brain and skeletal muscle. J Biol Chem 289:11293–11303. https://doi.org/10.1074/jbc.M113.529388
Mills JD, Sheahan PJ, Lai D, Kril JJ, Janitz M, Sutherland GT (2014) The alternative splicing of the apolipoprotein E gene is unperturbed in the brains of Alzheimer's disease patients. Mol Biol Rep 41:6365–6376. https://doi.org/10.1007/s11033-014-3516-8
Miura S, Kai Y, Kamei Y, Ezaki O (2008) Isoform-specific increases in murine skeletal muscle peroxisome proliferator-activated receptor-gamma coactivator-1alpha (PGC-1alpha) mRNA in response to beta2-adrenergic receptor activation and exercise. Endocrinology 149:4527–4533. https://doi.org/10.1210/en.2008-0466
Moore MJ, Wang Q, Kennedy CJ, Silver PA (2010) An alternative splicing network links cell-cycle control to apoptosis. Cell 142:625–636. https://doi.org/10.1016/j.cell.2010.07.019
Morikawa T, Manabe T, Ito Y, Yamada S, Yoshimi A, Nagai T, Ozaki N, Mayeda A (2010) The expression of HMGA1a is increased in lymphoblastoid cell lines from schizophrenia patients. Neurochem Int 56:736–739. https://doi.org/10.1016/j.neuint.2010.03.011
Mourich DV, Oda SK, Schnell FJ, Crumley SL, Hauck LL, Moentenich CA, Marshall NB, Hinrichs DJ, Iversen PL (2014) Alternative splice forms of CTLA-4 induced by antisense mediated splice-switching influences autoimmune diabetes susceptibility in NOD mice. Nucleic Acid Ther 24:114–126. https://doi.org/10.1089/nat.2013.0449
Mueckler M, Thorens B (2013) The SLC2 (GLUT) family of membrane transporters. Mol Asp Med 34:121–138. https://doi.org/10.1016/j.mam.2012.07.001
Murray A, Donger C, Fenske C, Spillman I, Richard P, Dong YB, Neyroud N, Chevalier P, Denjoy I, Carter N, Syrris P, Afzal AR, Patton MA, Guicheney P, Jeffery S (1999) Splicing mutations in KCNQ1: a mutation hot spot at codon 344 that produces in frame transcripts. Circulation 100:1077–1084
Musaro A, McCullagh K, Paul A, Houghton L, Dobrowolny G, Molinaro M, Barton ER, Sweeney HL, Rosenthal N (2001) Localized Igf-1 transgene expression sustains hypertrophy and regeneration in senescent skeletal muscle. Nat Genet 27:195–200. https://doi.org/10.1038/84839
Nutter CA, Jaworski EA, Verma SK, Deshmukh V, Wang Q, Botvinnik OB, Lozano MJ, Abass IJ, Ijaz T, Brasier AR, Garg NJ, Wehrens XHT, Yeo GW, Kuyumcu-Martinez MN (2016) Dysregulation of RBFOX2 is an early event in cardiac pathogenesis of diabetes. Cell Rep 15:2200–2213. https://doi.org/10.1016/j.celrep.2016.05.002
Ohno K, Rahman MA, Nazim M, Nasrin F, Lin Y, Takeda JI, Masuda A (2017) Splicing regulation and dysregulation of cholinergic genes expressed at the neuromuscular junction. J Neurochem. https://doi.org/10.1111/jnc.13954
Orengo JP, Cooper TA (2007) Alternative splicing in disease. Adv Exp Med Biol 623:212–223
Organization WH (2016) Global report on diabetes. World Health Organization
Panelli D, Lorusso F, Papa F, Sardanell A, Papa S (2013) Alternative splicing and nonsense mediated decay in mitochondrial complex-I biogenesis and its implication in human diseases. J Bioanal Biomed s3. https://doi.org/10.4172/1948-593x.s3-006
Paronetto MP, Passacantilli I, Sette C (2016) Alternative splicing and cell survival: from tissue homeostasis to disease. Cell Death Differ 23:1919–1929. https://doi.org/10.1038/cdd.2016.91
Phillips MI, Costales J, Lee RJ, Oliveira E, Burns AB (2015) Antisense therapy for cardiovascular diseases. Curr Pharm Des 21:4417–4426
Poltorak Z, Cohen T, Neufeld G (2000) The VEGF splice variants: properties, receptors, and usage for the treatment of ischemic diseases. Herz 25:126–129
Poon KL, Tan KT, Wei YY, Ng CP, Colman A, Korzh V, Xu XQ (2012) RNA-binding protein RBM24 is required for sarcomere assembly and heart contractility. Cardiovasc Res 94:418–427. https://doi.org/10.1093/cvr/cvs095
Popov DV, Bachinin AV, Lysenko EA, Miller TF, Vinogradova OL (2014) Exercise-induced expression of peroxisome proliferator-activated receptor gamma coactivator-1alpha isoforms in skeletal muscle of endurance-trained males. J Physiol Sci 64:317–323. https://doi.org/10.1007/s12576-014-0321-z
Rajan S, Jagatheesan G, Karam CN, Alves ML, Bodi I, Schwartz A, Bulcao CF, D'Souza KM, Akhter SA, Boivin GP, Dube DK, Petrashevskaya N, Herr AB, Hullin R, Liggett SB, Wolska BM, Solaro RJ, Wieczorek DF (2010) Molecular and functional characterization of a novel cardiac-specific human tropomyosin isoform. Circulation 121:410–418. https://doi.org/10.1161/CIRCULATIONAHA.109.889725
Rockenstein EM, McConlogue L, Tan H, Power M, Masliah E, Mucke L (1995) Levels and alternative splicing of amyloid beta protein precursor (APP) transcripts in brains of APP transgenic mice and humans with Alzheimer's disease. J Biol Chem 270:28257–28267
Rogaev EI, Sherrington R, Wu C, Levesque G, Liang Y, Rogaeva EA, Ikeda M, Holman K, Lin C, Lukiw WJ, de Jong PJ, Fraser PE, Rommens JM, St George-Hyslop P (1997) Analysis of the 5′ sequence, genomic structure, and alternative splicing of the presenilin-1 gene (PSEN1) associated with early onset Alzheimer disease. Genomics 40:415–424. https://doi.org/10.1006/geno.1996.4523
Roybal GA, Jurica MS (2010) Spliceostatin A inhibits spliceosome assembly subsequent to prespliceosome formation. Nucleic Acids Res 38:6664–6672. https://doi.org/10.1093/nar/gkq494
Ruas JL, White JP, Rao RR, Kleiner S, Brannan KT, Harrison BC, Greene NP, Wu J, Estall JL, Irving BA, Lanza IR, Rasbach KA, Okutsu M, Nair KS, Yan Z, Leinwand LA, Spiegelman BM (2012) A PGC-1alpha isoform induced by resistance training regulates skeletal muscle hypertrophy. Cell 151:1319–1331. https://doi.org/10.1016/j.cell.2012.10.050
Ryan M, Wong WC, Brown R, Akbani R, Su X, Broom B, Melott J, Weinstein J (2016) TCGASpliceSeq a compendium of alternative mRNA splicing in cancer. Nucleic Acids Res 44:D1018–D1022. https://doi.org/10.1093/nar/gkv1288
Salton M, Kasprzak WK, Voss T, Shapiro BA, Poulikakos PI, Misteli T (2015) Inhibition of vemurafenib-resistant melanoma by interference with pre-mRNA splicing. Nat Commun 6:7103. https://doi.org/10.1038/ncomms8103
Sammeth M, Foissac S, Guigo R (2008) A general definition and nomenclature for alternative splicing events. PLoS Comput Biol 4:e1000147. https://doi.org/10.1371/journal.pcbi.1000147
Sanchez-Caballero L, Ruzzenente B, Bianchi L, Assouline Z, Barcia G, Metodiev MD, Rio M, Funalot B, van den Brand MA, Guerrero-Castillo S, Molenaar JP, Koolen D, Brandt U, Rodenburg RJ, Nijtmans LG, Rotig A (2016) Mutations in Complex I assembly factor TMEM126B result in muscle weakness and isolated Complex I deficiency. Am J Hum Genet 99:208–216. https://doi.org/10.1016/j.ajhg.2016.05.022
Santini MP, Lexow J, Borsellino G, Slonimski E, Zarrinpashneh E, Poggioli T, Rosenthal N (2011) IGF-1Ea induces vessel formation after injury and mediates bone marrow and heart cross-talk through the expression of specific cytokines. Biochem Biophys Res Commun 410:201–207. https://doi.org/10.1016/j.bbrc.2011.05.081
Satoh N, Yokoyama C, Itamura N, Miyajima-Nakano Y, Hisatomi H (2015) Alternative splicing isoform in succinate dehydrogenase complex, subunit C causes downregulation of succinate-coenzyme Q oxidoreductase activity in mitochondria. Oncol Lett 9:330–334. https://doi.org/10.3892/ol.2014.2699
Sawada T, Minamino T, Fu HY, Asai M, Okuda K, Isomura T, Yamazaki S, Asano Y, Okada K, Tsukamoto O, Sanada S, Asanuma H, Asakura M, Takashima S, Kitakaze M, Komuro I (2010) X-box binding protein 1 regulates brain natriuretic peptide through a novel AP1/CRE-like element in cardiomyocytes. J Mol Cell Cardiol 48:1280–1289. https://doi.org/10.1016/j.yjmcc.2010.02.004
Scacco S, Petruzzella V, Budde S, Vergari R, Tamborra R, Panelli D, van den Heuvel LP, Smeitink JA, Papa S (2003) Pathological mutations of the human NDUFS4 gene of the 18-kDa (AQDQ) subunit of complex I affect the expression of the protein and the assembly and function of the complex. J Biol Chem 278:44161–44167. https://doi.org/10.1074/jbc.M307615200
Schoenauer R, Emmert MY, Felley A, Ehler E, Brokopp C, Weber B, Nemir M, Faggian GG, Pedrazzini T, Falk V, Hoerstrup SP, Agarkova I (2011) EH-myomesin splice isoform is a novel marker for dilated cardiomyopathy. Basic Res Cardiol 106:233–247. https://doi.org/10.1007/s00395-010-0131-2
Schommartz T, Loroch S, Alawi M, Grundhoff A, Sickmann A, Brune W (2016) Functional dissection of an alternatively spliced herpesvirus gene by splice site mutagenesis. J Virol 90:4626–4636. https://doi.org/10.1128/JVI.02987-15
Serena G, Yan S, Camhi S, Patel S, Lima RS, Sapone A, Leonard MM, Mukherjee R, Nath BJ, Lammers KM, Fasano A (2017) Proinflammatory cytokine interferon-gamma and microbiome-derived metabolites dictate epigenetic switch between forkhead box protein 3 isoforms in coeliac disease. Clin Exp Immunol 187:490–506. https://doi.org/10.1111/cei.12911
Shehadeh LA, Yu K, Wang L, Guevara A, Singer C, Vance J, Papapetropoulos S (2010) SRRM2, a potential blood biomarker revealing high alternative splicing in Parkinson's disease. PLoS One 5:e9104. https://doi.org/10.1371/journal.pone.0009104
Shi J, Qian W, Yin X, Iqbal K, Grundke-Iqbal I, Gu X, Ding F, Gong CX, Liu F (2011) Cyclic AMP-dependent protein kinase regulates the alternative splicing of tau exon 10: a mechanism involved in tau pathology of Alzheimer disease. J Biol Chem 286:14639–14648. https://doi.org/10.1074/jbc.M110.204453
Silvennoinen M, Ahtiainen JP, Hulmi JJ, Pekkala S, Taipale RS, Nindl BC, Laine T, Hakkinen K, Selanne H, Kyrolainen H, Kainulainen H (2015) PGC-1 isoforms and their target genes are expressed differently in human skeletal muscle following resistance and endurance exercise. Physiol Rep 3. https://doi.org/10.14814/phy2.12563
Smith CW, Valcarcel J (2000) Alternative pre-mRNA splicing: the logic of combinatorial control. Trends Biochem Sci 25:381–388
Smoot ME, Ono K, Ruscheinski J, Wang PL, Ideker T (2011) Cytoscape 2.8: new features for data integration and network visualization. Bioinformatics 27:431–432. https://doi.org/10.1093/bioinformatics/btq675
Soreq L, Bergman H, Israel Z, Soreq H (2012) Exon arrays reveal alternative splicing aberrations in Parkinson's disease leukocytes. Neurodegener Dis 10:203–206. https://doi.org/10.1159/000332598
Splawski I, Timothy KW, Decher N, Kumar P, Sachse FB, Beggs AH, Sanguinetti MC, Keating MT (2005) Severe arrhythmia disorder caused by cardiac L-type calcium channel mutations. Proc Natl Acad Sci U S A 102:8089–8096; discussion 8086-8088. https://doi.org/10.1073/pnas.0502506102
Splawski I, Timothy KW, Sharpe LM, Decher N, Kumar P, Bloise R, Napolitano C, Schwartz PJ, Joseph RM, Condouris K, Tager-Flusberg H, Priori SG, Sanguinetti MC, Keating MT (2004) Ca(V)1.2 calcium channel dysfunction causes a multisystem disorder including arrhythmia and autism. Cell 119:19–31. https://doi.org/10.1016/j.cell.2004.09.011
Sterne-Weiler T, Sanford JR (2014) Exon identity crisis: disease-causing mutations that disrupt the splicing code. Genome Biol 15:201. https://doi.org/10.1186/gb4150
Sveen A, Kilpinen S, Ruusulehto A, Lothe RA, Skotheim RI (2016) Aberrant RNA splicing in cancer; expression changes and driver mutations of splicing factor genes. Oncogene 35:2413–2427. https://doi.org/10.1038/onc.2015.318
Tadaishi M, Miura S, Kai Y, Kano Y, Oishi Y, Ezaki O (2011) Skeletal muscle-specific expression of PGC-1alpha-b, an exercise-responsive isoform, increases exercise capacity and peak oxygen uptake. PLoS One 6:e28290. https://doi.org/10.1371/journal.pone.0028290
Tazi J, Bakkour N, Stamm S (2009) Alternative splicing and disease. Biochim Biophys Acta 1792:14–26. https://doi.org/10.1016/j.bbadis.2008.09.017
Tian B, Manley JL (2017) Alternative polyadenylation of mRNA precursors. Nat Rev Mol Cell Biol 18:18–30. https://doi.org/10.1038/nrm.2016.116
Tomita N, Morishita R (2004) Antisense oligonucleotides as a powerful molecular strategy for gene therapy in cardiovascular diseases. Curr Pharm Des 10:797–803
Troncoso R, Ibarra C, Vicencio JM, Jaimovich E, Lavandero S (2014) New insights into IGF-1 signaling in the heart. Trends Endocrinol Metab 25:128–137. https://doi.org/10.1016/j.tem.2013.12.002
Tumurkhuu G, Dagvadorj J, Jones HD, Chen S, Shimada K, Crother TR, Arditi M (2015) Alternatively spliced myeloid differentiation protein-2 inhibits TLR4-mediated lung inflammation. J Immunol 194:1686–1694. https://doi.org/10.4049/jimmunol.1402123
van den Bosch L (1998) Regulation of alternative processing of the sarco/endoplasmic reticulum Ca2+ ATPase (SERCA2) gene transcripts during muscle differentiation. Verh K Acad Geneeskd Belg 60:441–461
Ver Heyen M, Heymans S, Antoons G, Reed T, Periasamy M, Awede B, Lebacq J, Vangheluwe P, Dewerchin M, Collen D, Sipido K, Carmeliet P, Wuytack F (2001) Replacement of the muscle-specific sarcoplasmic reticulum Ca(2+)-ATPase isoform SERCA2a by the nonmuscle SERCA2b homologue causes mild concentric hypertrophy and impairs contraction-relaxation of the heart. Circ Res 89:838–846
Villate O, Turatsinze JV, Mascali LG, Grieco FA, Nogueira TC, Cunha DA, Nardelli TR, Sammeth M, Salunkhe VA, Esguerra JL, Eliasson L, Marselli L, Marchetti P, Eizirik DL (2014) Nova1 is a master regulator of alternative splicing in pancreatic beta cells. Nucleic Acids Res 42:11818–11830. https://doi.org/10.1093/nar/gku861
Vinciguerra M, Santini MP, Martinez C, Pazienza V, Claycomb WC, Giuliani A, Rosenthal N (2012) mIGF-1/JNK1/SirT1 signaling confers protection against oxidative stress in the heart. Aging Cell 11:139–149. https://doi.org/10.1111/j.1474-9726.2011.00766.x
Wahbi K, Algalarrondo V, Becane HM, Fressart V, Beldjord C, Azibi K, Lazarus A, Berber N, Radvanyi-Hoffman H, Stojkovic T, Behin A, Laforet P, Eymard B, Hatem S, Duboc D (2013) Brugada syndrome and abnormal splicing of SCN5A in myotonic dystrophy type 1. Arch Cardiovasc Dis 106:635–643. https://doi.org/10.1016/j.acvd.2013.08.003
Wang Z, Burge CB (2008) Splicing regulation: from a parts list of regulatory elements to an integrated splicing code. RNA 14:802–813. https://doi.org/10.1261/rna.876308
Watkins H, Conner D, Thierfelder L, Jarcho JA, MacRae C, McKenna WJ, Maron BJ, Seidman JG, Seidman CE (1995) Mutations in the cardiac myosin binding protein-C gene on chromosome 11 cause familial hypertrophic cardiomyopathy. Nat Genet 11:434–437. https://doi.org/10.1038/ng1295-434
Weeland CJ, van den Hoogenhof MM, Beqqali A, Creemers EE (2015) Insights into alternative splicing of sarcomeric genes in the heart. J Mol Cell Cardiol 81:107–113. https://doi.org/10.1016/j.yjmcc.2015.02.008
Wei C, Qiu J, Zhou Y, Xue Y, Hu J, Ouyang K, Banerjee I, Zhang C, Chen B, Li H, Chen J, Song LS, Fu XD (2015) Repression of the central splicing regulator RBFox2 is functionally linked to pressure overload-induced heart failure. Cell Rep. https://doi.org/10.1016/j.celrep.2015.02.013
Westenbrink BD, Ling H, Divakaruni AS, Gray CB, Zambon AC, Dalton ND, Peterson KL, Gu Y, Matkovich SJ, Murphy AN, Miyamoto S, Dorn GW 2nd, Heller Brown J (2015) Mitochondrial reprogramming induced by CaMKIIdelta mediates hypertrophy decompensation. Circ Res 116:e28–e39. https://doi.org/10.1161/CIRCRESAHA.116.304682
Xu X, Yang D, Ding JH, Wang W, Chu PH, Dalton ND, Wang HY, Bermingham JR Jr, Ye Z, Liu F, Rosenfeld MG, Manley JL, Ross J Jr, Chen J, Xiao RP, Cheng H, Fu XD (2005) ASF/SF2-regulated CaMKIIdelta alternative splicing temporally reprograms excitation-contraction coupling in cardiac muscle. Cell 120:59–72. https://doi.org/10.1016/j.cell.2004.11.036
Yamankurt G, Wu HC, McCarthy M, Cunha SR (2015) Exon organization and novel alternative splicing of Ank3 in mouse heart. PLoS One 10:e0128177. https://doi.org/10.1371/journal.pone.0128177
Yoshioka T, Inagaki K, Noguchi T, Sakai M, Ogawa W, Hosooka T, Iguchi H, Watanabe E, Matsuki Y, Hiramatsu R, Kasuga M (2009) Identification and characterization of an alternative promoter of the human PGC-1alpha gene. Biochem Biophys Res Commun 381:537–543. https://doi.org/10.1016/j.bbrc.2009.02.077
Zeng L, Xiao Q, Chen M, Margariti A, Martin D, Ivetic A, Xu H, Mason J, Wang W, Cockerill G, Mori K, Li JY, Chien S, Hu Y, Xu Q (2013) Vascular endothelial cell growth-activated XBP1 splicing in endothelial cells is crucial for angiogenesis. Circulation 127:1712–1722. https://doi.org/10.1161/CIRCULATIONAHA.112.001337
Zhang J, Manley JL (2013) Misregulation of pre-mRNA alternative splicing in cancer. Cancer Discov 3:1228–1237. https://doi.org/10.1158/2159-8290.CD-13-0253
Zhang L, Vincent GM, Baralle M, Baralle FE, Anson BD, Benson DW, Whiting B, Timothy KW, Carlquist J, January CT, Keating MT, Splawski I (2004) An intronic mutation causes long QT syndrome. J Am Coll Cardiol 44:1283–1291. https://doi.org/10.1016/j.jacc.2004.06.045
Zhang T, Lin Y, Liu J, Zhang ZG, Fu W, Guo LY, Pan L, Kong X, Zhang MK, Lu YH, Huang ZR, Xie Q, Li WH, Xu XQ (2016) Rbm24 regulates alternative splicing switch in embryonic stem cell cardiac lineage differentiation. Stem Cells 34:1776–1789. https://doi.org/10.1002/stem.2366
Zhang X, Zhou T, Yang J, Lin Y, Shi J, Zhang X, Frabutt DA, Zeng X, Li S, Venta PJ, Zheng YH (2017) Identification of SERINC5-001 as the predominant spliced isoform for HIV-1 restriction. J Virol 91. https://doi.org/10.1128/JVI.00137-17
Zhang Y, Huypens P, Adamson AW, Chang JS, Henagan TM, Boudreau A, Lenard NR, Burk D, Klein J, Perwitz N, Shin J, Fasshauer M, Kralli A, Gettys TW (2009) Alternative mRNA splicing produces a novel biologically active short isoform of PGC-1alpha. J Biol Chem 284:32813–32826. https://doi.org/10.1074/jbc.M109.037556
Zhao P, Turdi S, Dong F, Xiao X, Su G, Zhu X, Scott GI, Ren J (2009) Cardiac-specific overexpression of insulin-like growth factor I (IGF-1) rescues lipopolysaccharide-induced cardiac dysfunction and activation of stress signaling in murine cardiomyocytes. Shock 32:100–107. https://doi.org/10.1097/SHK.0b013e31818ec609
Zhou H, Liu X, Yu R, Long T, Zhao R, Liu H, Xu Y, Liang JG, Liang P (2016) Alternative splicing directs two IL-20R2 isoforms and is responsible for the incomplete gene knockout via the exon I ablation. Genes Immun 17:220–227. https://doi.org/10.1038/gene.2016.13
Zhu H, Ding Q (2015) Lower expression level of two RAGE alternative splicing isoforms in Alzheimer's disease. Neurosci Lett 597:66–70. https://doi.org/10.1016/j.neulet.2015.04.032
Funding
This study was supported by grants from the Priority Research Centers Program and Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Science, and Technology (2010-0020224, 2015R1A2A1A13001900, and 2015R1D1A1A01057937).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, H.K., Pham, M.H.C., Ko, K.S. et al. Alternative splicing isoforms in health and disease. Pflugers Arch - Eur J Physiol 470, 995–1016 (2018). https://doi.org/10.1007/s00424-018-2136-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-018-2136-x