Abstract
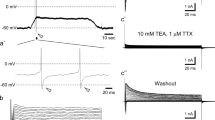
The powerful plant-derived irritant allyl isothiocyanate (AITC, aka mustard oil) induces hyperalgesia to heat in rodents and humans through mechanisms that are not yet fully understood. It is generally believed that AITC activates the broadly tuned chemosensory cation channel transient receptor potential cation channel subfamily A member 1 (TRPA1), triggering an inflammatory response that sensitizes the heat sensor transient receptor potential cation channel subfamily V member 1 (TRPV1). In the view of recent data demonstrating that AITC can directly activate TRPV1, we here explored the possibility that this compound sensitizes TRPV1 to heat stimulation in a TRPA1-independent manner. Patch-clamp recordings and intracellular Ca2+ imaging experiments in HEK293T cells over-expressing mouse TRPV1 revealed that the increase in channel activation induced by heating is larger in the presence of AITC than in control conditions. The analysis of the effects of AITC and heat on the current–voltage relationship of TRPV1 indicates that the mechanism of sensitization is based on additive shifts of the voltage dependence of activation towards negative voltages. Finally, intracellular Ca2+ imaging experiments in mouse sensory neurons isolated from Trpa1 KO mice yielded that AITC enhances the response to heat, specifically in the subpopulation expressing TRPV1. Furthermore, this effect was strongly reduced by the TRPV1 inhibitor capsazepine and virtually absent in neurons isolated from double Trpa1/Trpv1 KO mice. Taken together, these findings demonstrate that TRPV1 is a locus for cross sensitization between AITC and heat in sensory neurons and may help explaining, at least in part, the role of this channel in AITC-induced hyperalgesia to heat.




Similar content being viewed by others
References
Albin KC, Carstens MI, Carstens E (2008) Modulation of oral heat and cold pain by irritant chemicals. Chem Senses 33:3–15
Aneiros E, Cao L, Papakosta M, Stevens EB, Phillips S, Grimm C (2011) The biophysical and molecular basis of TRPV1 proton gating. EMBO J 30:994–1002
Bautista DM, Jordt SE, Nikai T, Tsuruda PR, Read AJ, Poblete J, Yamoah EN, Basbaum AI, Julius D (2006) TRPA1 mediates the inflammatory actions of environmental irritants and proalgesic agents. Cell 124:1269–1282
Carstens E, Mitsuyo T (2005) Neural correlates of oral irritation by mustard oil and other pungent chemicals: a hot topic. Chem Senses 30(Suppl 1):i203–i204
Caterina MJ, Leffler A, Malmberg AB, Martin WJ, Trafton J, Petersen-Zeitz KR, Koltzenburg M, Basbaum AI, Julius D (2000) Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science 288:306–313
Caterina MJ, Rosen TA, Tominaga M, Brake AJ, Julius D (1999) A capsaicin-receptor homologue with a high threshold for noxious heat. Nature 398:436–441
Descoeur J, Pereira V, Pizzoccaro A, Francois A, Ling B, Maffre V, Couette B, Busserolles J, Courteix C, Noel J, Lazdunski M, Eschalier A, Authier N, Bourinet E (2011) Oxaliplatin-induced cold hypersensitivity is due to remodelling of ion channel expression in nociceptors. EMBO Mol Med 3:266–278
Donnerer J, Liebmann I (2012) Phosphorylation of ERK1/2 in dorsal root ganglia following sequential mustard oil and thermal stimulation of the rat hind paw. Pharmacology 89:7–12
Dunham JP, Leith JL, Lumb BM, Donaldson LF (2010) Transient receptor potential channel A1 and noxious cold responses in rat cutaneous nociceptors. Neuroscience 165:1412–1419
Eckert WA 3rd, Julius D, Basbaum AI (2006) Differential contribution of TRPV1 to thermal responses and tissue injury-induced sensitization of dorsal horn neurons in laminae I and V in the mouse. Pain 126:184–197
Everaerts W, Gees M, Alpizar YA, Farre R, Leten C, Apetrei A, Dewachter I, van Leuven F, Vennekens R, de Ridder D, Nilius B, Voets T, Talavera K (2011) The capsaicin receptor TRPV1 is a crucial mediator of the noxious effects of mustard oil. Curr Biol 21:316–321
Fajardo O, Meseguer V, Belmonte C, Viana F (2008) TRPA1 channels mediate cold temperature sensing in mammalian vagal sensory neurons: pharmacological and genetic evidence. J Neurosci 28:7863–7875
Garcia-Martinez C, Humet M, Planells-Cases R, Gomis A, Caprini M, Viana F, de la Pena E, Sanchez-Baeza F, Carbonell T, de Felipe C, Perez-Paya E, Belmonte C, Messeguer A, Ferrer-Montiel A (2002) Attenuation of thermal nociception and hyperalgesia by VR1 blockers. Proc Natl Acad Sci USA 99:2374–2379
Gees M, Alpizar YA, Boonen B, Sanchez A, Everaerts W, Segal A, Xue F, Janssens A, Owsianik G, Nilius B, Voets T, Talavera K (2013) Mechanisms of TRPV1 activation and sensitization by allyl isothiocyanate. Mol Pharmacol doi:10.1124/mol.113.085548
Jain A, Bronneke S, Kolbe L, Stab F, Wenck H, Neufang G (2011) TRP-channel-specific cutaneous eicosanoid release patterns. Pain 152:2765–2772
Karashima Y, Talavera K, Everaerts W, Janssens A, Kwan KY, Vennekens R, Nilius B, Voets T (2009) TRPA1 acts as a cold sensor in vitro and in vivo. Proc Natl Acad Sci USA 106:1273–1278
Kwan KY, Allchorne AJ, Vollrath MA, Christensen AP, Zhang DS, Woolf CJ, Corey DP (2006) TRPA1 contributes to cold, mechanical, and chemical nociception but is not essential for hair-cell transduction. Neuron 50:277–289
Liu B, Hui K, Qin F (2003) Thermodynamics of heat activation of single capsaicin ion channels VR1. Biophys J 85:2988–3006
Malkia A, Madrid R, Meseguer V, de la Pena E, Valero M, Belmonte C, Viana F (2007) Bidirectional shifts of TRPM8 channel gating by temperature and chemical agents modulate the cold sensitivity of mammalian thermoreceptors. J Physiol 581:155–174
Mazario J, Basbaum AI (2007) Contribution of substance P and neurokinin A to the differential injury-induced thermal and mechanical responsiveness of lamina I and V neurons. J Neurosci 27:762–770
McMahon SB, Abel C (1987) A model for the study of visceral pain states: chronic inflammation of the chronic decerebrate rat urinary bladder by irritant chemicals. Pain 28:109–127
Merrill AW, Cuellar JM, Judd JH, Carstens MI, Carstens E (2008) Effects of TRPA1 agonists mustard oil and cinnamaldehyde on lumbar spinal wide-dynamic range neuronal responses to innocuous and noxious cutaneous stimuli in rats. J Neurophysiol 99:415–425
Meseguer VM, Denlinger BL, Talavera K (2011) Methodological considerations to understand the sensory function of TRP channels. Curr Pharm Biotechnol 12:3–11
Meseguer V, Karashima Y, Talavera K, D'Hoedt D, Donovan-Rodriguez T, Viana F, Nilius B, Voets T (2008) Transient receptor potential channels in sensory neurons are targets of the antimycotic agent clotrimazole. J Neurosci 28:576–586
Nilius B, Appendino G (2013) Spices: the savory and beneficial science of pungency. Rev Physiol Biochem Pharmacol PMID: 23605179 (in press)
Nilius B, Appendino G, Owsianik G (2012) The transient receptor potential channel TRPA1: from gene to pathophysiology. Pflugers Arch 464:425–458
Nilius B, Talavera K, Owsianik G, Prenen J, Droogmans G, Voets T (2005) Gating of TRP channels: a voltage connection? J Physiol 567:35–44
Ohta T, Imagawa T, Ito S (2007) Novel agonistic action of mustard oil on recombinant and endogenous porcine transient receptor potential V1 (pTRPV1) channels. Biochem Pharmacol 73:1646–1656
Raisinghani M, Zhong L, Jeffry JA, Bishnoi M, Pabbidi RM, Pimentel F, Cao DS, Evans MS, Premkumar LS (2011) Activation characteristics of transient receptor potential ankyrin 1 and its role in nociception. Am J Physiol Cell Physiol 301:C587–C600
Rask L, Andreasson E, Ekbom B, Eriksson S, Pontoppidan B, Meijer J (2000) Myrosinase: gene family evolution and herbivore defense in Brassicaceae. Plant Mol Biol 42:93–113
Reeh PW, Kocher L, Jung S (1986) Does neurogenic inflammation alter the sensitivity of unmyelinated nociceptors in the rat? Brain Res 384:42–50
Sawada Y, Hosokawa H, Hori A, Matsumura K, Kobayashi S (2007) Cold sensitivity of recombinant TRPA1 channels. Brain Res 1160:39–46
Sawyer CM, Carstens MI, Carstens E (2009) Mustard oil enhances spinal neuronal responses to noxious heat but not cooling. Neurosci Lett 461:271–274
Simons CT, Sudo S, Sudo M, Carstens E (2004) Mustard oil has differential effects on the response of trigeminal caudalis neurons to heat and acidity. Pain 110:64–71
Story GM, Peier AM, Reeve AJ, Eid SR, Mosbacher J, Hricik TR, Earley TJ, Hergarden AC, Andersson DA, Hwang SW, McIntyre P, Jegla T, Bevan S, Patapoutian A (2003) ANKTM1, a TRP-like channel expressed in nociceptive neurons, is activated by cold temperatures. Cell 112:819–829
Talavera K, Nilius B (2011) Electrophysiological methods for the study of TRP channels. In: Zhu MX (ed) Series methods in signal transduction. CRC, Boca Raton, pp 237–254
Talavera K, Nilius B, Voets T (2008) Neuronal TRP channels: thermometers, pathfinders and life-savers. Trends Neurosci 31:287–295
Voets T, Droogmans G, Wissenbach U, Janssens A, Flockerzi V, Nilius B (2004) The principle of temperature-dependent gating in cold- and heat-sensitive TRP channels. Nature 430:748–754
Vriens J, Owsianik G, Hofmann T, Philipp SE, Stab J, Chen X, Benoit M, Xue F, Janssens A, Kerselaers S, Oberwinkler J, Vennekens R, Gudermann T, Nilius B, Voets T (2011) TRPM3 is a nociceptor channel involved in the detection of noxious heat. Neuron 70:482–494
Wang S, Lee J, Ro JY, Chung MK (2012) Warmth suppresses and desensitizes damage-sensing ion channel TRPA1. Mol Pain 8:22
Xu M, Kim CJ, Neubert MJ, Heinricher MM (2007) NMDA receptor-mediated activation of medullary pro-nociceptive neurons is required for secondary thermal hyperalgesia. Pain 127:253–262
Acknowledgments
We would like to thank Kelvin Kwan and David Corey and Christopher Benham for providing us the Trpa1 and Trpv1 KO mice, respectively. We would like to thank Melissa Benoit for the maintenance of the cell cultures. This work was supported by grants from the Belgian Federal Government (IUAP P7/13), the Research Foundation-Flanders (grants G.0565.07, G.0686.09, and G.A022.11N), and the Research Council of the KU Leuven (grants GOA 2009/07, EF/95/010, and PFV/10/006). B.B. was funded by a Ph.D. grant of the Agency for Innovation by Science and Technology (IWT). M.G was supported by a doctoral fellowship from the F.W.O.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alpizar, Y.A., Boonen, B., Gees, M. et al. Allyl isothiocyanate sensitizes TRPV1 to heat stimulation. Pflugers Arch - Eur J Physiol 466, 507–515 (2014). https://doi.org/10.1007/s00424-013-1334-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-013-1334-9