Abstract.
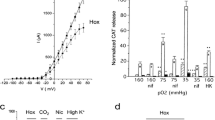
In mammals, adrenomedullary chromaffin cells (AMC) express a neutrophil-like NADPH oxidase and secrete catecholamines, which play a vital role in the ability of the neonate to survive hypoxic stress. To test whether NADPH oxidase functions as an O2 sensor, and whether mouse AMC express a developmentally regulated O2-sensing mechanism similar to rats, we compared the effects of hypoxia on cultured AMC from wild-type (WT) and transgenic oxidase-deficient (OD) mice, lacking the gp91phox subunit of NADPH oxidase. Hypoxia (pO2 ≅5 mmHg) caused a reversible inhibition of outward K+ current by ≅27% (n=6) in WT and ≅29% (n=9) in OD neonatal (P1-P5) chromaffin cells. O2-sensitive K+ currents included both Ca2+-dependent (I BK) and a delayed rectifier-like K+ current (I KV). Additionally, hypoxia depolarized WT and OD chromaffin cells and caused reversible broadening of the action potential. Exposure of both WT and OD neonatal AMC cultures to hypoxia (5% O2) for ≅1 h caused four- to sixfold stimulation of catecholamine (CA) secretion as determined by HPLC. In contrast, hypoxia had no significant effect on K+ currents or CA secretion in juvenile (P14-P15) AMC. Thus, mouse AMC possess a developmentally regulated O2-sensing mechanism, but NADPH oxidase does not function as the primary O2 sensor in these cells.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Thompson, R.J., Farragher, S.M., Cutz, E. et al. Developmental regulation of O2 sensing in neonatal adrenal chromaffin cells from wild-type and NADPH-oxidase-deficient mice. Pflügers Arch - Eur J Physiol 444, 539–548 (2002). https://doi.org/10.1007/s00424-002-0853-6
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00424-002-0853-6