Abstract
Purpose
To determine whether 14 inflammation-, angiogenesis-, and adhesion-related proteins in cord blood (CB), alone or in combination with conventional perinatal factors, could predict retinopathy of prematurity (ROP) in preterm infants.
Methods
Data from 111 preterm infants (born at ≤ 32.0 weeks) were retrospectively reviewed. The levels of endoglin, E-selectin, HSP70, IGFBP-3/4, LBP, lipocaline-2, M-CSFR, MIP-1α, pentraxin 3, P-selectin, TGFBI, TGF-β1, and TNFR2 were assessed in stored CB samples collected at birth using ELISA kits. The primary endpoints included severe ROP (≥ stage 3) and type 1 ROP requiring treatment.
Results
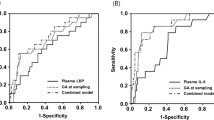
ROP was diagnosed in 29 infants (26.1%), among whom 14 (12.6%) had severe ROP and seven (6.3%) had type 1 ROP. Multivariate logistic regression showed that decreased CB TGFBI levels were significantly associated with severe ROP and type 1 ROP after adjusting for gestational age at birth. Stepwise regression analysis allowed to design prediction models with good accuracy, which comprised low CB TGFBI levels and low birth weight (BW) as predictors for severe ROP (area under the curve [AUC] = 0.888), and low CB endoglin levels and low BW as predictors for type 1 ROP (AUC = 0.950). None of the other CB proteins evaluated were found to be associated with severe ROP or type 1 ROP.
Conclusions
Low CB TGFBI levels are associated with severe ROP and type 1 ROP, independently of gestational age. Moreover, combined predictive models based on CB TGFBI and endoglin levels, along with BW data, may act as good indicators at birth for the neonatal risk of ROP progression.

Similar content being viewed by others
Data availability
The datasets used and analyzed in the current study are available from the corresponding author upon reasonable request.
References
Hong EH, Shin YU, Bae GH, Choi YJ, Ahn SJ, Sobrin L, Hong R, Kim I, Cho H (2021) Nationwide incidence and treatment pattern of retinopathy of prematurity in South Korea using the 2007–2018 national health insurance claims data. Sci Rep 11:1451. https://doi.org/10.1038/s41598-021-80989-z
Hellstrom A, Smith LE, Dammann O (2013) Retinopathy of prematurity. Lancet 382:1445–1457. https://doi.org/10.1016/S0140-6736(13)60178-6
Hartnett ME (2015) Pathophysiology and mechanisms of severe retinopathy of prematurity. Ophthalmology 122:200–210. https://doi.org/10.1016/j.ophtha.2014.07.050
Binenbaum G, Ying GS, Quinn GE, Dreiseitl S, Karp K, Roberts RS, Kirpalani H, Premature Infants in Need of Transfusion Study G (2011) A clinical prediction model to stratify retinopathy of prematurity risk using postnatal weight gain. Pediatrics 127:e607-614. https://doi.org/10.1542/peds.2010-2240
Hartnett ME, Penn JS (2012) Mechanisms and management of retinopathy of prematurity. N Engl J Med 367:2515–2526. https://doi.org/10.1056/NEJMra1208129
Dammann O, Rivera JC, Chemtob S (2021) The prenatal phase of retinopathy of prematurity. Acta Paediatr 110:2521–2528. https://doi.org/10.1111/apa.15945
Woo SJ, Park JY, Hong S, Kim YM, Park YH, Lee YE, Park KH (2020) Inflammatory and Angiogenic Mediators in Amniotic Fluid Are Associated With the Development of Retinopathy of Prematurity in Preterm Infants. Invest Ophthalmol Vis Sci 61:42. https://doi.org/10.1167/iovs.61.5.42
Park YJ, Woo SJ, Kim YM, Hong S, Lee YE, Park KH (2019) Immune and Inflammatory Proteins in Cord Blood as Predictive Biomarkers of Retinopathy of Prematurity in Preterm Infants. Invest Ophthalmol Vis Sci 60:3813–3820. https://doi.org/10.1167/iovs.19-27258
Lynch AM, Berning AA, Thevarajah TS, Wagner BD, Post MD, McCourt EA, Cathcart JN, Hodges JK, Mandava N, Gibbs RS, Palestine AG (2018) The role of the maternal and fetal inflammatory response in retinopathy of prematurity. Am J Reprod Immunol 80:e12986. https://doi.org/10.1111/aji.12986
Lynch AM, Wagner BD, Hodges JK, Thevarajah TS, McCourt EA, Cerda AM, Mandava N, Gibbs RS, Palestine AG (2017) The relationship of the subtypes of preterm birth with retinopathy of prematurity. Am J Obstet Gynecol 217:354e351-354 e358. https://doi.org/10.1016/j.ajog.2017.05.029
Mor F, Quintana FJ, Cohen IR (2004) Angiogenesis-inflammation cross-talk: vascular endothelial growth factor is secreted by activated T cells and induces Th1 polarization. J Immunol 172:4618–4623. https://doi.org/10.4049/jimmunol.172.7.4618
Aguilar-Cazares D, Chavez-Dominguez R, Carlos-Reyes A, Lopez-Camarillo C, Hernadez de la Cruz ON, Lopez-Gonzalez JS (2019) Contribution of Angiogenesis to Inflammation and Cancer. Front Oncol 9:1399. https://doi.org/10.3389/fonc.2019.01399
Thapa N, Lee BH, Kim IS (2007) TGFBIp/betaig-h3 protein: a versatile matrix molecule induced by TGF-beta. Int J Biochem Cell Biol 39:2183–2194. https://doi.org/10.1016/j.biocel.2007.06.004
Koch AE, Halloran MM, Haskell CJ, Shah MR, Polverini PJ (1995) Angiogenesis mediated by soluble forms of E-selectin and vascular cell adhesion molecule-1. Nature 376:517–519. https://doi.org/10.1038/376517a0
Ley K (2003) The role of selectins in inflammation and disease. Trends Mol Med 9:263–268. https://doi.org/10.1016/s1471-4914(03)00071-6
Pieh C, Kruger M, Lagreze WA, Gimpel C, Buschbeck C, Zirrgiebel U, Agostini HT (2010) Plasma sE-selectin in premature infants: a possible surrogate marker of retinopathy of prematurity. Invest Ophthalmol Vis Sci 51:3709–3713. https://doi.org/10.1167/iovs.09-4723
Sood BG, Madan A, Saha S, Schendel D, Thorsen P, Skogstrand K, Hougaard D, Shankaran S, Carlo W, network Nnr (2010) Perinatal systemic inflammatory response syndrome and retinopathy of prematurity. Pediatr Res 67:394–400. https://doi.org/10.1203/PDR.0b013e3181d01a36
Fierson WM, American Academy of Pediatrics Section on O, American Academy of O, American Association for Pediatric O, Strabismus, American Association of Certified O (2013) Screening examination of premature infants for retinopathy of prematurity. Pediatrics 131:189–195. https://doi.org/10.1542/peds.2012-2996
Fierson WM (2018) Screening examination of premature infants for retinopathy of prematurity. Pediatrics 142:e20183061. https://doi.org/10.1542/peds.2018-3061
Early Treatment for Retinopathy of Prematurity Cooperative G, Good WV, Hardy RJ, Dobson V, Palmer EA, Phelps DL, Tung B, Redford M (2010) Final visual acuity results in the early treatment for retinopathy of prematurity study. Arch Ophthalmol 128:663–671. https://doi.org/10.1001/archophthalmol.2010.72
Cooperative ETFROP, G, (2003) Revised indications for the treatment of retinopathy of prematurity: results of the early treatment for retinopathy of prematurity randomized trial. Arch Ophthalmol 121:1684–1694. https://doi.org/10.1001/archopht.121.12.1684
Song JS, Woo SJ, Park KH, Kim H, Lee KN, Kim YM (2022) Association of inflammatory and angiogenic biomarkers in maternal plasma with retinopathy of prematurity in preterm infants. Eye (Lond). https://doi.org/10.1038/s41433-022-02234-9
Woo SJ, Park KH, Lee SY, Ahn SJ, Ahn J, Park KH, Oh KJ, Ryu A (2013) The relationship between cord blood cytokine levels and perinatal factors and retinopathy of prematurity: a gestational age-matched case-control study. Invest Ophthalmol Vis Sci 54:3434–3439. https://doi.org/10.1167/iovs.13-11837
Jung EY, Choi BY, Rhee J, Park J, Cho SH, Park KH (2017) Relation between amniotic fluid infection or cytokine levels and hearing screen failure in infants at 32 wk gestation or less. Pediatr Res 81:349–355. https://doi.org/10.1038/pr.2016.219
Kim CJ, Romero R, Chaemsaithong P, Chaiyasit N, Yoon BH, Kim YM (2015) Acute chorioamnionitis and funisitis: definition, pathologic features, and clinical significance. Am J Obstet Gynecol 213:S29-52. https://doi.org/10.1016/j.ajog.2015.08.040
Park KH, Kim SN, Oh KJ, Lee SY, Jeong EH, Ryu A (2012) Noninvasive prediction of intra-amniotic infection and/or inflammation in preterm premature rupture of membranes. Reprod Sci 19:658–665. https://doi.org/10.1177/1933719111432869
Lee JY, Park KH, Kim A, Yang HR, Jung EY, Cho SH (2017) Maternal and Placental Risk Factors for Developing Necrotizing Enterocolitis in Very Preterm Infants. Pediatr Neonatol 58:57–62. https://doi.org/10.1016/j.pedneo.2016.01.005
Lee JJ (2007) Birth weight for gestational age patterns by sex, plurality, and parity in Korean population. Korean J Pediatr 50:732–739
Doubilet PM, Benson CB, Nadel AS, Ringer SA (1997) Improved birth weight table for neonates developed from gestations dated by early ultrasonography. J Ultrasound Med 16:241–249. https://doi.org/10.7863/jum.1997.16.4.241
Vandenbroucke JP, von Elm E, Altman DG, Gotzsche PC, Mulrow CD, Pocock SJ, Poole C, Schlesselman JJ, Egger M, Initiative S (2007) Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): explanation and elaboration. PLoS Med 4:e297. https://doi.org/10.1371/journal.pmed.0040297
Puhr R, Heinze G, Nold M, Lusa L, Geroldinger A (2017) Firth’s logistic regression with rare events: accurate effect estimates and predictions? Stat Med 36:2302–2317. https://doi.org/10.1002/sim.7273
DeLong ER, DeLong DM, Clarke-Pearson DL (1988) Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44:837–845. https://doi.org/10.2307/2531595
Madan A, El-Ferzli G, Carlson SM, Whitin JC, Schilling J, Najmi A, Yu TT, Lau K, Dimmitt RA, Cohen HJ (2007) A potential biomarker in the cord blood of preterm infants who develop retinopathy of prematurity. Pediatr Res 61:215–221. https://doi.org/10.1203/pdr.0b013e31802d776d
Cakir U, Tayman C, Yucel C, Ozdemir O (2019) Can IL-33 and Endocan be New Markers for Retinopathy of Prematurity? Comb Chem High Throughput Screen 22:41–48. https://doi.org/10.2174/1386207322666190325120244
Bae JS, Lee W, Nam JO, Kim JE, Kim SW, Kim IS (2014) Transforming growth factor beta-induced protein promotes severe vascular inflammatory responses. Am J Respir Crit Care Med 189:779–786. https://doi.org/10.1164/rccm.201311-2033OC
Chen Y, Zhao H, Feng Y, Ye Q, Hu J, Guo Y, Feng Y (2021) Pan-Cancer Analysis of the Associations of TGFBI Expression With Prognosis and Immune Characteristics. Front Mol Biosci 8:745649. https://doi.org/10.3389/fmolb.2021.745649
Stark A, Dammann C, Nielsen HC, Volpe MV (2018) A Pathogenic Relationship of Bronchopulmonary Dysplasia and Retinopathy of Prematurity? A Review of Angiogenic Mediators in Both Diseases. Front Pediatr 6:125. https://doi.org/10.3389/fped.2018.00125
Ahlfeld SK, Wang J, Gao Y, Snider P, Conway SJ (2016) Initial Suppression of Transforming Growth Factor-beta Signaling and Loss of TGFBI Causes Early Alveolar Structural Defects Resulting in Bronchopulmonary Dysplasia. Am J Pathol 186:777–793. https://doi.org/10.1016/j.ajpath.2015.11.024
Liu M, Iosef C, Rao S, Domingo-Gonzalez R, Fu S, Snider P, Conway SJ, Umbach GS, Heilshorn SC, Dewi RE, Dahl MJ, Null DM, Albertine KH, Alvira CM (2021) Transforming Growth Factor-induced Protein Promotes NF-κB-mediated Angiogenesis during Postnatal Lung Development. Am J Respir Cell Mol Biol 64:318–330. https://doi.org/10.1165/rcmb.2020-0153OC
Jang JH, Kim YC (2020) Retinal vascular development in an immature retina at 33–34 weeks postmenstrual age predicts retinopathy of prematurity. Sci Rep 10:18111. https://doi.org/10.1038/s41598-020-75151-0
Lofqvist C, Hansen-Pupp I, Andersson E, Holm K, Smith LE, Ley D, Hellstrom A (2009) Validation of a new retinopathy of prematurity screening method monitoring longitudinal postnatal weight and insulinlike growth factor I. Arch Ophthalmol 127:622–627. https://doi.org/10.1001/archophthalmol.2009.69
Lofqvist C, Andersson E, Sigurdsson J, Engstrom E, Hard AL, Niklasson A, Smith LE, Hellstrom A (2006) Longitudinal postnatal weight and insulin-like growth factor I measurements in the prediction of retinopathy of prematurity. Arch Ophthalmol 124:1711–1718. https://doi.org/10.1001/archopht.124.12.1711
Holm M, Morken TS, Fichorova RN, VanderVeen DK, Allred EN, Dammann O, Leviton A, Neonatology ES, Ophthalmology C (2017) Systemic Inflammation-Associated Proteins and Retinopathy of Prematurity in Infants Born Before the 28th Week of Gestation. Invest Ophthalmol Vis Sci 58:6419–6428. https://doi.org/10.1167/iovs.17-21931
Yu H, Yuan L, Zou Y, Peng L, Wang Y, Li T, Tang S (2014) Serum concentrations of cytokines in infants with retinopathy of prematurity. APMIS 122:818–823. https://doi.org/10.1111/apm.12223
Hellgren G, Lofqvist C, Hansen-Pupp I, Gram M, Smith LE, Ley D, Hellstrom A (2018) Increased postnatal concentrations of pro-inflammatory cytokines are associated with reduced IGF-I levels and retinopathy of prematurity. Growth Horm IGF Res 39:19–24. https://doi.org/10.1016/j.ghir.2017.11.006
Zasada M, Suski M, Bokiniec R, Szwarc-Duma M, Borszewska-Kornacka MK, Madej J, Bujak-Gizycka B, Madetko-Talowska A, Revhaug C, Baumbusch LO, Saugstad OD, Pietrzyk JJ, Kwinta P (2018) An iTRAQ-Based Quantitative Proteomic Analysis of Plasma Proteins in Preterm Newborns With Retinopathy of Prematurity. Invest Ophthalmol Vis Sci 59:5312–5319. https://doi.org/10.1167/iovs.18-24914
Smith LE, Hard AL, Hellstrom A (2013) The biology of retinopathy of prematurity: how knowledge of pathogenesis guides treatment. Clin Perinatol 40:201–214. https://doi.org/10.1016/j.clp.2013.02.002
Goldstein GP, Leonard SA, Kan P, Koo EB, Lee HC, Carmichael SL (2019) Prenatal and postnatal inflammation-related risk factors for retinopathy of prematurity. J Perinatol 39:964–973. https://doi.org/10.1038/s41372-019-0357-2
Siatkowski RM, Dobson V, Quinn GE, Summers CG, Palmer EA, Tung B (2007) Severe visual impairment in children with mild or moderate retinal residua following regressed threshold retinopathy of prematurity. J AAPOS 11:148–152. https://doi.org/10.1016/j.jaapos.2006.11.113
Acknowledgements
We would like to thank the Division of Statistics in Medical Research Collaborating Center (MRCC) at Seoul National University Bundang Hospital for statistical analysis.
Funding
This research was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (No. 2020R1F1A1048362). The funders had no role in the design of this study, data collection, data analyses, data interpretation, or in the writing of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
The study adhered to the tenets of the Declaration of Helsinki and was approved by the Institutional Review Board of the Ethics Committee of the Seoul National University Bundang Hospital, Seongnamsi, Korea (approval no. B-1006/103–102).
Consent to participate
Written informed consent was obtained from the parents of all infants for use of their biological samples and clinical data for research purposes.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Song, J.S., Woo, S.J., Park, K.H. et al. Cord blood transforming growth factor-β-induced as predictive biomarker of retinopathy of prematurity in preterm infants. Graefes Arch Clin Exp Ophthalmol 261, 2477–2488 (2023). https://doi.org/10.1007/s00417-023-06056-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-023-06056-7