Abstract
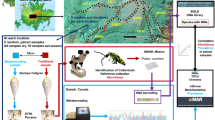
Soil is often collected from a suspect’s tire, vehicle, or shoes during a criminal investigation and subsequently submitted to a forensic laboratory for analysis. Plant and insect material recovered in such samples is rarely analyzed, as morphological identification is difficult. In this study, DNA barcoding was used for taxonomic identifications by targeting the gene regions known to permit discrimination in plants [maturase K (matK) and ribulose 1,5-biphosphate carboxylase (rbcL)] and insects [cytochrome oxidase subunit I (COI)]. A DNA barcode protocol suitable for processing forensic-type biological fragments was developed and its utility broadly tested with forensic-type fragments (e.g., seeds, leaves, bark, head, legs; n, 213) isolated from soils collected within Virginia, USA (n, 11). Difficulties with PCR inhibitors in plant extracts and obtaining clean Sanger sequence data from insect amplicons were encountered during protocol development; however, the final protocol produced sequences specific to the expected locus and taxa. The overall quantity and quality of DNA extracted from the 213 forensic-type biological fragments was low (< 15 ng/μL). For plant fragments, only the rbcL sequence data was deemed reliable; thus, taxonomic identifications were limited to the family level. The majority of insect sequences matched COI in both GenBank and Barcode of Life DataSystems; however, they were identified as an undescribed environmental contaminant. Although limited taxonomic information was gleaned from the forensic-type fragments processed in this study, the new protocol shows promise for obtaining reliable and specific identifications through DNA barcoding, which could ultimately enhance the information gleaned from soil examinations.




Similar content being viewed by others
References
Post RJ, Flook PK, Millest AL (1993) Method for the preservation of insects for DNA studies. Biochem Syst Ecol 21(1):85–92. https://doi.org/10.1016/0305-1978(93)90012-G
Drábková LZ (2014) DNA extraction from herbarium specimens. Methods Mol Biol 1115:69–84. https://doi.org/10.1007/978-1-62703-767-9_4
Peterson PM, Romaschenko K, Soreng RJ (2014) A laboratory guide for generating DNA barcodes in grasses: a case study of Leptochloa s.l. (Poaceae: Chloridoideae). J Plant Taxon Geogr 69(1):1–12. https://doi.org/10.1080/00837792.2014.927555
Hebert PDN, Cywinska A, Ball SL, deWaard JR (2003) Biological identifications through DNA barcodes. Proc Royal Soc London: Biol Sci 270(1512):313–321. https://doi.org/10.1098/rspb.2002.2218
Moritz C, Cicero C (2004) DNA barcoding: promise and pitfalls. PLoS Biol 2(10):e354. https://doi.org/10.1371/journal.pbio.0020354
Will KW, Rubinoff D (2004) Myth of the molecule: DNA barcodes for species cannot replace morphology for identification and classification. Cladistics 20(1):47–55. https://doi.org/10.1111/j.1096-0031.2003.00008.x
Ebach MC, Holdrege C (2005) More taxonomy, not DNA barcoding. Bioscience 55(10):822–824. https://doi.org/10.1641/0006-3568(2005)055[0823:MTNDB]2.0.CO;2
Will KW, Mishler BD, Wheeler QD, Savolainen V (2005) The perils of DNA barcoding and the need for integrative taxonomy. Syst Biol 54(5):844–851. https://doi.org/10.1080/10635150500354878
Meier R, Shiyang K, Vaidya G et al (2006) DNA barcoding and taxonomy in Diptera: a tale of high intraspecific variability and low identification success. Syst Biol 55(5):715–728. https://doi.org/10.1080/10635150600969864
Witt JDS, Threloff DL, Hebert PDN (2006) DNA barcoding reveals extraordinary cryptic diversity in an amphipod genus: implications for desert spring conservation. Mol Ecol 15(10):3073–3082. https://doi.org/10.1111/j.1365-294X.2006.02999.x
Hajibabaei M, Singer GAC, Hebert PDN, Hickey DA (2007) DNA barcoding: how it complements taxonomy, molecular phylogenetics and population genetics. Trends Genet 23(4):167–172. https://doi.org/10.1016/j.tig.2007.02.001
Ward RD, Holmes BH, White WT, Last PR (2007) DNA barcoding Australasian chondrichthyans: results and potential uses in conservation. Mar Freshw Res 59(1):57–71. https://doi.org/10.1071/MF07148
Ardura A, Linde AR, Moreira JC, Garcia-Vazquez E (2010) DNA barcoding for conservation and management of Amazonian commercial fish. Biol Conserv 143(6):1438–1443. https://doi.org/10.1016/j.biocon.2010.03.019
Kress WJ, García-Robledo C, Uriarte M, Erickson DL (2015) DNA barcodes for ecology, evolution and conservation. Trends Ecol Evol 30(1):25–65. https://doi.org/10.1016/j.tree.2014.10.008
Ball SL, Armstrong KF (2006) DNA barcodes for insect pest identification: a test case with tussock moths (Lepidoptera: Lymantriidae). Can J Forest Res 36(2):337–350. https://doi.org/10.1139/x05-276
Saunders GW (2009) Routine DNA barcoding of Canadian Gracilariales (Rhodophyta) reveals the invasive species Gracilaria vermiculophylla in British Columbia. Mol Ecol Resour 9:140–150. https://doi.org/10.1111/j.1755-0998.2009.02639.x
Stoeckle MY, Gamble CC, Kirpekar et al (2011) Commercial teas highlight plant DNA barcode identification successes and obstacles. Sci Rep 1(1):42. https://doi.org/10.1038/srep00042
Galimberti A, De Mattia F, Losa A et al (2013) DNA barcoding as a new tool for food traceability. Food Res Int 50:55–63. https://doi.org/10.1016/j.foodres.2012.09.036
Ivanova NV, Kuzmina ML, Braukmann TWA, Borisenko AV, Zakharov EV (2016) Authentication of herbal supplements using next-generation sequencing. PLoS One 11(5):e0156426. https://doi.org/10.1371/journal.pone.0156426
Mishra P, Kumar A, Nagireddy A, Mani DN, Shukla AK, Tiwari R, Sundaresan V (2016) DNA barcoding: an efficient tool to overcome authentication challenges in the herbal market. Plant Biotechnol J 14(1):8–21. https://doi.org/10.1111/pbi.12419
Zhang T, Wang Y-J, Guo W et al (2016) DNA barcoding, species-specific PCR and real-time PCR techniques for the identification of six Trobolium pests of stored products. Sci Rep 6:28494. https://doi.org/10.1038/srep28494
Bruni I, De Mattia F, Galimberti A et al (2010) Identification of poisonous plants by DNA barcoding approach. Int J Legal Med 124(6):595–603. https://doi.org/10.1007/s00414-010-0447-3
Dalton DL, Kotze A (2011) DNA barcoding as a tool for species identification in three forensic wildlife cases in South Africa. Forensic Sci Int 207:e51–e54. https://doi.org/10.1016/j.forsciint.2010.12.017
Jones YL, Peters SM, Weland C et al (2013) Potential use of DNA barcodes in regulatory science: identification of the U.S. Food and Drug Administration’s “dirty 22,” contributors to the spread of foodborne pathogens. J Food Prot 76(1):144–149. https://doi.org/10.4315/0362-028X.JFP-12-168
Ng KKS, Lee SL, Tnah LH, Nurul-Farhanah Z, Ng CH, Lee CT, Tani N, Diway B, Lai PS, Khoo E (2016) Forensic timber identification: a case study of CITES listed species, Gonystylus bancanus (Thymelaeaceae). Forensic Sci Int Genet 23:197–209. https://doi.org/10.1016/j.fsigen.2016.05.002
Ratnasingham S, Hebert PDN (2007) BOLD: the barcode of life data system. (www.barcodinglife.org). Mol Ecol Notes 7(3):355–364. https://doi.org/10.1111/j.1471-8286.2007.01678.x
Benson DA, Karsch-Mizrachi I, Lipman DJ et al (2002) GenBank. Nucleic Acids Res 30(1):17–20
PDN H, Ratnasingham S, de Waard JR (2003) Barcoding animal life: cytochrome c oxidase subunit 1 divergences among closely related species. Proceedings of the Royal Society of London. Biological Sciences 270:s96–s99
Kress WJ, Erickson DL (2012) DNA barcodes: methods and protocols. Methods Mol Biol 858:3–8. https://doi.org/10.1007/978-1-61779-591-6_1
Dasmahapatra KK, Mallet J (2006) DNA barcodes: recent successes and future prospects. Heredity 97(4):254–255. https://doi.org/10.1038/sj.hdy.6800858
Ward RD, Zemlak TS, Innes BH, Last PR, Hebert PD (2005) DNA barcoding Australia’s fish species. Philos Trans Royal Soc Biol 360(1462):1847–1857. https://doi.org/10.1098/rstb.2005.1716
Tavares ES, Baker AJ (2008) Single mitochondrial gene barcodes reliably identify sister-species in diverse clades of birds. BMC Evol Biol 8(1):81. https://doi.org/10.1186/1471-2148-8-81
Hebert PDN, Penton EH, Burns JM, Janzen DH, Hallwachs W (2004) Ten species in one: DNA barcoding reveals cryptic species in the neotropical skipper butterfly Astraptes fulgerator. Proc Natl Acad Sci U S A 101(41):14812–14817. https://doi.org/10.1073/pnas.0406166101
Ball SL, Hebert PDN, Burian SK, Webb JM (2005) Biological identifications of mayflies (Epheneroptera) using DNA barcodes. J N Am Benthol Soc 24(3):508–524. https://doi.org/10.1899/04-142.1
Nelson LA, Wallman JF, Dowton M (2007) Using COI barcodes to identify forensically and medically important blowflies. Med Vet Entomol 21(1):44–52. https://doi.org/10.1111/j.1365-2915.2007.00664.x
Meiklejohn KA, Wallman JF, Dowton M (2011) DNA-based identification of forensically important Australian Sarcophagidae (Diptera). Int J Legal Med 125(1):27–32. https://doi.org/10.1007/s00414-009-0395-y
Meiklejohn KA, Wallman JF, Cameron SL, Dowton M (2012) Comprehensive evaluation of DNA barcoding of the forensically important Australian Sarcophagidae (Diptera). Invertebr Syst 26(6):515–525. https://doi.org/10.1071/IS12008
Hollingsworth PM, Forrest LL, Spouge JL et al (2009) A DNA barcode for land plants. PNAS 106:12794–12797. https://doi.org/10.1073/pnas.0905845106
Hollingsworth PM, Graham SW, Little DP (2011) Choosing and using a plant DNA barcode. PLoS One 6(5):e19254. https://doi.org/10.1371/journal.pone.0019254
Ferri G, Corradini B, Ferrari F, Santunione AL, Palazzoli F, Alu M (2015) Forensic botany II, DNA barcode for land plants: which markers after international agreement? Forensic Sci Int Genet 15:131–136. https://doi.org/10.1016/j.fsigen.2014.10.005
Epp LS, Boessenkool S, Bellemain EP et al (2012) New environmental metabarcodes for analyzing soil DNA: potential for studying past and present ecosystems. Mol Ecol 21(8):1821–1833. https://doi.org/10.1111/j.1365-294X.2012.05537.x
Taberlet P, Prud’Homme SM, Campione E et al (2012) Soil sampling and isolation of extracellular DNA from large amount of starting material suitable for metabarcoding studies. Mol Ecol 21(8):1816–1820. https://doi.org/10.1111/j.1365-294X.2011.05317.x
Cristescu ME (2014) From barcoding single individuals to metabarcoding biological communities: towards an integrative approach to the study of global biodiversity. TREE 29(10):566–571. https://doi.org/10.1016/j.tree.2014.08.001
Giampaoli S, Berti A, Di Maggio RM et al (2014) The environmental biological signature: NGS profiling for forensic comparison of soils. Forensic Sci Int 240:41–47. https://doi.org/10.1016/j.forsciint.2014.02.028
Khodakova AS, Smith RJ, Bugoyne L et al (2014) Random whole metagenomic sequencing for forensic discrimination of soils. PLoS One 9(8):e104996. https://doi.org/10.1371/journal.pone.0104996
Yang C, Wang X, Miller JA et al (2014) Using metabarcoding to ask if easily collected soil and leaf-litter samples can be used as a general biodiversity indicator. Ecol Indic 46:379–389. https://doi.org/10.1016/j.ecolind.2014.06.028
Young JM, Weyrich LS, Cooper A (2014) Forensic soil DNA analysis using high-throughput sequencing: a comparison of four molecular markers. Forensic Sci Int Genet 13:176–184. https://doi.org/10.1016/j.fsigen.2014.07.014
Orgiazzi A, Dunbar MB, Panagos et al (2015) Soil biodiversity and DNA barcodes: opportunities and challenges. Soil Biol Biochem 80:244–250. https://doi.org/10.1016/j.soilbio.2014.10.014
Jesmok EM, Hopkins JM, Foran DR (2016) Next-generation sequencing of the bacterial 16S rRNA gene for forensic soil comparison: a feasibility study. J Forensic Sci 61(3):607–617. https://doi.org/10.1111/1556-4029.13049
Burgoyne L, Koh LY, Catcheside DEA (2017) Extracting information from vehicle exteriors via soil and insect DNA. Forensic Sci Int Genet Suppl Ser 6:e593–e595. https://doi.org/10.1016/j.fsigss.2017.10.004
Meyer JM, Hoy MA (2008) Removal of fungal contaminants and their DNA from the surface of Diaphorina citri (Hemiptera: Psyllidae) prior to a molecular survey of endosymbionts. Fla Entomol 91(4):702–705. https://doi.org/10.1653/0015-4040-91.4.702
Meusnier I, Singer GAC, Landry J-F, Hickey DA, Hebert PDN, Hajibabaei M (2008) A universal DNA mini-barcode for biodiversity analysis. BMC Genomics 9(1):214. https://doi.org/10.1186/1471-2164-9-214
Levin RA, Wagner WL, Hoch PC, Nepokroeff M, Pires JC, Zimmer EA, Sytsma KJ (2003) Family-level relationships of Onagraceae based on chloroplast rbcL and ndhF data. Am J Bot 90(1):107–115. https://doi.org/10.3732/ajb.90.1.107
Little DP (2014) A DNA mini-barcode for land plants. Mol Ecol Resour 14(3):437–446. https://doi.org/10.1111/1755-0998.12194
Wojciechowski MF, Lavin M, Sanderson MJ (2004) A phylogeny of legumes (Leguminosae) based on the analysis of the plastid matK gene resolves many well-supported subclades within the family. Am J Bot 91(11):1846–1862. https://doi.org/10.3732/ajb.91.11.1846
Maurin O (2012) DNA barcoding of plants: matK primers for angiosperms. Royal Botanic Garden Edinburgh. Available at http://api.ning.com/files/3inNVyMeyJHWn4ZQeytwnpIODPXqirXGN9er*vXdBQiB1Il-SDyKKYlFEtxTQU6635m4OKaVOEWfzs8158svE8JMqemREGz3/Angiosperm_matK_RBGE_PROTOCOLv1.0.pdf. (accessed on November 21st 2017)
Schrader C, Schielke A, Ellerbroek L, Johne R (2012) PCR inhibitors—occurrence, properties and removal. J Appl Microbiol 113(5):1014–1026. https://doi.org/10.1111/j.1365-2672.2012.05384.x
Hirano M, Nakamura S, Okada M, Ueda M, Mukai R (2000) Rapid discrimination of monkey B virus from human herpes simplex viruses by PCR in the presence of betaine. J Clin Microbiol 38(3):1255–1257
Katcher HL, Schwartz I (1994) A distinctive property of Tth DNA polymerase: enzymatic amplification in the presence of phenol. BioTechniques 16(1):84–92
Sutlović D, Definis GM, Andelinovic S, Gugic D, Primorac D (2005) Taq polymerase reverse inhibition of quantitative real time polymerase chain reaction by humic acid. Croat Med J 46(4):556–562
Peist R, Honsel D, Twieling G, Löffert D (2001) PCR inhibitors in plant DNA preparations. QIAGEN News 3:7–9
Miller DN, Bryant JE, Madsen EL, Ghiorse WC (1999) Evaluation and optimization of DNA extraction and purification procedures for soil and sediment samples. Appl Environ Microb 65(11):4715–4724
Koonjul PK, Brandt WF, Farrant JM, Lindsey GG (1999) Inclusion of polyvinylpyrrolidone in the polymerase chain reaction reverses the inhibitory effects of polyphenolic contamination of RNA. Nucleic Acids Res 27(3):915–916. https://doi.org/10.1093/nar/27.3.915
Demeke T, Adams RP (1992) The effects of plant polysaccharides and buffer additives on PCR. BioTechniques 12(3):332–334
Kreader CA (1996) Relief of amplification inhibition in PCR with bovine serum albumin or T4 gene 32 protein. Appl Environ Microbiol 62(3):1102–1106
Muzzalupo I, Pisani F, Greco F, Chiappetta A (2015) Direct DNA amplification from virgin olive oil for traceability and authenticity. Eur Food Res Technol 241(1):151–155. https://doi.org/10.1007/s00217-015-2455-5
Nilsson M, Grånemo J, Buś MM et al (2016) Comparison of DNA polymerases for improved forensic analysis of challenging samples. Forensic Sci Int Genet 24:55–59. https://doi.org/10.1016/j.fsigen.2016.05.017
Ficetola GF, Coissac E, Zundel S et al (2010) An in silico approach for the evaluation of DNA barcodes. BMC Genomics 11:434. https://doi.org/10.1186/1471-2164-11-434
Zhan A, Bailey SA, Heath DD, Macisaac HJ (2014) Performance comparison of genetic markers for high-throughput sequencing-based biodiversity assessment in complex communities. Mol Ecol Resour 14(5):1049–1059. https://doi.org/10.1111/1755-0998.12254
Finley SJ, Lorenco N, Mulle J, Robertson BK, Javan GT (2016) Assessment of microbial DNA extraction methods of cadaver soil samples for criminal investigations. Aust J Forensic Sci 48(3):265–272. https://doi.org/10.1080/00450618.2015.1063690
Ribeiro RA, Lovato MB (2007) Comparative analysis of different DNA extraction protocols in fresh and herbarium specimens of the genus Dalbergia. Genet Mol Res 6(1):173–187
Li Y, Gao L-M, Poudel RC, Li D-Z, Forrest A (2011) High universality of matK primers for barcoding of gymnosperms. J Syst Evol 49(3):169–175
Soller DR, Reheis MC, Garrity CP, Van Sistine DR (2009) Map database for surficial materials in the conterminous United States: U.S. Geological Survey Data Series 425, scale 1:5,000,000. (available at http://pubs.usgs.gov/ds/425/)
Schweitzer PN (2011) Combined geologic map of the conterminous US derived from the USGS state geological map compilation. (available at mrdata.usgs.gov/geology/state/geol_poly.zip)
Acknowledgments
This is publication number 17-03 of the Laboratory Division of the Federal Bureau of Investigation. Names of commercial manufacturers are provided for information only and inclusion does not imply endorsement by the FBI or the US Government. The views expressed are those of the authors and do not necessarily reflect the official policy or position of the FBI or the US Government. This research was supported in part by an appointment to the Visiting Scientist Program at the FBI Laboratory Division, administered by the Oak Ridge Institute of Science and Education, through an interagency agreement between the US Department of Energy and the FBI. The authors thank JenaMarie Baldaino and Dr. Jack Hietpas for assistance with isolating biological fragments from forensic-type soils.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(PDF 243 kb)
Rights and permissions
About this article
Cite this article
Meiklejohn, K.A., Jackson, M.L., Stern, L.A. et al. A protocol for obtaining DNA barcodes from plant and insect fragments isolated from forensic-type soils. Int J Legal Med 132, 1515–1526 (2018). https://doi.org/10.1007/s00414-018-1772-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00414-018-1772-1