Abstract
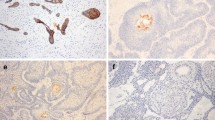
This study evaluated the expression of fibronectin, tenascin and type I collagen in ameloblastomas and adenomatoid odontogenic tumors (AOTs) aiming to contribute with the comprehension of the differences in the biological behavior of these tumors. Immunohistochemical technique was performed in 20 cases of ameloblastoma (16 solid and 4 desmoplastic) and in 10 cases of AOT. All tumors presented moderate fibronectin expression in the stroma. Solid ameloblastomas showed intense expression of fibronectin at the epithelial–mesenchymal interface, whereas desmoplastic ameloblastomas revealed no immunoexpression of fibronectin at this site. Ameloblastomas presented stronger immunoreactivity to tenascin than AOTs, especially at the epithelial–mesenchymal interface. AOTs and desmoplastic ameloblastomas showed intense labeling for type I collagen. The patterns of expression of the proteins studied agree with the locally more invasive behavior of ameloblastomas in comparison to AOTs. Our results might suggest a less invasive behavior of desmoplastic ameloblastoma in comparison to solid ameloblastoma.


Similar content being viewed by others
References
Gardner DG, Heikinheimo K, Shear M, Philipsen HP, Coleman H (2005) Ameloblastomas. In: Barnes L, Eveson JW, Reichart HP, Sidransky D (eds) World Health Organization classification of tumors. Pathology and genetics of head and neck tumours. IARC press, Lyon, pp 296–300
Philipsen HP, Reichart PA (1999) Adenomatoid odontogenic tumour: facts and figures. Oral Oncol 35:125–131. doi:10.1016/S1368-8375(98)00111-0
Philipsen HP, Nikai H (2005) Adenomatoid odontogenic tumor. In: Barnes L, Eveson JW, Reichart HP, Sidransky D (eds) World Health Organization classification of tumors. Pathology and genetics of head and neck tumours. IARC press, Lyon, pp 304–305
Kim SG, Jang HS (2001) Ameloblastoma: a clinical, radiographic, and histopathologic analysis of 71 cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 91:649–653. doi:10.1067/moe.2001.114160
Santos JN, Souza VF, Azevedo RA, Sarmento VA, Souza LB (2006) Hybrid lesion of desmoplastic and conventional ameloblastoma: immunohistochemical aspects. Rev Bras Otorrinolaringol (Engl Ed) 72:709–713
Funaoka K, Arisue M, Kobayashi I, Iizuka T, Kohgo T, Amemiya A, Totsuka Y (1996) Immunohistochemical detection of proliferating cell nuclear antigen (PCNA) in 23 cases of ameloblastoma. Eur J Cancer B Oral Oncol 32:328–332. doi:10.1016/0964-1955(96)00007-3
Kumamoto H, Ooya K (2006) Immunohistochemical detection of retinoblastoma protein and E2 promoter-binding factor-I in ameloblastomas. J Oral Pathol Med 35:183–189. doi:10.1111/j.1600-0714.2006.00381.x
Luo HY, Yu SF, Li TJ (2006) Differential expression of apoptosis-related proteins in various cellular components of ameloblastomas. Int J Oral Maxillofac Surg 35:750–755. doi:10.1016/j.ijom.2006.03.012
Sathi GS, Han PP, Tamamura R, Nagatsuka H, Hu H, Katase N, Nagai N (2007) Immunolocalization of cell signaling molecules in the granular cell ameloblastoma. J Oral Pathol Med 36:609–614
Nagai N, Yamachika E, Nishijima K, Inoue M, Shin HI, Suh MS, Nagatsuka H (1994) Immunohistochemical demonstration of tenascin and fibronectin in odontogenic and human fetal tooth germs. Eur J Cancer B Oral Oncol 30:191–195. doi:10.1016/0964-1955(94)90090-6
Mori M, Yamada T, Doi T, Ohmura H, Takai Y, Shrestha P (1995) Expression of tenascin in odontogenic tumors. Eur J Cancer B Oral Oncol 31:275–279. doi:10.1016/0964-1955(95)00002-Y
Ida-Yonemochi H, Ikarashi T, Nagata M, Hoshina H, Takagi R, Saku T (2002) The basement membrane-type heparan sulfate proteoglycan (perlecan) in ameloblastomas: its intercellular localization in stellate reticulum-like foci and biosynthesis by tumor cells in culture. Virchows Arch 441:165–173. doi:10.1007/s00428-001-0556-y
Poomsawat S, Punyasingh J, Vejchapipat P (2007) Expression of basement membrane components in odontogenic tumors. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 104:666–675. doi:10.1016/j.tripleo.2006.08.025
Matias MA, Li H, Young WG, Bartold PM (2003) Immunohistochemical localisation of extracellular matrix proteins in the periodontium during cementogenesis in the rat molar. Arch Oral Biol 48:709–716. doi:10.1016/S0003-9969(03)00131-6
Chiquet-Ehrismann R (2004) Tenascins. Int J Biochem Cell Biol 36:986–990. doi:10.1016/j.biocel.2003.12.002
Orend G (2005) Potential oncogenic action of tenascin-C in tumorigenesis. Int J Biochem Cell Biol 37:1066–1083. doi:10.1016/j.biocel.2004.12.002
Larsen M, Artym VV, Green JA, Yamada KM (2006) The matrix reorganized: extracellular matrix remodeling and integrin signaling. Curr Opin Cell Biol 18:463–471. doi:10.1016/j.ceb.2006.08.009
Orend G, Chiquet-Ehrismann R (2006) Tenascin-C induced signaling in cancer. Cancer Lett 244:143–163. doi:10.1016/j.canlet.2006.02.017
Ioachim E, Charchanti A, Briasoulis E, Karavasilis V, Tsanou H, Arvanitis DL, Agnantis NJ, Pavlidis N (2002) Immunohistochemical expression of extracellular matrix components tenascin, fibronectin, collagen type IV and laminin in breast cancer: their prognostic value and role in tumour invasion and progression. Eur J Cancer 38:2362–2370. doi:10.1016/S0959-8049(02)00210-1
Pinheiro JJV, Freitas VM, Moretti AIS, Jorge AG, Jaeger RG (2004) Local invasiveness of ameloblastoma. Role played by matrix metalloproteinases and proliferative activity. Histopathology 45:65–72. doi:10.1111/j.1365-2559.2004.01902.x
Armstrong PB, Armstrong MT (2000) Intercellular invasion and the organizational stability of tissues: a role for fibronectin. Biochim Biophys Acta 1470:O9–O20
Christgau M, Caffesse RG, Schmalz G, D’Souza RN (2007) Extracellular matrix expression and periodontal wound-healing dynamics following guided tissue regeneration therapy in canine furcation defects. J Clin Periodontol 34:691–708. doi:10.1111/j.1600-051X.2007.01097.x
Lyons AJ, Jones J (2007) Cell adhesion molecules, the extracellular matrix and oral squamous carcinoma. Int J Oral Maxillofac Surg 36:671–679. doi:10.1016/j.ijom.2007.04.002
Ziober BL, Turner MA, Palefsky JM, Banda MJ, Kramer RH (2000) Type I collagen degradation by invasive oral squamous cell carcinoma. Oral Oncol 36:365–372. doi:10.1016/S1368-8375(00)00019-1
Pilcher BK, Dumin JA, Sudbeck BD, Krane SM, Welgus HG, Parks WC (1997) The activity of collagenase-1 is required for keratinocyte migration on a type I collagen matrix. J Cell Biol 137:1445–1457. doi:10.1083/jcb.137.6.1445
Borkakoti N (2004) Matrix metalloproteinase inhibitors: design from structure. Biochem Soc Trans 32:17–20. doi:10.1042/BST0320017
Kadler KE, Baldock C, Bella J, Boot-Handford RP (2007) Collagens at a glance. J Cell Sci 120:1955–1958. doi:10.1242/jcs.03453
Heikinheimo K, Morgan PR, Happonen RP, Stenman G, Virtanen I (1991) Distribution of extracellular matrix proteins in odontogenic tumours and developing teeth. Virchows Arch B Cell Pathol Incl Mol Pathol 61:101–109. doi:10.1007/BF02890411
Oliveira MDC, Miranda JL, Amorim RFB, Souza LB, Freitas RA (2004) Tenascin and fibronectin expression in odontogenic cysts. J Oral Pathol Med 33:354–359
Murata M, Cheng J, Horino K, Hara K, Shimokawa H, Saku T (2000) Enamel proteins and extracellular matrix molecules are co-localized in the pseudocystic stromal space of adenomatoid odontogenic tumor. J Oral Pathol Med 29:483–490. doi:10.1034/j.1600-0714.2000.291002.x
Eversole I, Leider AS, Hansen LS (1984) Ameloblastoma with pronounced stromal desmoplasia. J Oral Maxillofac Surg 42:735–740. doi:10.1016/0278-2391(84)90423-3
Andrade ESS, Miguel MCC, Pinto LP, Souza LB (2007) Ameloblastoma and adenomatoid odontogenic tumor: the role of α2β1, α3β1 and α5β1 integrins in local invasiveness and architectural characteristics. Ann Diagn Pathol 11:199–205. doi:10.1016/j.anndiagpath.2006.04.005
Andrade ESS, Miguel MCC, Freitas RA, Pinto LP, Souza LB (2008) Immunoexpression of integrins in ameloblastoma, adenomatoid odontogenic tumor, and human tooth germs. Int J Surg Pathol 16:277–285. doi:10.1177/1066896907306084
Philipsen HP, Reichart PA, Takata T (2001) Desmoplastic ameloblastoma (including “hybrid” lesion of ameloblastoma). Biological profile based on 100 cases from the literature and own cases. Oral Oncol 37:455–460. doi:10.1016/S1368-8375(00)00111-1
Delzangles G, Boy-Lefevre ML, Forest N (1997) Glycoproteins expression in apical pathologic tissues: clinical incidences. J Endod 23:965–968. doi:10.1016/S0099-2399(06)81122-2
Takata T, Miyauchi M, Ogawa I, Kudo Y, Takekoshi T, Zhao M, Sato S, Nikai H, Komiyama K (2000) Immunoexpression of transforming growth factor β in desmoplastic ameloblastoma. Virchows Arch 436:319–323. doi:10.1007/s004280050453
Conflict of interest statement
We inform that we do not have a financial relationship with the organization that sponsored the research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Medeiros, A.M.C., Nonaka, C.F.W., Galvão, H.C. et al. Expression of extracellular matrix proteins in ameloblastomas and adenomatoid odontogenic tumors. Eur Arch Otorhinolaryngol 267, 303–310 (2010). https://doi.org/10.1007/s00405-009-0996-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-009-0996-6