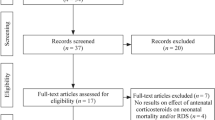
Abstract
Purpose
To critically analyse the literature on the antenatal corticosteroids (ACS)-to-birth interval from a causal point of view and to present a solution to the problem of bias caused by post hoc analysis.
Methods
Due to the post hoc nature of the ACS-to-birth interval, a randomised controlled trial (RCT) of ACS versus placebo is not able to examine the importance of the interval. When an RCT is not feasible, for whatsoever reason, a target trial can be set up and an attempt can be made to answer the causal question of interest using observational data. An attempt was made to set up a target trial which could enable to examine the causal effect of the ACS-to-birth interval on neonatal outcomes. An analysis of current literature on the ACS-to-birth interval was done.
Results
The majority of studies aimed to examine the causal effect of the interval, but their study design only permitted to find associations between the interval and neonatal outcomes. Barriers for setting up a target trial are highlighted.
Conclusion
Evidence on the superiority of any ACS-to-birth interval is lacking and the question can only be addressed causally and become clinically relevant if baseline randomisation to ACS-to-birth intervals is made possible.

Similar content being viewed by others
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Liggins GC, Howie RN (1972) A controlled trial of antepartum glucocorticoid treatment for prevention of the respiratory distress syndrome in premature infants. Pediatrics 50(4):515–525
WHO recommendations on interventions to improve preterm birth outcomes. https://apps.who.int/iris/bitstream/handle/10665/183037/9789241508988_eng.pd?sequence=1. Retrieved 4 Feb 2019.
Crowley P (1995) Antenatal corticosteroid therapy: a meta-analysis of the randomized trials, 1972 to 1994. Am J Obstet Gynecol 173(1):322–335
Crowther C, McKinlay C, Middleton P, Harding J (2015) Repeat doses of prenatal corticosteroids for women at risk of preterm birth for improving neonatal health outcomes. Cochrane Database Syst Rev 7:CD003935
Bruijn M, Vis J, Wilms F, Oudijk M, Kwee A, Porath M et al (2016) Comparison of the Actim Partus test and the fetal fibronectin test in the prediction of spontaneous preterm birth in symptomatic women undergoing cervical length measurement. Eur J Obstet Gynecol Reprod Biol 206:220–224
Watson H, Carter J, Seed P, Tribe R, Shennan A (2017) The QuiPP App: a safe alternative to a treat-all strategy for threatened preterm labor. Ultrasound Obstet Gynecol 50(3):342–346
Demirci O, Ünal A, Demirci E, Sözen H, Akdemir Y, Boybek E et al (2011) Sonographic measurement of cervical length and risk of preterm birth. J Obstet Gynaecol Res 37(7):809–814
Crowther C, Aghajafari F, Askie L, Asztalos E, Brocklehurst P, Bubner T et al (2012) Repeat prenatal corticosteroid prior to preterm birth: a systematic review and individual participant data meta-analysis for the PRECISE study group (prenatal repeat corticosteroid international IPD study group: assessing the effects ousing the best level of evidence)—study protocol. Syst Rev 1:12
Crowther C, Middleton P, Voysey M, Askie L, Zhang S, Martlow T et al (2019) Effects of repeat prenatal corticosteroids given to women at risk of preterm birth: an individual participant data meta-analysis. PLoS Med 16(4):e1002771
Melamed N, Asztalos E, Murphy K, Zaltz A, Redelmeier D, Shah B, Barret J (2019) Neurodevelopmental disorders among term infants exposed to antenatal corticosteroids during pregnancy: a population-based study. BMJ Open 9:e031197. https://doi.org/10.1136/bmjopen-2019-031197
Parnell L, Ayuk P (2020) Prophylactic antenatal corticosteroids for fetal lung maturity: known unknowns and unknown unknowns. Case Rep Womens Health 28:e00242. https://doi.org/10.1016/j.crwh.2020.e00242
Murphy D, Caukwell S, Joels L, Wardle P (2002) Cohort study of the neonatal outcome of twin pregnancies that were treated with prophylactic or rescue antenatal corticosteroids. Am J Obstet Gynecol 187(2):483–488
Jobe A, Goldenberg G (2018) Antenatal corticosteroids: an assessment of anticipated benefits and potential risks. Am J Obstet Gynecol 219(1):62–74
Espinoza J, Kusanovic JP, Kim CJ, Kim YM, Kim JS, Hassan S et al (2007) An episode of preterm labor is a risk factor for the birth of a small-for-gestational-age neonate. Am J Obstet Gynecol 196:574.e1-574.e6
Paules C, Pueyo V, Marti E, de Vilchez S, Burd I, Calvo P, Oros D (2017) Threatened preterm labor is a risk factor for impaired cognitive development in early childhood. Am J Obstet Gynecol 216:157.e1–7
Romero R, Erez O, Maymon E, Pacora P (2017) Is an episode of suspected preterm labor that subsequently leads to a term delivery benign? Am J Obstet Gynecol 216:89–94
Sterne J, Savović J, Page M, Elbers R, Blencowe N, Boutron I et al (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366:l4898. https://doi.org/10.1136/bmj.l4898
Hernán M (2018) The C-Word: scientific euphemisms do not improve causal inference from observational data. Am J Public Health 108:616–619
Hernán M, Robins J (2016) Using big data to emulate a target trial when a randomized trial is not available. Am J Epidemiol 183(8):758–764
Prosperi M, Guo Y, Sperrin M, Koopman J, Min J, He X et al (2020) Causal inference and counterfactual prediction in machine learning for actionable healthcare. Nat Mach Intell 2:369–375
Hernán M, Robins J (2020) Causal inference. What if. CRC Press, Taylor & Francis Group A, Chapman & Hall book. https://cdn1.sph.harvard.edu/wp-content/uploads/sites/1268/2019/02/hernanrobins_v1.10.38.pdf. Retrieved 30 Mar 2020.
Hernán M, Taubman S (2008) Does obesity shorten life? The importance of well-defined interventions to answer causal questions. Int J Obest (Lond) 32:S8–S14
Hernán M, Sauer B, Hernandez-Dias S, Platt R, Shrier I (2016) Specifying a target trial prevents immortal time bias and other self-inflicted injuries in observational analyses. J Clin Epidemiol 79:70–75
Schutte M, Treffers P, Koppe J (1983) Threatened preterm labor: the influence of time factors on the incidence of respiratory distress syndrome. Obstet Gynecol 146(4):395–400
McNamara M, Bottoms S (1998) The incidence of respiratory distress syndrome does not increase when preterm delivery occurs greater than 7 days after steroid administration. Aust N Z J Obstet Gynaecol 38(1):8–10
Vemillion S, Soper D, Newman R (2001) Is betamethasone effective longer than 7 days after treatment? Obstet Gynecol 97(4):491–493
Smrcek J, Schwarau N, Kohl M, Berg C, Geipel A, Krapp M (2005) Antenatal corticosteroid therapy in premature infants. Arch Gynecol Obstet 271:26–32
Sehdev H, Abbasi S, Robertson P, Fischer L, Marchiano D, Gerdes J et al (2004) The effects of the time interval from antenatal corticosteroid exposure to delivery on neonatal outcome of very low birth weight infants. Am J Obstet Gynecol 191:1409–1413
Peaceman A, Bajaj K, Kumar P, Grobman W (2005) The interval between a single course of antenatal steroids and delivery and its association with neonatal outcomes. Am J Obstet Gynecol 193:1165–1169
Ring A, Garland J, Stafeil B, Carr M, Peckman G, Pircon R (2007) The effect of a prolonged time between antenatal corticosteroid administration and delivery on outcomes in preterm neonates: a cohort study. Am J Obstet Gynecol 196(5):457.e1–6
Ferguson S, Allen V, Craig C, Allen A, Dodds L (2009) Timing of indicated delivery after antenatal steroids in preterm pregnancies with severe hypertension. Hypertens Pregnancy 28:63–75
Waters T, Mercer B (2009) Impact of timing of antenatal corticosteroid exposure on neonatal outcomes. J Matern Fetal Neonatal Med 22(4):311–314
Wilms F, Vis Y, Pattinaja D, Duin R, Stam M, Reuvers J et al (2011) Relationship between the time interval from antenatal corticosteroid administration until preterm birth and the occurence of respiratory morbidity. Am J Obstet Gynecol 205:e1-7
Kuk J-Y, An J-J, Cha H-H, Choi S-J, Vargas J, Oh S-y et al (2013) Optimal time interval between a single course of antenatal corticosteroids and delivery for reduction of respiratory distress syndrome in preterm twins. Am J Obstet Gynecol 209:256.e1–7
Melamed N, Shah J, Soraisham A, Yoon E, Lee S, Shah P et al (2015) Association between antenatal corticosteroid administration-to-birth interval and outcomes of preterm neonates. Obstet Gynecol 125:1377–1384
Kosinska-Kazzynska K, Szymusik I, Urban P, Zachara M, Wielgos M (2016) Relation between time interval from antenatal corticosteroids administration to delivery and neonatal outcomes in twins. J Obstet Gynaecol Res 42(6):625–631
Liebowitz M, Clyman R (2016) Antenatal betamethasone: a prolonged time interval from administration to delivery is associated with an increased incidence of severe intraventricular hemorrhage in infants born before 28 weeks gestation. J Pediatr 177:114–120
Fuller K, DeGroff S, Borgida A (2017) Neonatal outcomes based on antenatal corticosteroid exposure time for infants delivered between 23 and 34 weeks’ gestation. Clin Exp Obstet Gynecol 44(2):252–258
Yasuhi I, Myoga M, Suga S, Sugimi S, Umezaki Y, Fukuda M et al (2017) Influence of the interval between antenatal corticosteroid therapy and delivery on respiratory distress syndrome. J Obstet Gynaecol Res 43(3):486–491
Norman M, Piedvache A, Borch K, Huusom LD, Edstedt Bonamy A-K, Howell E et al (2017) Association of short antenatal corticosteroid administration-to-birth intervals with survival and morbidity among very preterm infants. JAMA Pediatr 171(7):678–686
Norberg H, Kowalski J, Marsal K, Norman M (2017) Timing of antenatal corticosteroid administration and survival in extremely preterm infants: a national population-based cohort study. BJOG 124:1567–1574
Lau H, Tung J, Wong T, Tan P, Tagore S (2017) Timing of antenatal steroids exposure and its effects on neonates. Arch Gynecol Obstet 296:1091–1096
Frändberg J, Sandblom J, Bruschettini M, Marsal K, Kristensen K (2018) Antenatal corticosteroids: a retrospective cohort study on timing, indications and neonatal outcome. Acta Obstet Gynecol Scand 97:591–597
Gates S, Brocklehurst P (2007) Decline in effectiveness of antenatal corticosteroids with time to birth: real or artefact. BMJ 335:77–79
Field A, Miles J, Field Z (2012) Discovering statistics in R 2. SAGE Publications
Naggara O, Raymond J, Guilbert F, Altman D (2011) The problem of subgroup analyses: an example from a trial on ruptured intracranial aneurysms. Am J Neuroradiol 32:633–636
Hirji K, Fagerland M (2009) Outcome based subgroup analysis: a neglected concern. Trials 10:33. https://doi.org/10.1186/1745-6215-10-33
Desai M, Pieper K, Mahaffey K (2014) Challenges and solutions to pre- and post-randomization subgroup analyses. Curr Cardiol Rep 16:531
Snowden J, Basso O (2017) Causal inference in studies in preterm babies: a simulation study. BJOG 125(6):686–692. https://doi.org/10.1111/1471-0528.14942
VanderWeele T, Mumford S, Schisterman E (2013) Conditioning on intermediates in perinatal epidemiology. Epidemiology 23(1):1–9
Sterne J, Hernán M, Reeves B, Savovic J, Berkman N, Viswanathan M et al (2016) ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355:i4949. https://doi.org/10.1136/bmj.i4919
Acknowledgements
This article is part of the PhD project of Isabelle Dehaene, called “PREdiction tool for preTerm laboUr and Neonatal outcome” (PRETURN). We would like to thank all other researchers involved in PRETURN, in particular Johan Steen, statistician at Ghent University and Ghent University Hospital, Belgium. He is a specialist in causal inference with a special interest in time-varying treatments. This work would not have been possible without his input.
Funding
Isabelle Dehaene is funded by a scholarship of FWO (1700520N). The funding body played no role in the creation of this manuscript.
Author information
Authors and Affiliations
Contributions
ID: conceptualization, writing—original draft, writing—review and editing; KR: writing—review and editing, supervision; KS: writing—review and editing, supervision; JD: writing—review and editing, supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Not applicable.
Consent to participate/for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dehaene, I., Roelens, K., Smets, K. et al. Relevance of the antenatal corticosteroids-to-delivery interval in the prevention of neonatal respiratory distress syndrome through the eyes of causal inference: a review and target trial. Arch Gynecol Obstet 305, 885–892 (2022). https://doi.org/10.1007/s00404-021-06213-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-021-06213-5