Abstract
Purpose
We investigated the association between serum ionized calcium and prognosis of EOC and determined the optimal cutoff value of ionized calcium level to predict the prognosis of EOC.
Methods
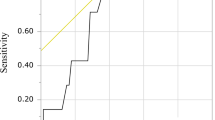
The medical records of patients who were newly diagnosed with EOC from 2001 to 2016 were retrieved. Preoperative ionized calcium test was performed within 2 weeks before surgery, and the cutoff of high normocalcemia was defined based on the receiver operating characteristic (ROC) curve for recurrence. Cox proportional hazards regression models were used to identify independent prognostic factors for progression-free survival (PFS).
Results
From 2001 to 2016, 83 patients diagnosed with EOC were identified at a single institution. The optimal cutoff value was set to 4.7 mg/dL (high normocalcemia vs. control group) by plotting the ROC curve for recurrence. Stages III/IV were more frequent in high normocalcemia, with borderline significance (72.9% vs. 52.2%, p = 0.053). Recurrence (67.6% vs. 43.5%, p = 0.029) and death (46.0% vs. 15.2%, p < 0.01) were significantly more frequent in the high normocalcemia group. In multivariate analysis, high normocalcemia (HR 1.9, 95% CI 1.03–3.61, p = 0.04), age (HR 1.04, 95% CI 1.01–1.08, p = 0.02), stage (HR 3.67, 95% CI 1.13–11.92, p = 0.03), residual tumor > 1 cm (HR 3.79, 95% CI 1.61–8.95, p < 0.01), and lymph node metastasis (HR 2.46, 95% CI 1.27–4.78, p < 0.01) were independent risk factors for recurrence.
Conclusion
This study showed positive association between relatively high level of ionized calcium level and recurrence risk of EOC. High normocalcemia showed the potential as a biomarker for prognosis of EOC.



Similar content being viewed by others
References
Webb PM, Jordan SJ (2017) Epidemiology of epithelial ovarian cancer. Best Pract Res Clin Obstet Gynaecol 41:3–14. https://doi.org/10.1016/j.bpobgyn.2016.08.006
Goodman MT, Correa CN, Tung KH, Roffers SD, Cheng WuX, Young JL Jr, Wilkens LR, Carney ME, Howe HL (2003) Stage at diagnosis of ovarian cancer in the United States, 1992–1997. Cancer 97:2648–2659. https://doi.org/10.1002/cncr.11347
Kelly MG, Winkler SS, Lentz SS, Berliner SH, Swain MF, Skinner HG, Schwartz GG (2015) Serum calcium and serum albumin are biomarkers that can discriminate malignant from benign pelvic masses. Cancer Epidemiol Biomarkers Prev 24:1593–1598. https://doi.org/10.1158/1055-9965.Epi-15-0443
Stewart SL, Rim SH, Richards TB (2011) Gynecologic oncologists and ovarian cancer treatment: avenues for improved survival. J Womens Health (Larchmt) 20:1257–1260. https://doi.org/10.1089/jwh.2011.3053
So KA, Hong JH, Jin HM, Kim JW, Song JY, Lee JK, Lee NW (2014) The prognostic significance of preoperative leukocytosis in epithelial ovarian carcinoma: a retrospective cohort study. Gynecol Oncol 132:551–555. https://doi.org/10.1016/j.ygyno.2014.01.010
Zhang W, Ye B, Liang W, Ren Y (2017) Preoperative prognostic nutritional index is a powerful predictor of prognosis in patients with stage III ovarian cancer. Sci Rep 7:9548. https://doi.org/10.1038/s41598-017-10328-8
Goldner W (2016) Cancer-related hypercalcemia. J Oncol Pract 12:426–432. https://doi.org/10.1200/jop.2016.011155
Gellhorn A, Plimpton CH (1956) Hypercalcemia in malignant disease without evidence of bone destruction. Am J Med 21:750–759
Schwartz GG, Skinner HG (2013) Prospective studies of total and ionized serum calcium in relation to incident and fatal ovarian cancer. Gynecol Oncol 129:169–172. https://doi.org/10.1016/j.ygyno.2012.12.046
Schwartz GG, Tretli S, Vos L, Robsahm TE (2017) Prediagnostic serum calcium and albumin and ovarian cancer: a nested case–control study in the Norwegian Janus Serum Bank Cohort. Cancer Epidemiol 49:225–230. https://doi.org/10.1016/j.canep.2017.07.004
Toriola AT, Surcel HM, Calypse A, Grankvist K, Luostarinen T, Lukanova A, Pukkala E, Lehtinen M (2010) Independent and joint effects of serum 25-hydroxyvitamin D and calcium on ovarian cancer risk: a prospective nested case–control study. Eur J Cancer 46:2799–2805. https://doi.org/10.1016/j.ejca.2010.05.019
Song X, Li Z, Ji X, Zhang D (2017) Calcium intake and the risk of ovarian cancer: a meta-analysis. Nutrients 9:679. https://doi.org/10.3390/nu9070679
Qin B, Moorman PG, Alberg AJ et al (2016) Dairy, calcium, vitamin D and ovarian cancer risk in African–American women. Br J Cancer 115:1122–1130. https://doi.org/10.1038/bjc.2016.289
Hobson SA, McNeil SE, Lee F, Rodland KD (2000) Signal transduction mechanisms linking increased extracellular calcium to proliferation in ovarian surface epithelial cells. Exp Cell Res 258:1–11. https://doi.org/10.1006/excr.2000.4910
Tennakoon S, Aggarwal A, Kállay E (2016) The calcium-sensing receptor and the hallmarks of cancer. Biochim Biophys Acta 1863:1398–1407. https://doi.org/10.1016/j.bbamcr.2015.11.017
Akino K, Ohtsuru A, Kanda K, Yasuda A, Yamamoto T, Akino Y, Naito S, Kurokawa M, Iwahori N, Yamashita S (2000) Parathyroid hormone-related peptide is a potent tumor angiogenic factor. Endocrinology 141:4313–4316. https://doi.org/10.1210/endo.141.11.7875
Hoey RP, Sanderson C, Iddon J, Brady G, Bundred NJ, Anderson NG (2003) The parathyroid hormone-related protein receptor is expressed in breast cancer bone metastases and promotes autocrine proliferation in breast carcinoma cells. Br J Cancer 88:567–573. https://doi.org/10.1038/sj.bjc.6600757
Iwamura M, Hellman J, Cockett AT, Lilja H, Gershagen S (1996) Alteration of the hormonal bioactivity of parathyroid hormone-related protein (PTHrP) as a result of limited proteolysis by prostate-specific antigen. Urology 48:317–325. https://doi.org/10.1016/s0090-4295(96)00182-3
Powell GJ, Southby J, Danks JA, Stillwell RG, Hayman JA, Henderson MA, Bennett RC, Martin TJ (1991) Localization of parathyroid hormone-related protein in breast cancer metastases: increased incidence in bone compared with other sites. Cancer Res 51:3059–3061
Nishihara M, Kanematsu T, Taguchi T, Razzaque MS (2007) PTHrP and tumorigenesis: is there a role in prognosis? Ann N Y Acad Sci 1117:385–392. https://doi.org/10.1196/annals.1402.046
Shen X, Mula RV, Evers BM, Falzon M (2007) Increased cell survival, migration, invasion, and Akt expression in PTHrP-overexpressing LoVo colon cancer cell lines. Regul Pept 141:61–72. https://doi.org/10.1016/j.regpep.2006.12.017
Singh RJ, Kumar R (2003) Fibroblast growth factor 23 concentrations in humoral hypercalcemia of malignancy and hyperparathyroidism. Mayo Clin Proc 78:826–829. https://doi.org/10.4065/78.7.826
Tebben PJ, Kalli KR, Cliby WA, Hartmann LC, Grande JP, Singh RJ, Kumar R (2005) Elevated fibroblast growth factor 23 in women with malignant ovarian tumors. Mayo Clin Proc 80:745–751. https://doi.org/10.1016/s0025-6196(11)61528-0
Mansinho A, Ferreira AR, Casimiro S et al (2019) Levels of circulating fibroblast growth factor 23 (FGF23) and prognosis in cancer patients with bone metastases. Int J Mol Sci 20:695. https://doi.org/10.3390/ijms20030695
Asher V, Lee J, Bali A (2012) Preoperative serum albumin is an independent prognostic predictor of survival in ovarian cancer. Med Oncol 29:2005–2009. https://doi.org/10.1007/s12032-011-0019-5
Parker D, Bradley C, Bogle SM, Lay J, Masood M, Hancock AK, Naylor B, Price JJ (1994) Serum albumin and CA125 are powerful predictors of survival in epithelial ovarian cancer. Br J Obstet Gynaecol 101:888–893
Maier JD, Levine SN (2015) Hypercalcemia in the intensive care unit: a review of pathophysiology, diagnosis, and modern therapy. J Intensive Care Med 30:235–252. https://doi.org/10.1177/0885066613507530
Ong GS, Walsh JP, Stuckey BG, Brown SJ, Rossi E, Ng JL, Nguyen HH, Kent GN, Lim EM (2012) The importance of measuring ionized calcium in characterizing calcium status and diagnosing primary hyperparathyroidism. J Clin Endocrinol Metab 97:3138–3145. https://doi.org/10.1210/jc.2012-1429
Sorva A, Elfving S, Pohja P, Tilvis RS (1988) Assessment of calcaemic status in geriatric hospital patients: serum ionized calcium versus albumin-adjusted total calcium. Scand J Clin Lab Invest 48:489–494
Young RH, Oliva E, Scully RE (1994) Small cell carcinoma of the ovary, hypercalcemic type. A clinicopathological analysis of 150 cases. Am J Surg Pathol 18:1102–1116
Piura B (2008) Hypercalcemia in malignancies of the female genital tract. Harefuah 147(229–234):277
Acknowledgements
The authors would like to thank all the members of Gynecological Cancer Research Institute in Korea University. This study was sponsored by Hanmi Pharm Co., Ltd., Republic of Korea.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
HWC summarized available literature and drafted the paper. JHH and YTO collected and analyzed the clinical data. JKL and HWC edited the paper and supervised the research group. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
This study was approved by the Institutional Review Boards of Korea University Medical Center (KUGH17256). Institutional Review Boards of Korea University Medical Center allows the use of medical record without informed consent where data are fully anonymized.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Data availability
Not applicable.
Code availability
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cho, HW., Ouh, YT., Hong, J.H. et al. Exploring the prognostic significance of preoperative high normocalcemia in epithelial ovarian carcinoma. Arch Gynecol Obstet 303, 803–810 (2021). https://doi.org/10.1007/s00404-020-05834-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-020-05834-6