Abstract
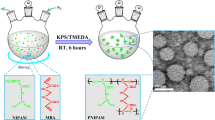
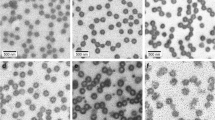
Thermosensitive poly(N-isopropylacrylamide-co-methacrylic acid) (poly(NIPAM-co-MAA)) microgels were prepared via semi-batch free radical copolymerization in which the functional monomer (methacrylic acid) was continuously fed into the reaction vessel at various speeds. Microgels with the same bulk MAA contents (and thus the same overall compositions) but different radial functional group distributions were produced, with batch copolymerizations resulting in core-localized functional groups, fast-feed semi-batch copolymerizations resulting in near-uniform functional group distributions, and slow-feed semi-batch copolymerizations resulting in shell-localized functional groups. Functional group distributions in the microgels were probed using titration analysis, electrophoresis, and transmission electron microscopy. The induced functional group distributions have particularly significant impacts on the pH-induced swelling and cationic drug binding behavior of the microgels; slower monomer feeds result in increased pH-induced swelling but lower drug binding. This work suggests that continuous semi-batch feed regimes can be used to synthesize thermoresponsive microgels with well-defined internal morphologies if an understanding of the relative copolymerization kinetics of each comonomer relative to NIPAM is achieved.







Similar content being viewed by others
References
Pelton R (2000) Temperature-sensitive aqueous microgels. Adv Colloid Interface Sci 85(1):1–33
Snowden MJ, Chowdhry BZ, Vincent B, Morris GE (1996) Colloidal copolymer microgels of N-isopropylacrylamide and acrylic acid: pH, ionic strength and temperature effects. J Chem Soc-Faraday Trans 92(24):5013–5016
Tan BH, Ravi P, Tam KC (2006) Synthesis and characterization of novel pH-responsive polyampholyte microgels. Macromol Rapid Commun 27:522–528
Liu R, Milani AH, Saunders JM, Freemont TJ, Saunders BR (2011) Tuning the swelling and mechanical properties of pH-responsive doubly crosslinked microgels using particle composition. Soft Matter 7:9297–9306
Tan BH, Tam KC, Lam YC, Tan CB (2005) Microstructure and rheological properties of pH-responsive core-shell particles. Polymer 46:10066–10076
Pelton RH, Chibante P (1986) Preparation of aqueous lattices with N-isopropylacrylamide. Colloids Surf 20(3):247–256
Lu Y, Ballauff M (2011) Thermosensitive core-shell microgels: from colloidal model systems to nanoreactors. Prog Polym Sci 36:767–792
Lapeyre V, Gosse I, Chevreux S, Ravaine V (2006) Monodispersed glucose-responsive microgels operating at physiological salinity. Biomacromolecules 7(12):3356–3363
Hoare T, Pelton R (2008) Charge-switching, amphoteric glucose-responsive microgels with physiological swelling activity. Biomacromolecules 9(2):733–740
Zhao Y, He J, Yan B, Tremblay L (2011) Both core- and shell-cross-linked nanogels: photoinduced size change, intraparticle LCST, and interparticle UCST thermal behaviors. Langmuir 27(1):436–444
Das M, Zhang H, Kumacheva E (2006) Microgels: old materials with new applications. Ann Rev Mat Res 36:117–142
Lu Y, Ballauff M (2007) “Smart” nanoparticles: preparation, characterization and applications. Polymer 48(7):1815–1823
Lyon LA, Meng ZY, Singh N, Sorrell CD, John AS (2009) Thermoresponsive microgel-based materials. Chem Soc Rev 38(4):865–874
Oh JK, Drumright R, Siegwart DJ, Matyjaszewski K (2008) The development of microgels/nanogels for drug delivery applications. Prog Polym Sci 33(4):448–477
Saunders BR, Laajam N, Daly E, Teow S, Hu XH, Stepto R (2009) Microgels: from responsive polymer colloids to biomaterials. Adv Colloid Interface Sci 147–48:251–262
Vinogradov SV (2006) Colloidal microgels in drug delivery applications. Curr Pharm Design 12(36):4703–4712
Hoare T, Pelton R (2004) Highly pH and temperature responsive microgels functionalized with vinylacetic acid. Macromolecules 37(7):2544–2550
Hoare T, Pelton R (2005) Electrophoresis of functionalized microgels: morphological insights. Polymer 46(4):1139–1150
Hoare T, Pelton R (2006) Titrametric characterization of pH-induced phase transitions in functionalized microgels. Langmuir 22(17):7342–7350
Hoare T, Pelton R (2007) Calorimetric analysis of thermal phase transitions in functionalized microgels. J Phys Chem B 111(6):1334–1342
Hoare T, Pelton R (2008) Impact of microgel morphology on functionalized microgel-drug interactions. Langmuir 24(3):1005–1012
Sahiner N, Ozay O, Aktas N (2011) 4-Vinylpyridine-based smart nanoparticles with N-isopropylacrylamide, 2-hydroxyethyl methacrylate, acrylic acid, and methacrylic acid for potential biomedical applications. Curr Nanosci 7(3):453–462
Bradley M, Vincent B (2008) Poly(vinylpyridine) core/poly(N-isopropylacrylamide) shell microgel particles: their characterization and the uptake and release of an anionic surfactant. Langmuir 24(6):2421–2425
Liu WJ, Huang YM, Liu HL (2007) Preparation and characterization of temperature and pH responsive core-shell microgel. Acta Chim Sinica 65(2):91–94
Richtering W, Kleinen J, Klee A (2010) Influence of architecture on the interaction of negatively charged multisensitive poly(N-isopropylacrylamide)-co-methacrylic acid microgels with oppositely charged polyelectrolyte: absorption vs adsorption. Langmuir 26(13):11258–11265
Bradley M, Vincent B, Burnett G (2007) Uptake and release of anionic surfactant into and from cationic core-shell microgel particles. Langmuir 23(18):9237–9241
Gan DJ, Lyon LA (2001) Tunable swelling kinetics in core-shell hydrogel nanoparticles. J Am Chem Soc 123(31):7511–7517
Gan DJ, Lyon LA (2003) Fluorescence nonradiative energy transfer analysis of crosslinker heterogeneity in core-shell hydrogel nanoparticles. Anal Chim Acta 496(1–2):53–63
Berndt I, Richtering W (2003) Doubly temperature sensitive core-shell microgels. Macromolecules 36(23):8780–8785
Richtering W, Scherzinger C, Lindner P, Keerl M (2010) Cononsolvency of poly(N,N-diethylacrylamide) (PDEAAM) and poly(N-isopropylacrylamide) (PNIPAM) based microgels in water/methanol mixtures: copolymer vs core-shell microgel. Macromolecules 43(16):6829–6833
Hu ZB, Chi CL, Cai T (2009) Oligo(ethylene glycol)-based thermoresponsive core-shell microgels. Langmuir 25(6):3814–3819
Suzuki D, Yoshida R (2010) Self-oscillating core/shell microgels: effect of a crosslinked nanoshell on autonomous oscillation of the core. Polym J 42(6):501–508
Lyon LA, Suzuki D, McGrath JG, Kawaguchi H (2007) Colloidal crystals of thermosensitive, core/shell hybrid microgels. J Phys Chem C 111(15):5667–5672
Kuckling D, Vo CD, Wohlrab SE (2002) Preparation of nanogels with temperature-responsive core and pH-responsive arms by photo-cross-linking. Langmuir 18(11):4263–4269
Huang YM, Liu WJ, Liu HL, Hu Y (2007) Composite structure of temperature sensitive chitosan microgel and anomalous behavior in alcohol solutions. J Colloid Interface Sci 313(1):117–121
Wang PX, Chen Q, Xu K, Zhang WD, Song CL (2009) Preparation and characterization of poly(N-isopropylacrylamide)/polyvinylamine core-shell microgels. Colloid Polym Sci 287(11):1339–1346
Wu C, Hu TJ, You YZ, Pan CY (2002) The coil-to-globule-to-brush transition of linear thermally sensitive poly(N-isopropylacrylamide) chains grafted on a spherical microgel. J Phys Chem B 106(26):6659–6662
Charleux B, Rieger J, Grazon C, Alaimo D, Jerome C (2009) Pegylated thermally responsive block copolymer micelles and nanogels via in situ RAFT aqueous dispersion polymerization. J Polym Sci Pol Chem 47(9):2373–2390
Nagasaki Y, Oishi M (2007) Synthesis, characterization, and biomedical applications of core-shell-type stimuli-responsive nanogels—nanogel composed of poly[2-(N,N-diethylamino)ethyl methacrylate] core and PEG tethered chains. React Funct Polym 67(11):1311–1329
Thurecht KJ, Zheng Y, Turner W, Zong MM, Irvine DJ, Howdle SM (2011) Biodegradable core-shell materials via RAFT and ROP: characterization and comparison of hyperbranched and microgel particles. Macromolecules 44(6):1347–1354
Seiffert S, Thiele J, Abate AR, Weitz DA (2010) Smart microgel capsules from macromolecular precursors. J Am Chem Soc 132(18):6606–6609
Hoare T, McLean D (2006) Kinetic prediction of functional group distributions in thermosensitive microgels. J Phys Chem B 110(41):20327–20336
Hoare T, McLean D (2006) Multi-component kinetic modeling for controlling local compositions in thermosensitive polymers. Macromol Theory Simul 15(8):619–632
Hoare T, Pelton R (2007) Functionalized microgel swelling: comparing theory and experiment. J Phys Chem B 111(41):11895–11906
Hoare T, Pelton R (2007) Engineering glucose swelling responses in poly(N-isopropylacrylamide)-based microgels. Macromolecules 40(3):670–678
Acciaro R, Gilányi T, Varga I (2011) Preparation of monodisperse poly(N-isopropylacrylamide) microgel particles with homogenous cross-link density distribution. Langmuir 27(12):7917–7925. doi:10.1021/la2010387
Zha LS, Zhang QS, Ma JH, Liang BR (2009) A novel route to prepare pH- and temperature-sensitive nanogels via a semibatch process. J Colloid Interf Sci 330(2):330–336
Forcada J, Imaz A, Miranda JI, Ramos J (2008) Evidences of a hydrolysis process in the synthesis of N-vinylcaprolactam-based microgels. Eur Polym J 44(12):4002–4011
Sun GX, Zhang MZ, Xu Y, Lu YM, Ni PH (2009) Synthesis and properties of pH-responsive cationic microgels. Acta Chim Sinica 67(14):1685–1690
Ponratnam S, Kapur SL (1977) Reactivity ratios of ionizing monomers in aqueous-solution—copolymerization of acrylic and methacrylic acids with acrylamide. Makromol Chem 178(4):1029–1038
Brandrup J, Immergut EH, Grulke EA (eds) (1999) Polymer handbook, 4th edn. Wiley, New York
Ohshima H (1994) Electrophoretic mobility of soft particles. J Colloid Interface Sci 163(2):474–483
Elmas B, Tuncel M, Senel S, Patir S, Tuncel A (2007) Hydroxyl functionalized thermosensitive microgels with quadratic crosslinking density distribution. J Colloid Interface Sci 313(1):174–183
Fernandez-Barbero A, Fernandez-Nieves A, Grillo I, Lopez-Cabarcos E (2002) Structural modifications in the swelling of inhomogeneous microgels by light and neutron scattering. Phys Rev E 66(5):051803
Stieger M, Richtering W, Pedersen JS, Lindner P (2004) Small-angle neutron scattering study of structural changes in temperature sensitive microgel colloids. J Chem Phys 120(13):6197–6206
Acknowledgments
The Natural Sciences and Engineering Research Council of Canada (NSERC) is acknowledged for funding. Kevin DeFrance is acknowledged for his assistance in synthesizing the semi-batch microgels.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Information
Excess Gibbs free energy of ionization and degree of ionization versus pH plots are provided for the batch and semi-batch microgels studied as well as gel permeation chromatography traces of supernatant polymers for fast and slow-feed microgels. (PDF 84 kb)
Rights and permissions
About this article
Cite this article
Sheikholeslami, P., Ewaschuk, C.M., Ahmed, S.U. et al. Semi-batch control over functional group distributions in thermoresponsive microgels. Colloid Polym Sci 290, 1181–1192 (2012). https://doi.org/10.1007/s00396-012-2642-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-012-2642-x