Abstract
Introduction
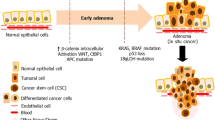
Despite the various therapeutic combinations and the emergence of new targeted therapies, there is still no curative treatment for all stages of colorectal cancer. Through the query for the best possible combination and solution, a new theory approaching colorectal cancer as a stem cell disease appeared, with a continuously growing body of evidence supporting this idea. The inability to directly recognize cancer stem cells has led researchers to an attempt of distinguishing those using indirect markers.
Discussion
This review focuses on colon cancer stem cell theory, the various findings supporting and contradicting this hypothesis, and the markers used up to now in characterizing stem cell populations in colorectal cancer. Despite the numerous unanswered questions on this new cancer hypothesis, it appears to have a justifiable role to play in colorectal cancer tumor biology, and furthermore, it may be the basis for the development of new therapeutic agents of the future. Therefore, every surgeon, oncologist, and physician who is implicated with this disease should be familiar with this novel colorectal cancer theory.

Similar content being viewed by others
References
Ferlay J, Autier P, Heanue M et al (2007) Estimates of the cancer incidence and mortality in Europe in 2006. Ann Oncol 18:581–592
Boman BM, Wicha MS (2008) Cancer stem cells: a step toward the cure. J Clin Oncol 26:2795–2799
Potten CS, Loeffler M (1990) Stem cells: attributes, cycles, spirals, pitfalls and uncertainties. Lessons from a crypt. Development 110:1001–1020
Brittan M, Wright NA (2002) Gastrointestinal stem cells. J Pathol 197:492–509
Reya T, Clevers H (2005) Wnt signalling in stem cells and cancer. Nature 434:843–850
Willis ND, Przyborski SA, Hutchison CJ et al (2008) Colonic and colorectal cancer stem cells: progress in the search for putative biomarkers. J Anat 213:59–65
Potten CS, Grant HK (1998) The relationship between ionizing radiation-induced apoptosis and stem cells in the small and large intestine. Brit J Cancer 78:993–1003
Meritt AJ, Potten CS, Watson AJM et al (1995) Differential expression of bcl-2 in intestinal epithelia. Correlation with attenuation of apoptosis in colonic crypts and the incidence of colonic neoplasia. J Cell Sci 198:2261–2271
O’Brien CA, Pollett A, Gallinger S et al (2007) A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature 445:106–110
Nguyen NP, Almeida FS, Chi A et al (2010) Molecular biology of breast cancer stem cells: potential clinical applications. Cancer Treat Rev 2010 (In press)
Oliveira LR, Jeffrey SS, Ribeiro-Silva A (2010) Stem cells in human breast cancer. Histol Histopathol 25:371–385
Singh SK, Hawkins C, Clarke ID et al (2004) Identification of human brain tumour initiating cells. Nature 432:396–401
Kim CF, Jackson EL, Woolfenden AE et al (2005) Identification of bronchioalveolar stem cells in normal lung and lung cancer. Cell 121:823–835
Ma S, Chan KW, Hu L et al (2007) Identification and characterization of tumorigenic liver cancer stem/progenitor cells. Gastroenterology 132:2542–2556
Schatton T, Murphy GF, Frank NY et al (2008) Identification of cells initiating human melanomas. Nature 451:345–349
Li C, Heidt DG, Dalerba P et al (2007) Identification of pancreatic cancer stem cells. Cancer Res 67:1030–1037
Pardal R, Clarke MF, Morrison SJ (2003) Applying the principles of stem-cell biology to cancer. Nat Rev Cancer 3:895–902
Gaziova I, Bhat KM (2007) Generating asymmetry: with and without renewal. Prog Mol Subcell Biol 45:143–178
Bach S, Renehan A, Potten C (2000) Stem cells: the intestinal stem cell as a paradigm. Carcinogenesis 21:469–476
Karam SM (1999) Lineage commitment and maturation of epithelial cells in the gut. Frontiers Biosci 4:286–298
Fevr T, Robine S, Louvard D, Huelsken J (2007) Wnt/beta-catenin is essential for intestinal homeostasis and maintenance of intestinal stem cells. Mol Cell Biol 27:7551–7559
Crosnier C, Stamataki D, Lewis J (2006) Organizing cell renewal in the intestine: stem cells, signals and combinatorial control. Nat Rev Genet 7:349–359
van Es JH, Clevers H (2005) Notch and Wnt inhibitors as potential new drugs for intestinal neoplastic disease. Trends Mol Med 11:496–502
Kosinski C, Li VSW, Chan ASY et al (2007) Gene expression patterns of human colon tops and basal crypts and BMP antagonists as intestinal stem cell niche factors. Proc Natl Acad Sci USA 104:15418–15423
Potten CS (1992) The significance of spontaneous and induced apoptosis in the gastrointestinal tract of mice. Cancer Metastasis Rev 179–195
Watson AJ, Merritt AJ, Jones LS et al (1996) Evidence of reciprocity of bcl-2 and p53 expression in human colorectal adenomas and carcinomas. Br J Cancer 73:889–895
Wilson JW, Nostro MC, Balzi M et al (2000) Bcl-w expression in colorectal adenocarcinoma. Br J Cancer 82:178–185
Kirkland SC (1988) Clonal origin of columnar, mucous, and endocrine cell lineages in human colorectal epithelium. Cancer 61:1359–1363
Yatabe Y, Tavare S, Shibata D (2001) Investigating stem cells in human colon by using methylation patterns. Proc Natl Acad Sci USA 98:10839–10844
Reya T, Morrison SJ, Clarke MF, Weissman IL (2001) Stem cells, cancer, and cancer stem cells. Nature 414:105–111
Cicalese A, Bonizzi G, Pasi CE et al (2009) The tumor suppressor p53 regulates polarity of self-renewing divisions in mammary stem cells. Cell 138:1083–1095
Kinzler KW, Vogelstein B (1996) Lessons from hereditary colorectal cancer. Cell 87:159–170
Fearon ER, Hamilton SR, Vogelstein B (1987) Clonal analysis of human colorectal tumors. Science 238:193–197
Fabian A, Barok M, Vereb G et al (2009) Die hard: are cancer stem cells the Bruce Willises of tumor biology? Cytom A 75:67–74
Bonnet D, Dick JE (1997) Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 3:730–737
Houghton J, Stoicov C, Nomura S et al (2004) Gastric cancer originating from bone marrow-derived cells. Science 306:1568–1571
Ricci-Vitiani L, Fabrizi E, Palio E et al (2009) Colon cancer stem cells. J Mol Med 87:1097–1104
Cotsarelis G, Sun TT, Lavker RM (1990) Label-retaining cells reside in the bulge area of pilosebaceous unit: implications for follicular stem cells, hair cycle, and skin carcinogenesis. Cell 61:1329–1337
Kiel MJ, Yilmaz OH, Iwashita T, Terhorst C, Morrison SJ (2005) SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 121:1109–1121
Shackleton M, Vaillant F, Simpson KJ, Stingl J et al (2006) Generation of a functional mammary gland from a single stem cell. Nature 439:84–88
Corbeil D, Roper K, Hellwig A et al (2000) The human AC133 hematopoietic stem cell antigen is also expressed in epithelial cells and targeted to plasma membrane protrusions. J Biol Chem 275:5512–5520
Corbeil D, Roper K, Fargeas CA et al (2001) Prominin: a story of cholesterol, plasma membrane protrusions and human pathology. Traffic 2:82–91
Haraguchi N, Utsunomiya T, Inoue H et al (2006) Characterization of a side population of cancer cells from human gastrointestinal system. Stem Cells 24:506–513
Ricci-Vitiani L, Lombardi DG, Pilozzi E et al (2007) Identification and expansion of human colon-cancer-initiating cells. Nature 445:111–115
Hermann PC, Huber SL, Herrler T et al (2007) Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell 1:313–323
Zhu L, Gibson P, Currle DS et al (2009) Promini1 marks intestinal stem cells that are susceptible to neoplastic transformation. Nature 457:603–607
Immervoll H, Hoem D, Sakariassen PO et al (2008) Expression of the “stem cell marker” CD133 in pancreas and pancreatic ductal adenocarcinomas. BMC Cancer 8:48
Saigusa S, Tanaka K, Toiyama Y et al (2009) Correlation of CD133, OCT4, and SOX2 in rectal cancer and their association with distant recurrence after chemoradiotherapy. Ann Surg Oncol 6(12):3488–3498
Horst D, Kriegl L, Engel J et al (2008) CD133 expression is an independent prognostic marker for low survival in colorectal cancer. Br J Cancer 99(8):1285–1289
Sparks AB, Morin PJ, Vogelstein B, Kinzler KW (1998) Mutational analysis of the APC/beta-catenin/Tcf pathway in colorectal cancer. Cancer Res 58:1130–1134
Bienz M, Clevers H (2000) Linking colorectal cancer to Wnt signaling. Cell 103:311–320
Horst D, Kriegl L, Engel J et al (2009) CD133 and nuclear β-catenin: the marker combination to detect high risk cases of low stage colorectal cancer. Eur J Cancer 45:2034–40
Corbeil D, Röper K, Hellwig A et al (2000) The human AC133 hematopoietic stem cell antigen is also expressed in epithelial cells and targeted to plasma membrane protrusions. J Biol Chem 275:5512–5520
Shmelkov SV, Butler JM, Hooper AT et al (2008) CD133 expression is not restricted to stem cells, and both CD133+ and CD133- metastatic colon cancer cells initiate tumors. J Clin Invest 118:2111–2120
Lin EH, Hassan M, Li Y et al (2007) Elevated circulating endothelial progenitor marker CD133 messenger RNA levels predict colon cancer recurrence. Cancer 110:534–542
Ieta K, Tanaka F, Haraguchi N et al (2008) Biological and genetic characteristics of tumor-initiating cells in colon cancer. Ann Surg Oncol 15:638–648
Choi D, Lee HW, Hur KY et al (2009) Cancer stem cell markers CD133 and CD24 correlate with invasiveness and differentiation in colorectal adenocarcinoma. World J Gastroenterol 15:2258–2264
Lim SC, Oh SH (2005) The role of CD24 in various human epithelial neoplasias. Pathol Res Pract 201:479–486
Weichert W, Denkert C, Burkhardt M et al (2005) Cytoplasmic CD24 expression in colorectal cancer independently correlates with shortened patient survival. Clin Cancer Res 11:6574–6581
Horst D, Sheel SK, Liebmann S et al (2009) The cancer stem cell marker CD133 has high prognostic impact but unknown functional relevance for the metastasis of human colon cancer. J Pathol 219:427–434
Cheng L, Sung MT, Cossu-Rocca P et al (2007) OCT4: biological functions and clinical applications as a marker of germ cell neoplasia. J Pathol 211:1–9
Sotomayor P, Godoy A, Smith GJ (2008) Oct4A is expressed by a subpopulation of prostate neuroendocrine cells. Prostate 69:401–410
Hochedlinger K, Yamada Y, Beard C, Jaenisch R (2005) Ectopic expression of Oct-4 blocks progenitor-cell differentiation and causes dysplasia in epithelial tissues. Cell 121:465–477
Yamanaka S (2008) Induction of pluripotent stem cells from mouse fibroblasts by four transcription factors. Cell Prolif 41:51–56
Ben-Porath I, Thomson MW, Carey VJ et al (2008) An embryonic stem cell-like gene expression signature in poorly differentiated aggressive human tumors. Nat Genet 40:499–507
Schoenhals M, Kassambara A, De Vos J et al (2009) Embryonic stem cell markers expression in cancers. Biochem Biophys Res Commun 383:157–162
Rodriguez-Pinilla SM, Sarrio D, Moreno-Bueno G et al (2007) Sox2: a possible driver of the basal-like phenotype in sporadic breast cancer. Mod Pathol 20:474–481
Gu G, Yuan J, Wills M, Kasper S (2007) Prostate cancer cells with stem cell characteristics reconstitute the original human tumor in vivo. Cancer Res 67:4807–4815
Tsukamoto T, Mizoshita T, Mihara M et al (2005) Sox2 expression in human stomach adenocarcinomas with gastric and gastric-and intestinal-mixed phenotypes. Histopathology 46:649–658
Evans PM, Liu C (2008) Roles of Kruppel-like factor 4 in normal homeostasis, cancer and stem cells. Acta Biochem Biophys Sin (Shanghai) 40:554–564
McConnell BB, Ghaleb AM, Nandan MO, Yang VW (2007) The diverse functions of Kruppel-like factors 4 and 5 in epithelial biology and pathobiology. Bioessays 29:549–557
Wei D, Kanai M, Huang S, Xie K (2006) Emerging role of KLF4 in human gastrointestinal cancer. Carcinogenesis 27:23–31
Rowland BD, Peeper DS (2006) KLF4, p21 and context-dependent opposing forces in cancer. Nat Rev Cancer 6:11–23
Adhikary S, Eilers M (2005) Transcriptional regulation and transformation by Myc proteins. Nat Rev Mol Cell Biol 6:635–645
Kim JB, Zaehres H, Wu G et al (2008) Pluripotent stem cells induced from adult neural stem cells by reprogramming with two factors. Nature 454:646–650
Okita K, Ichisaka T, Yamanaka S (2007) Generation of germline-competent induced pluripotent stem cells. Nature 448:313–317
Wernig M, Meissner A, Foreman R et al (2007) In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature 448:318–324
Eilers M, Eisenman RN (2008) Myc’s broad reach. Genes Dev 22:2755–2766
Schulein C, Eilers M (2009) An unsteady scaffold for Myc. EMBO 28:453–454
Welcker M, Orian A, Jin J et al (2004) The Fbw7 tumor suppressor regulates glycogen synthase kinase 3 phosphorylation-dependent c-Myc protein degradation. Proc Natl Acad Sci USA 101:9085–9090
Arnold HK, Zhang X, Daniel CJ et al (2009) The Axin1 scaffold protein promotes formation of a degradation complex for c-Myc. EMBO J 28:500–512
Yada M, Hatakeyama S, Kamura T et al (2004) Phosphorylation-dependent degradation of c-Myc is mediated by the F-box protein Fbw7. EMBO J 23:2116–2125
Rajagopalan H, Jallepalli PV, Rago C, Velculescu VE, Kinzler KW, Vogelstein B, Lengauer C (2004) Inactivation of hCDC4 can cause chromosomal instability. Nature 428:77–81
Popov N, Wanzel M, Madiredjo M et al (2007) The ubiquitin-specific protease USP28 is required for MYC stability. Nat Cell Biol 9:765–774
Haegebarth A, Clevers H (2009) Wnt signaling, Lgr5, and stem cells in the intestine and the skin. Am J Pathol 174:715–21
Barker N, van Es JH, Kuipers J et al (2007) Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449:1003–1007
Sato T, Vries RG, Snippert HJ et al (2009) Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459:262–265
Barker N, Ridgway RA, van Es JH et al (2009) Crypt stem cells as the cells-of-origin of intestinal cancer. Nature 457:608–612
Kaneko Y, Sakakibara S, Imai T et al (2000) Musashi1: an evolutionally conserved marker for CNS progenitor cells including neural stem cells. Dev Neurosci 22:139–153
Booth C, Potten CS (2000) Gut instincts: thoughts on intestinal epithelial stem cells. J Clin Invest 105:1493–1499
Nishimura S, Wakabayashi N, Toyoda K et al (2003) Expression of Musashi-1 in human normal colon crypt cells: a possible stem cell marker of human colon epithelium. Dig Dis Sci 48:1523–1529
Fujimoto K, Beauchamp RD, Whitehead RH (2002) Identification and isolation of candidate human colonic clonogenic cells based on cell surface integrin expression. Gastroenterology 123:1941–1948
Howe A, Aplin AE, Alahari SK et al (1998) Integrin signaling and cell growth control. Curr Opin Cell Biol 10:220–231
Huang EH, Hynes MJ, Zhang T et al (2009) Aldehyde dehydrogenase 1 is a marker for normal and malignant human colonic stem cells (SC) and tracks SC overpopulation during colon tumorigenesis. Cancer Res 69:3382–3389
Bao S, Wu Q, McLendon RE et al (2006) Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 444:756–760
Todaro M, Perez Alea M, Scopelliti A et al (2008) IL-4-mediated drug resistance in colon cancer stem cells. Cell Cycle 7:309–313
Donnenberg VS, Landreneau RJ, Donnenberg AD (2007) Tumorigenic stem and progenitor cells: implications for the therapeutic index of anti-cancer agents. J Control Release 122:385–391
May R, Riehl TE, Hunt C, Sureban SM, Anant S, Houchen CW (2008) Identification of a novel putative gastrointestinal stem cell and adenoma stem cell marker, doublecortin and CaM kinase-like-1, following radiation injury and in adenomatous polyposis coli/multiple intestinal neoplasia mice. Stem Cells 26:630–337
Dalebra P, Dylla SJ, Park IK et al (2007) Phenotypic characterization of human colorectal cancer stem cells. Proc Natl Acad Sci USA 104:10158–10163
Todaro M, Francipane MG, Medema JP, Stassi G (2001) Colon cancer stem cells: promise of targeted therapy. Gastroenterology 138:2151–2162
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Papailiou, J., Bramis, K.J., Gazouli, M. et al. Stem cells in colon cancer. A new era in cancer theory begins. Int J Colorectal Dis 26, 1–11 (2011). https://doi.org/10.1007/s00384-010-1022-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-010-1022-6