Abstract
Background
Gastroschisis is an index neonatal malformation with an increasing incidence from earlier decades. The aim of this study was to investigate regional variations in patient demographics, outcomes, and costs in gastroschisis.
Materials and methods
The 2016 kid’s inpatient database (KID) obtained from the Healthcare Cost and Utilization Project (HCUP) was used to obtain gastroschisis birth, demographic, and outcome data across four U.S. geographical regions: Northeast, Midwest, South, and West. Categorical variables were analyzed with Pearson’s Chi-squared test. Odds ratios (OR) with 95% confidence intervals (CI) were reported for variables with significance (p < 0.05). Appropriate logistic regressions were used for comparing categorical, cardinal, and continuous data.
Results
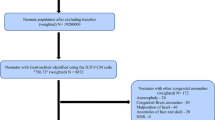
2043 gastroschisis cases were obtained from the 2016 KID database with a weighted incidence of 5.1 per 10,000 live births. Incidence of gastroschisis across regions was similar when controlling for a number of hospitals reporting to KID. Prematurity was common, with 59% (1204) of neonates with gastroschisis born at less than 37 weeks. 52% (N = 1067) of gastroschisis infants were male. 46% (955) were White, 9% (178) African American, 22% (450) Hispanic, and 8% (155) other. The overall mortality of gastroschisis in this database was 3.6% (75). The average length of stay was 39 days and average cost of treatment was $357,436. Though without significant differences in mortality, there were significant differences in demographics, costs, and lengths of stay between regions.
Conclusions
There has been a significant increase in gastroschisis incidence over the last three decades. This significant increase appears to be related to an increase in socioeconomic inequality and an alteration in the population demographics in the United States. Further study of regional variations will facilitate the development of best practice guidelines that benefit all.
Similar content being viewed by others
Data availability
This article is based on the KID database. The data in this database is HIPAA compliant from a de-identified subset of patients and retrospective in nature. Data use permission was obtained from the KID database prior to presentation and publication.
References
Arnold MA, Chang DC, Nabaweesi R et al (2007) Risk stratification of 4344 patients with gastroschisis into simple and complex categories. J Pediatr Surg 42:1520–1525
Bhatt P, Lekshminarayanan A, Donda K et al (2018) Trends in incidence and outcomes of gastroschisis in the United States: analysis of the national inpatient sample 2010–2014. Pediatr Surg Int 34:919–929
Hook-Dufresne DM, Yu X, Bandla V et al (2015) The economic burden of gastroschisis: costs of a birth defect. J Surg Res 195:16–20
Wilmore DW, Groff DB, Bishop HC et al (1969) Total parenteral nutrition in infants with catastrophic gastrointestinal anomalies. J Pediatr Surg 4:181–189
Bermejo E, Mendioroz J, Cuevas L et al (2006) The incidence of gastroschisis: is also increasing in Spain, particularly among babies of young mothers. BMJ 332:424
Di Tanna GL, Rosano A, Mastroiacovo P (2002) Prevalence of gastroschisis at birth: retrospective study. BMJ 325:1389–1390
Kilby MD (2006) The incidence of gastroschisis. BMJ 332:250–251
Mastroiacovo P, Lisi A, Castilla EE (2006) The incidence of gastroschisis: research urgently needs resources. BMJ 332:423–424
Werler MM, Mitchell AA, Moore CA et al (2009) Is there epidemiologic evidence to support vascular disruption as a pathogenesis of gastroschisis? Am J Med Genet A 149A:1399–1406
Interrante JD, Ailes EC, Lind JN et al (2017) Risk comparison for prenatal use of analgesics and selected birth defects, national birth defects prevention study 1997–2011. Ann Epidemiol 27:645.e642-653.e642
Short TD, Stallings EB, Isenburg J et al (2019) Gastroschisis trends and ecologic link to opioid prescription rates—United States, 2006–2015. MMWR Morb Mortal Wkly Rep 68:31–36
Getz KD, Anderka MT, Werler MM et al (2012) Short interpregnancy interval and gastroschisis risk in the national birth defects prevention study. Birth Defects Res A Clin Mol Teratol 94:714–720
Feldkamp ML, Enioutina EY, Botto LD et al (2015) Chlamydia trachomatis IgG3 seropositivity is associated with gastroschisis. J Perinatol 35:930–934
Reefhuis J, Devine O, Friedman JM et al (2015) Specific SSRIs and birth defects: Bayesian analysis to interpret new data in the context of previous reports. BMJ 351:h3190
Richardson S, Browne ML, Rasmussen SA et al (2011) Associations between periconceptional alcohol consumption and craniosynostosis, omphalocele, and gastroschisis. Birth Defects Res A Clin Mol Teratol 91:623–630
Padula AM, Yang W, Schultz K et al (2016) Gene variants as risk factors for gastroschisis. Am J Med Genet A 170:2788–2802
(HCUP) (2016) HCaUP, HCUP Kid’s Inpatient Database (KID). Agency for Healthcare Research and Quality, Rockville, MD
Organization, WH (2016) International statistical classification of diseases and related health problems, 10th revision. https://icd.who.int/browse10/2016/en
Bureau, USC (2019) Selected economic characteristics. https://data.census.gov/cedsci/table?d=ACS%205-Year%20Estimates%20Data%20Profiles&table=DP03&tid=ACSDP5Y2016.DP03&y=2016&lastDisplayedRow=127. Accessed 12 Oct 2016
Martin JA, Hamilton BE, Osterman MJK, Driscoll AK, Drake P (2018) National vital statistics reports: births: final data for 2016, p 67
Palmer SR, Evans A, Broughton H et al (2013) The role of maternal stress in early pregnancy in the aetiology of gastroschisis: an incident case control study. PLoS ONE 8:e80103
Carmichael SL, Ma C, Tinker S et al (2017) Maternal stressors and social support and risks of delivering babies with gastroschisis or hypospadias. Am J Epidemiol 185:1240–1246
Paranjothy S, Broughton H, Evans A et al (2012) The role of maternal nutrition in the aetiology of gastroschisis: an incident case–control study. Int J Epidemiol 41:1141–1152
Deguen S, Kihal W, Jeanjean M et al (2016) Neighborhood deprivation and risk of congenital heart defects, neural tube defects and orofacial clefts: a systematic review and meta-analysis. PLoS ONE 11:e0159039
Elliott P, Briggs D, Morris S et al (2001) Risk of adverse birth outcomes in populations living near landfill sites. BMJ 323:363–368
Hinton CF, Siffel C, Correa A et al (2017) Survival disparities associated with congenital diaphragmatic hernia. Birth Defects Res 109:816–823
Lupo PJ, Danysh HE, Symanski E et al (2015) Neighborhood-based socioeconomic position and risk of oral clefts among offspring. Am J Public Health 105:2518–2525
Moore TC, Collins DL, Catanzarite V et al (1999) Pre-term and particularly pre-laborcesarean section to avoid complications of gastroschisis. Pediatr Surg Int 15:97–104
Sakala EP, Erhard LN, White JJ (1993) Elective cesarean section improves outcomes of neonates with gastroschisis. Am J Obstet Gynecol 169:1050–1053
Gupta R, Cabacungan ET (2018) Outcome of neonates with gastroschisis at different gestational ages using a national database. J Pediatr Surg 53:661–665
Carnaghan H, Baud D, Lapidus-Krol E et al (2016) Effect of gestational age at birth on neonatal outcomes in gastroschisis. J Pediatr Surg 51:734–738
Palatnik A, Loichinger M, Wagner A et al (2020) The association between gestational age at delivery, closure type and perinatal outcomes in neonates with isolated gastroschisis. J Matern Fetal Neonatal Med 33(8):1393–1399
Corey KM, Hornik CP, Laughon MM et al (2014) Frequency of anomalies and hospital outcomes in infants with gastroschisis and omphalocele. Early Hum Dev 90:421–424
de Buys Roessingh AS, Damphousse A, Ballabeni P et al (2015) Predictive factors at birth of the severity of gastroschisis. World J Gastrointest Pathophysiol 6:228–234
Lao OB, Larison C, Garrison MM et al (2010) Outcomes in neonates with gastroschisis in US children’s hospitals. Am J Perinatol 27:97–101
Marinovic VM, Lukac M, Mikovic Z et al (2016) Outcome differences between simple and complex gastroschisis. Ann Ital Chir 87:525–530
Payne NR, Pfleghaar K, Assel B et al (2009) Predicting the outcome of newborns with gastroschisis. J Pediatr Surg 44:918–923
Austin MT, Nguyen H, Eberth JM et al (2015) Health disparities are important determinants of outcome for children with solid tumor malignancies. J Pediatr Surg 50:161–166
Chu DI, Canning DA, Tasian GE (2016) Race and 30-day morbidity in pediatric urologic surgery. Pediatrics 138:e20154574
Song YK, Nunez Lopez O, Mehta HB et al (2017) Race and outcomes in gastroschisis repair: a nationwide analysis. J Pediatr Surg 52:1755–1759
Stone ML, Lapar DJ, Kane BJ et al (2013) The effect of race and gender on pediatric surgical outcomes within the United States. J Pediatr Surg 48:1650–1656
Author information
Authors and Affiliations
Contributions
ANS contributed to the data collection, data analysis, and manuscript drafting. Professor SB contributed to the conceptual development, finalization of data, and manuscript editing.
Corresponding author
Ethics declarations
Conflict of interest
Both authors do not have any conflict of interest. The authors report no proprietary or commercial interest in any product mentioned or concept discussed in this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix
The following lists the categorical variables that were grouped differently than the HCUP groupings. Groups were only combined. Grouping was changed only when criteria for Pearson’s Chi-squared test was not met due to a cell size < 5.
Race
The HCUP categorization of this variable an indicator of race:
-
1.
White,
-
2.
Black,
-
3.
Hispanic,
-
4.
Asian or Pacific Islander,
-
5.
Native American,
-
6.
Other
This variable was regrouped as:
-
1.
White
-
2.
Black
-
3.
Hispanic
-
4.
Other
Primary expected payer
The HCUP categorization of this variable an indicator of expected primary payer:
-
1.
Medicare,
-
2.
Medicaid,
-
3.
Private including HMO,
-
4.
Self-pay,
-
5.
No charge,
-
6.
Other
This variable was regrouped as:
(1) Medicare, Medicaid, or no charge or (2) Private, HMO, Self-pay, other.
Highlights
-
Incidence of gastroschisis is rising (~ 1 in 2000), causes are likely multifactorial
-
Mortality of gastroschisis remains low, around 3% across the U.S.
-
Premature infants were 44% of the sample, with lowest incidence in the Northeast
-
Incidence varied significantly among gender, race, and economic status by region
-
Length of stay and costs were highest in the Northeast U.S.
Rights and permissions
About this article
Cite this article
Burjonrappa, S., Snyder, A.N. Regional variation in gastroschisis: a nationwide database review of demographics and outcomes. Pediatr Surg Int 37, 911–917 (2021). https://doi.org/10.1007/s00383-021-04894-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-021-04894-2