Abstract
Purpose
Epilepsy following non-accidental trauma (NAT) occurs in 18% of pediatric patients. About 33% of patients with epilepsy develop drug-resistant epilepsy. For these patients, vagus nerve stimulation (VNS) is a palliative treatment option. We aimed to investigate the effectiveness of VNS among pediatric NAT-related epilepsy patients compared to those with non-NAT-related epilepsy.
Methods
We performed an 11-year retrospective analysis of VNS implantations for drug-resistant epilepsy at UPMC Children’s Hospital of Pittsburgh. Patients were split into two groups: NAT vs. non-NAT. The primary outcome was the attainment of ≥ 50% seizure frequency reduction at 1-year post-VNS implantation. Fisher’s exact tests and Wilcoxon rank-sum tests were used to compare groups. Significance was assessed at the alpha = 0.05 level.
Results
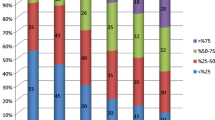
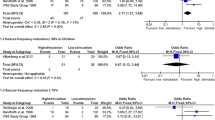
This analysis included data from 370 pediatric VNS patients, of whom 9 had NAT-related epilepsy. NAT patients had a significantly younger age of epilepsy onset than non-NAT patients (0.3 years vs. 3.3 years). Otherwise, there were no statistically significant baseline differences between groups, including patient sex and quantity of antiseizure medications pre-VNS. Overall, 71% of NAT patients experienced ≥ 50% seizure frequency reduction compared to 48% of non-NAT patients (p = 0.269).
Conclusion
VNS may allow a higher proportion of pediatric patients with NAT-related epilepsy to achieve ≥ 50% seizure frequency reduction compared to other epilepsy etiologies. While the results of this study were not statistically significant, the effect size was large. Our results underscore the need for larger, multi-center studies to validate the effectiveness of VNS for this patient population.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Iqbal O'Meara AM, Sequeira J, Ferguson NM (2020) Advances and future directions of diagnosis and management of pediatric abusive head trauma: a review of the literature. Front Neurol 11:118
Estroff JM, Foglia RP, Fuchs JR (2015) A comparison of accidental and nonaccidental trauma: it is worse than you think. J Emerg Med 48(3):274–279
Oluigbo CO et al (2012) Comparison of outcomes following decompressive craniectomy in children with accidental and nonaccidental blunt cranial trauma. J Neurosurg Pediatr 9(2):125–132
Irimia A, Van Horn JD (2015) Epileptogenic focus localization in treatment-resistant post-traumatic epilepsy. J Clin Neurosci 22(4):627–631
Keenan HT et al (2004) A population-based comparison of clinical and outcome characteristics of young children with serious inflicted and noninflicted traumatic brain injury. Pediatrics 114(3):633–639
Larkin M et al (2016) Post-traumatic, drug-resistant epilepsy and review of seizure control outcomes from blinded, randomized controlled trials of brain stimulation treatments for drug-resistant epilepsy. Cureus 8(8):e744
Kwan P et al (2010) Definition of drug resistant epilepsy: consensus proposal by the ad hoc Task Force of the ILAE Commission on Therapeutic Strategies. Epilepsia 51(6):1069–1077
Muthiah N et al (2022) Comparison of traditional and closed loop vagus nerve stimulation for treatment of pediatric drug-resistant epilepsy: a propensity-matched retrospective cohort study. Seizure 94:74–81
Hachem LD, Wong SM, Ibrahim GM (2018) The vagus afferent network: emerging role in translational connectomics. Neurosurg Focus 45(3):E2
Koo B (2001) EEG changes with vagus nerve stimulation. J Clin Neurophysiol 18(5):434–441
Smith DC et al (2005) Electrical stimulation of the vagus nerve enhances cognitive and motor recovery following moderate fluid percussion injury in the rat. J Neurotrauma 22(12):1485–1502
Zack MM, Kobau R (2017) National and state estimates of the numbers of adults and children with active epilepsy - United States, 2015. MMWR Morb Mortal Wkly Rep 66(31):821–825
Park JT, Chugani HT (2015) Post-traumatic epilepsy in children-experience from a tertiary referral center. Pediatr Neurol 52(2):174–181
Chevignard MP, Lind K (2014) Long-term outcome of abusive head trauma. Pediatr Radiol 44(Suppl 4):S548–S558
Hwang SY et al (2019) Long-term outcomes in children with moderate to severe traumatic brain injury: a single-centre retrospective study. Brain Inj 33(11):1420–1424
Paul AR, Adamo MA (2014) Non-accidental trauma in pediatric patients: a review of epidemiology, pathophysiology, diagnosis and treatment. Transl Pediatr 3(3):195–207
Araki T, Yokota H, Morita A (2017) Pediatric traumatic brain injury: characteristic features, diagnosis, and management. Neurol Med Chir (Tokyo) 57(2):82–93
Joyce T, Gossman W, Huecker MR (2022) Pediatric abusive head trauma, in StatPearls. Treasure Island (FL)
Mai T et al (2018) Infantile colic: new insights into an old problem. Gastroenterol Clin North Am 47(4):829–844
Barr RG (2014) Crying as a trigger for abusive head trauma: a key to prevention. Pediatr Radiol 44(Suppl 4):S559–S564
Temkin NR (2001) Antiepileptogenesis and seizure prevention trials with antiepileptic drugs: meta-analysis of controlled trials. Epilepsia 42(4):515–524
Garga N, Lowenstein DH (2006) Posttraumatic epilepsy: a major problem in desperate need of major advances. Epilepsy Curr 6(1):1–5
Muthiah N et al (2022) Development of a clinical model to predict vagus nerve stimulation response in pediatric patients with drug-resistant epilepsy. medRxiv 1(13):22269237
Englot DJ et al (2016) Rates and predictors of seizure freedom with vagus nerve stimulation for intractable epilepsy. Neurosurgery 79(3):345–353
Englot DJ, Chang EF, Auguste KI (2011) Vagus nerve stimulation for epilepsy: a meta-analysis of efficacy and predictors of response. J Neurosurg 115(6):1248–1255
Greenland S (2019) Valid P-values behave exactly as they should: some misleading criticisms of P-values and their resolution with S-values. Am Stat 73(sup1):106–114
Englot DJ et al (2012) Efficacy of vagus nerve stimulation in posttraumatic versus nontraumatic epilepsy. J Neurosurg 117(5):970–977
Author information
Authors and Affiliations
Contributions
Conceptualization: Nallammai Muthiah, Taylor J. Abel; Methodology: Nallammai Muthiah, Brigit Joseph, Lena Vodovotz, Nikhil Sharma; Formal analysis and investigation: Nallammai Muthiah; Writing—original draft preparation: Nallammai Muthiah, Brigit Joseph; Writing—review and editing: Nallammai Muthiah, Brigit Joseph, Gregory Varga, Taylor J. Abel; Funding acquisition: n/a; Resources: Nallammai Muthiah, Taylor J. Abel; Supervision: Taylor J. Abel.
Corresponding author
Ethics declarations
Ethics approval
This retrospective chart review study involving human participants was in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The Human Investigation Committee (IRB) of the University of Pittsburgh approved this study (STUDY20070281).
Consent for participate
Informed consent was obtained from legal guardians or patients (when applicable). No identifying images or patient data are presented in this manuscript.
Conflict of interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Muthiah, N., Joseph, B., Varga, G. et al. Investigation of the effectiveness of vagus nerve stimulation for pediatric drug-resistant epilepsies secondary to nonaccidental trauma. Childs Nerv Syst 39, 1201–1206 (2023). https://doi.org/10.1007/s00381-022-05817-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-022-05817-9