Abstract
Purpose
The craniometrics of head circumference (HC) and ventricular size are part of the clinical assessment of infants with hydrocephalus and are often utilized in conjunction with other clinical and radiological parameters to determine the success of treatment. We aimed to assess the effect of endoscopic third ventriculostomy (ETV) and shunting on craniometric measurements during the follow-up of a cohort of infants with symptomatic triventricular hydrocephalus secondary to aqueductal stenosis.
Methods
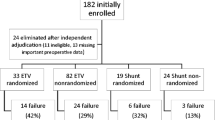
We performed a post hoc analysis of data from the International Infant Hydrocephalus Study (IIHS)—a prospective, multicenter study of infants (< 24 months old) with hydrocephalus from aqueductal stenosis who were treated with either an ETV or shunt. During various stages of a 5-year follow-up period, the following craniometrics were measured: HC, HC centile, HC z-score, and frontal-occipital horn ratio (FOR). Data were compared in an analysis of covariance, adjusting for baseline variables including age at surgery and sex.
Results
Of 158 enrolled patients, 115 underwent an ETV, while 43 received a shunt. Both procedures led to improvements in the mean HC centile position and z-score, a trend which continued until the 5-year assessment point. A similar trend was noted for FOR which was measured at 12 months and 3 years following initial treatment. Although the values were consistently higher for ETV compared with shunt, the differences in HC value, centile, and z-score were not significant. ETV was associated with a significantly higher FOR compared with shunting at 12 months (0.52 vs 0.44; p = 0.002) and 3 years (0.46 vs 0.38; p = 0.03) of follow-up.
Conclusion
ETV and shunting led to improvements in HC centile, z-score, and FOR measurements during long-term follow-up of infants with hydrocephalus secondary to aqueductal stenosis. Head size did not significantly differ between the treatment groups during follow-up, however ventricle size was greater in those undergoing ETV when measured at 1 and 3 years following treatment.


Similar content being viewed by others
References
Kulkarni AV, Drake JM, Armstrong DC, Dirks PB (2000) Imaging correlates of successful endoscopic third ventriculostomy. J Neurosurg 92:915–919. https://doi.org/10.3171/jns.2000.92.6.0915
Nowosławska E, Polis L, Kaniewska D, Mikołajczyk W, Krawczyk J, Szymański W, Zakrzewski K, Podciechowska J (2004) Influence of neuroendoscopic third ventriculostomy on the size of ventricles in chronic hydrocephalus. J Child Neurol 19:579–587. https://doi.org/10.1177/088307380401900803
St George E, Natarajan K, Sgouros S (2004) Changes in ventricular volume in hydrocephalic children following successful endoscopic third ventriculostomy. Childs Nerv Syst 20:834–838. https://doi.org/10.1007/s00381-004-0939-x
Hopf NJ, Grunert P, Fries G et al (1999) Endoscopic third ventriculostomy: outcome analysis of 100 consecutive procedures. Neurosurgery 44:795–804. https://doi.org/10.1097/00006123-199904000-00062
Sgouros S, Kulkharni AV, Constantini S (2006) The International Infant Hydrocephalus Study: concept and rational. Child’s Nerv Syst ChNS Off J Int Soc Pediatr Neurosurg 22:338–345. https://doi.org/10.1007/s00381-005-1253-y
Kulkarni AV, Sgouros S, Constantini S, Investigators I (2016) International Infant Hydrocephalus Study: initial results of a prospective, multicenter comparison of endoscopic third ventriculostomy (ETV) and shunt for infant hydrocephalus. Child’s Nerv Syst ChNS Off J Int Soc Pediatr Neurosurg 32:1039–1048. https://doi.org/10.1007/s00381-016-3095-1
Kulkarni AV, Sgouros S, Leitner Y, Constantini S, International Infant Hydrocephalus Study Investigators (2018) International Infant Hydrocephalus Study (IIHS): 5-year health outcome results of a prospective, multicenter comparison of endoscopic third ventriculostomy (ETV) and shunt for infant hydrocephalus. Child’s Nerv Syst ChNS Off J Int Soc Pediatr Neurosurg 34:2391–2397. https://doi.org/10.1007/s00381-018-3896-5
Olschewski M, Schumacher M, Davis KB (1992) Analysis of randomized and nonrandomized patients in clinical trials using the comprehensive cohort follow-up study design. Control Clin Trials 13:226–239
O’Hayon BB, Drake JM, Ossip MG et al (1998) Frontal and occipital horn ratio: a linear estimate of ventricular size for multiple imaging modalities in pediatric hydrocephalus. Pediatr Neurosurg 29:245–249. https://doi.org/10.1159/000028730
Kulkarni AV, Drake JM, Armstrong DC, Dirks PB (1999) Measurement of ventricular size: reliability of the frontal and occipital horn ratio compared to subjective assessment. Pediatr Neurosurg 31:65–70. https://doi.org/10.1159/000028836
Kulkarni AV, Sgouros S, Constantini S, Investigators IIHS (2017) Outcome of treatment after failed endoscopic third ventriculostomy (ETV) in infants with aqueductal stenosis: results from the International Infant Hydrocephalus Study (IIHS). Child’s Nerv Syst ChNS Off J Int Soc Pediatr Neurosurg 33:747–752. https://doi.org/10.1007/s00381-017-3382-5
Azab WA, Mijalcic RM, Nakhi S Ben, Mohammad MH (2016) Ventricular volume and neurocognitive outcome after endoscopic third ventriculostomy: is shunting a better option? A review. Childs Nerv Syst 32:775–780. https://doi.org/10.1007/s00381-016-3032-3
Kulkarni AV, Donnelly R, Mabbott DJ, Widjaja E (2015) Relationship between ventricular size, white matter injury, and neurocognition in children with stable, treated hydrocephalus. J Neurosurg Pediatr 16:267–274. https://doi.org/10.3171/2015.1.PEDS14597
Dewan MC, Lim J, Gannon SR, Heaner D, Davis MC, Vaughn B, Chern JJ, Rocque BG, Klimo P, Wellons JC, Naftel RP (2018) Comparison of hydrocephalus metrics between infants successfully treated with endoscopic third ventriculostomy with choroid plexus cauterization and those treated with a ventriculoperitoneal shunt: a multicenter matched-cohort analysis. J Neurosurg Pediatr 21:339–345. https://doi.org/10.3171/2017.10.PEDS17421
Dewan MC, Lim J, Morgan CD et al (2016) Endoscopic third ventriculostomy with choroid plexus cauterization outcome: distinguishing success from failure. J Neurosurg Pediatr 18:655–662. https://doi.org/10.3171/2016.6.PEDS1675
Nilsson D, Svensson J, Korkmaz BA et al (2013) Decreased head circumference in shunt-treated compared with healthy children. J Neurosurg Pediatr 12:483–490. https://doi.org/10.3171/2013.8.PEDS1370
Warf B, Ondoma S, Kulkarni A, Donnelly R, Ampeire M, Akona J, Kabachelor CR, Mulondo R, Nsubuga BK (2009) Neurocognitive outcome and ventricular volume in children with myelomeningocele treated for hydrocephalus in Uganda. J Neurosurg Pediatr 4:564–570. https://doi.org/10.3171/2009.7.PEDS09136
Kulkarni AV, Schiff SJ, Mbabazi-Kabachelor E, Mugamba J, Ssenyonga P, Donnelly R, Levenbach J, Monga V, Peterson M, MacDonald M, Cherukuri V, Warf BC (2017) Endoscopic treatment versus shunting for infant hydrocephalus in Uganda. N Engl J Med 377:2456–2464. https://doi.org/10.1056/NEJMoa1707568
Mandell JG, Kulkarni AV, Warf BC, Schiff SJ (2015) Volumetric brain analysis in neurosurgery: part 2. Brain and CSF volumes discriminate neurocognitive outcomes in hydrocephalus. J Neurosurg Pediatr 15:125–132. https://doi.org/10.3171/2014.9.PEDS12427
Acknowledgments
The authors would like to extend a special thanks to Adina Sherer, who ran the organizational logistics of this study and without whom, the IIHS would not have been possible.
Steering Committee: Shlomi Constantini (Principal Investigator), Spyros Sgouros, and Abhaya V. Kulkarni
Consultant Neurologist: Yael Leitner
Data Safety Monitoring Committee: John RW Kestle (Chair), Douglas D Cochrane, Maurice Choux, and Fleming Gjerris
Coordinating Administrator: Adina Sherer
Participating Investigator Authors: Nejat Akalan, Burçak Bilginer (Ankara, Turkey); Ramon Navarro (Barcelona, Spain); Ljiljana Vujotic (Belgrade, Serbia); Hannes Haberl, Ulrich-Wilhelm Thomale (Berlin, Germany); Spyros Sgouros (Birmingham, UK); Graciela Zúccaro, Roberto Jaimovitch (Buenos Aires, Argentina); David Frim, Lori Loftis (Chicago, USA); Dale M. Swift, Brian Robertson, Lynn Gargan (Dallas, USA); László Bognár, László Novák, Georgina Cseke (Debrecen, Hungary); Armando Cama, Giuseppe Marcello Ravegnani (Genova, Italy); Matthias Preuß (Giessen/Leipzig, Germany); Henry W. Schroeder, Michael Fritsch, Joerg Baldauf (Greifswald, Germany); Marek Mandera, Jerzy Luszawski, Patrycja Skorupka (Katowice, Poland); Conor Mallucci, Dawn Williams (Liverpool, UK); Krzysztof Zakrzewski, Emilia Nowoslawska (Lodz, Poland); Chhitij Srivastava, Ashok K. Mahapatra, Raj Kumar, Rabi Narayan Sahu (Lucknow, India); Armen G. Melikian, Anton Korshunov, Anna Galstyan (Moscow, Russia); Ashish Suri, Deepak Gupta (New Delhi, India); J. André Grotenhuis, Erik J. van Lindert (Nijmegen, The Netherlands); José Aloysio da Costa Val (Nova Lima, Brazil); Concezio Di Rocco, Gianpiero Tamburrini (Rome, Italy); Samuel Tau Zymberg, Sergio Cavalheiro (São Paulo, Brazil); Ma Jie, Jiang Feng (Shanghai, China); Shlomi Constantini, Orna Friedman (Tel Aviv, Israel); Abhaya V. Kulkarni, Naheeda Rajmohamed (Toronto, Canada); Marcin Roszkowski, and Slawomir Barszcz (Warsaw, Poland)
The following centres (and investigators) participated in the IIHS, but did not enroll any patients: Baltimore, Maryland, USA (George Jallo); Gainesville, Florida, USA (David W. Pincus, Bridget Richter); Kiel, Germany (HM Mehdorn, Susan Schultka); London, Ontario, Canada (Sandrine de Ribaupierre); London, UK (Dominic Thompson, Silvia Gatscher); Mainz, Germany (Wolfgang Wagner, Dorothee Koch); Reggio Calabria, Italy (Saverio Cipri, Claudio Zaccone); and Winnipeg, Manitoba, Canada (Patrick McDonald).
Author information
Authors and Affiliations
Consortia
Contributions
AVK, SS, CS, and ICC contributed to the study conception and design. Data collection was performed by AVK, SS, CS, and collaborators from the IIHS. Material preparation and data analysis were performed by ICC and AVK. The first draft of the manuscript was written by ICC, and all authors commented on previous versions of the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
The IIHS was publicly registered (clinicaltrials.gov, NCT00652470) and received ethics approval from all participating institutions.
Conflict of interest
The members of the Steering Committee have no conflicts of interest with respect to this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Coulter, I.C., Kulkarni, A.V., Sgouros, S. et al. Cranial and ventricular size following shunting or endoscopic third ventriculostomy (ETV) in infants with aqueductal stenosis: further insights from the International Infant Hydrocephalus Study (IIHS). Childs Nerv Syst 36, 1407–1414 (2020). https://doi.org/10.1007/s00381-020-04503-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-020-04503-y