Abstract
Purpose
Posterior fossa arachnoid cysts (PFAC) are mostly considered as benign lesions of the cerebellum. Although many studies have shown the major role of the cerebellum in modulating movement, language, cognition, and social interaction, there are few studies on the cognitive impact and surgical decompression of PFAC.
Methods
We present the cases of two brothers successively diagnosed with PFAC and neuropsychological delay. After multidisciplinary discussion with the boys’ parents, it was decided to drain these lesions. Clinical signs, cerebral images, and neuropsychological status were assessed on admission and then 1 and 3 years after surgery.
Results
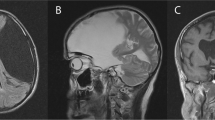
At presentation, both children had mild cerebellar signs, associated with cognitive and visual-motor impairments and academic regression. CT scans revealed retrovermian cysts, which were shunted. Post-operatively, both brothers demonstrated improved visual-motor skills and behavior. At follow-up, we observed disappearance of dysarthria and academic delay and significant improvement in cognition especially at the intelligence scale and in language. Fine motor skills had improved but remained slower than the average and writing skills appeared limited.
Conclusion
Except for PFAC which impair cerebrospinal fluid circulation or which are responsible for a significant mass effect, most PFAC are usually considered as “asymptomatic” and do not require surgical treatment. The two cases reported herein suggest that these lesions might be responsible for some associated but potentially reversible neuropsychological impairment. In the future, clinical assessment should include neuropsychological evaluation to help inform decision for surgical decompression in these children with PFAC.


Similar content being viewed by others
References
Aarhus M, Helland CA, Lund-Johansen M, Wester K, Knappskog PM (2010) Microarray-based gene expression profiling and DNA copy number variation analysis of temporal fossa arachnoid cysts. Cerebrospinal Fluid Res 7:6
Aarsen F, Van Dongen HR, Paquier PF, Van Mourik M, Castam-Berrvoets CE (2004) Long-term sequelae in children after cerebellar astrocytoma surgery. Neurology Apr 27 62(8):1311–1316
Ackermann H, Mathiak K, Riecker A (2007) The contribution of the cerebellum to speech production and speech perception: clinical and functional imaging data. Cerebellum 6(3):202–213. doi:10.1080/14734220701266742
Bellebaum C, Daum I (2011) Mechanisms of cerebellar involvement in associative learning. Cortex 47:128–136. doi:10.1016/j.cortex.2009.07.016
Boltshauser E, Martin F, Altermatt S (2002) Outcome in children with space-occupying posterior fossa arachnoid cysts. Neuropediatrics 33:118–121
Booth JR, Wood L, Lu D, Houk JC, Bitan T (2007) The role of the basal ganglia and cerebellum in language processing. Brain Res Feb 16;1133(1):136–144. doi:10.1016/j.brainres.2006.11.074
Bright R (1831) Serous cysts in the arachnoid. In: Reports of medical cases selected with a view of illustrating the symptoms and cure of diseases by a reference to morbid anatomy, volume 2: diseases of the brain nervous system, part 1. Published by Longman, Rees, Orme, Brown, Paternoster-Row and S. Highley, London, pp 437–439
Buckner RL (2013) The cerebellum and cognitive function: 25 years of insight from anatomy and neuroimaging. Neuron 80:807–815. doi:10.1016/j.neuron.2013.10.044
Cincu R, Agrawal A, Eiras J (2007) Intracranial arachnoid cysts: current concepts and treatment alternatives. Clin Neurol Neurosurg 109:837–843. doi:10.1016/j.clineuro.2007.07.013
De Volder AG, Michel C, Thauvoy C, Willems G, Ferriere G (1994) Brain glucose utilisation in acquired childhood aphasia associated with a sylvian arachnoid cyst: recovery after shunting as demonstrated by PET. J Neurol Neurosurg Psychiatry 57:296–300
Di Rocco C (2010) Sylvian fissure arachnoid cysts: we do operate on them but should it be done? Childs Nerv Syst 26:173–175. doi:10.1007/s00381-009-1041-1
D’Mello A, Stoodley C (2015) Cerebro-cerebellar circuits in autism spectrum disorder. Front Neurosci. doi:10.3389/fnins.2015.00408
Erdincler P, Kaynar MY, Bozkus H, Ciplak N (1999) Posterior fossa arachnoid cysts. Br J Neurosurg 13(1):10–17
Eskandary H, Sabba M, Khajehpour F, Eskandari M (2005) Incidental findings in brain computed tomography scans of 3000 head trauma patients. Surg Neurol 63:550–553. doi:10.1016/j.surneu.2004.07.049
Gjerde P, Schmid M, Hammar A, Wester K (2013) Intracranial arachnoid cysts: impairment of higher cognitive functions and postoperative improvement. J Neurodev Disord 5:21
Grill J, Viguier D, Kieffer V, Bulteau C, Sainte-Rose C, Hartmann O, Kalifa C, Dellatolas G (2004) Critical risk factors for intellectual impairment in children with posterior fossa tumors: the role of cerebellar damage. J Neurosurg 101(2 Suppl):152–158
Helland CA, Lund-Johansen M, Wester K (2010) Location, sidedness, and sex distribution of intracranial arachnoid cysts in a population-based sample. J Neurosurg 113(5):934–939
Huang J-H, Mei W-Z, Chen Y, Chen JW, Lin ZX (2015) Analysis on clinical characteristics of intracranial arachnoid cysts in 488 pediatric cases. Int J Clin Exp Med 8(10):18343–18350
Isaksen E, Leet TH, Helland CA, Wester K (2013) Maze learning in patients with intracranial arachnoid cysts. Acta Neurochir 155:841–848. doi:10.1007/s00701-013-1641-0
Jack A, Englander ZA, Morris JP (2011) Subcortical contributions to effective connectivity in brain networks supporting imitation. Neuropsychologia 49:3689–3698. doi:10.1016/j.neuropsychologia.2011.09.024
Karatekin C, Lazareff JA, Asarnow RF (2000) Relevance of the cerebellar hemispheres for executive functions. Pediatr Neurol 22:106–112
Koziol LF, Budding D, Andreasen N, D’Arrigo S, Bulgheroni S, Imamizu H, Ito M, Manto M, Marvel C, Parker K, Pezzulo G, Ramnani N, Riva D, Schmahmann JD, Vandervert L, Yamazaki T (2014) Consensus paper: the cerebellum’s role in movement and cognition. Cerebellum 13(1):151–177. doi:10.1007/s12311-013-0511-x
Laporte N, De Volder A, Bonnier C, Raftopoulos C, Sébire G (2012) Language impairment associated with arachnoid cysts: recovery after surgical treatment. Pediatr Neurol 46:44–47. doi:10.1016/j.pediatrneurol.2011.10.001
Levisohn L, Cronin-Golomb A, Schmahmann JD (2000) Neuropsychological consequences of cerebellar tumor resection in children: cerebellar cognitive affective syndrome in a paediatric population. Brain 123:1041–1050
Matsuda W, Akutsu H, Miyamoto S, Noguchi S, Tsunoda T, Sasaki M, Matsumura A (2010) Apparently asymptomatic arachnoid cyst: postoperative improvement of subtle neuropsychological impediment. Neurol Med Chir (Tokyo) 50:430–433
Morris Z, Whiteley WN, Longstreth WT, Weber F, Lee YC, Tsushima Y, Alphs H, Ladd SC, Warlow C, Wardlaw JM, Al-Shahi Salman R (2009) Incidental findings on brain magnetic resonance imaging: systematic review and meta-analysis. BMJ 339:b3016–b3016. doi:10.1136/bmj.b3016
Park YS, Eom S, Shim K-W, Kim D-S (2009) Neurocognitive and psychological profiles in pediatric arachnoid cyst. Childs Nerv Syst 25:1071–1076. doi:10.1007/s00381-009-0872-0
Pierce K, Courchesne E (2001) Evidence for a cerebellar role in reduced exploration and stereotyped behavior in autism. Biol Psychiatry 49:655–664
Pliatsikas C, Johnstone T, Marinis T (2014) Grey matter volume in the cerebellum is related to the processing of grammatical rules in a second language: a structural voxel-based morphometry study. Cerebellum 13:55–63. doi:10.1007/s12311-013-0515-6
Pomeranz S, Constantini S, Lubetzki-Korn I, Amir N (1991) Familial intracranial arachnoid cysts. Childs Nerv Syst Apr,7(2):100–102
Puget S, Boddaert N, Viguier D, Kieffer V, Bulteau C, Garnett M, Callu D, Sainte-Rose C, Kalifa C, Dellatolas G, Grill J (2009) Injuries to inferior vermis and dentate nuclei predict poor neurological and neuropsychological outcome in children with malignant posterior fossa tumors. Cancer 115:1338–1347. doi:10.1002/cncr.24150
Raeder MB, Helland CA, Hugdahl K, Wester K (2005) Arachnoid cysts cause cognitive deficits that improve after surgery. Neurology 64:160–162
Rengachary SS, Watanabe I (1981) Ultrastructure and pathogenesis of intracranial arachnoid cysts. J Neuropathol Exp Neurol 40:61–83. doi:10.1097/00005072-198140010-00007
Riva D, Giorgi C (2000) The cerebellum contributes to higher functions during development. Evidence from a Series of Children Surgically Treated for Posterior Fossa Tumours Brain 123:1051–1061
Riva D, Annunziata S, Contarino V, Erbetta A, Aquino D, Bulgheroni S (2013) Gray matter reduction in the vermis and CRUS-II is associated with social and interaction deficits in low-functioning children with autistic spectrum disorders: a VBM-DARTEL study. Cerebellum 12:676–685. doi:10.1007/s12311-013-0469-8
Riva D (2000) Cerebellar contribution to behaviour and cognition in children. J Neurolinguistics 13:215–225
Schmahmann JD, Sherman J (1998) The cerebellar cognitive affective syndrome. Brain 121:561–579
Schmahmann JD (2010) The role of the cerebellum in cognition and emotion: personal reflections since 1982 on the dysmetria of thought hypothesis, and its historical evolution from theory to therapy. Neuropsychol Rev 20:236–260. doi:10.1007/s11065-010-9142-x
Scott RB, Stoodley CJ, Anslow P, Paul C, Stein JF, Sugden EM, Mitchell CD (2001) Lateralized cognitive deficits in children following cerebellar lesions. Dev Med Child Neurol 43:685–691
Sinha S, Brown JIM (2004) Familial posterior fossa arachnoid cyst. Childs Nerv Syst 20:100–103. doi:10.1007/s00381-003-0808-z
Soukup VM, Patterson J, Trier TT, Chen JW (1998) Cognitive improvement despite minimal arachnoid cyst decompression. Brain Dev 20:589–593
Stoodley CJ, Schmahmann JD (2010) Evidence for topographic organization in the cerebellum of motor control versus cognitive and affective processing. Cortex 46(7):831–844. doi:10.1016/j.cortex.2009.11.008
Strick PL, Dum RP, Fiez JA (2009) Cerebellum and nonmotor function. Annu Rev Neurosci 32:413–434. doi:10.1146/annurev.neuro.31.060407.125606
Tamburrini G, Del Fabbro M, Di Rocco C (2008) Sylvian fissure arachnoid cysts: a survey on their diagnostic workout and practical management. Childs Nerv Syst 24:593–604. doi:10.1007/s00381-008-0585-9
Tavano A, Grasso R, Gagliardi C, Triulzi F, Bresolin N, Fabbro F, Borgatti R (2007) Disorders of cognitive and affective development in cerebellar malformations. Brain 130:2646–2660. doi:10.1093/brain/awm201
Vernooij MW, Arfan Ikram M, Tanghe HL, Vincent AJPE, Hofman A, Krestin GP, Niessen WJ, Breteler MMB, Van der Lugt A (2007) Incidental findings on brain MRI in the general population. N Engl J Med 357:1821–1828
Wester K (2008) Intracranial arachnoid cysts—do they impair mental functions? J Neurol 255:1113–1120. doi:10.1007/s00415-008-0011-y
Wester K, Hugdahl K (1995) Arachnoid cysts of the left temporal fossa: impaired preoperative cognition and postoperative improvement. J Neurol, Neurosurg & Psychiatry 59:293–298
Acknowledgments
The authors are grateful to the patients’ parents for their kindness answering questionnaires. This study was presented in part at the 40th annual meeting of the International Society of Pediatric Neurosurgery, Sydney, Australia, 2012, September 9–13.
We are indebted to Nikki Sabourin-Gibbs for editing the manuscript.
Authors’ contributions
Conception and design: MLC, PP, SP
Data analysis: MLC, MP, HP, PP, SP
Editing: MLC, MP, CSR, LVD, PP, SP
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
P. Piolino and S. Puget are co-last authors.
An erratum to this article is available at http://dx.doi.org/10.1007/s00381-017-3368-3.
Rights and permissions
About this article
Cite this article
Cuny, M.L., Pallone, M., Piana, H. et al. Neuropsychological improvement after posterior fossa arachnoid cyst drainage. Childs Nerv Syst 33, 135–141 (2017). https://doi.org/10.1007/s00381-016-3285-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-016-3285-x