Abstract
Purpose
The Butterfly Prostatic Retraction Device (“Butterfly”) is a permanent nitinol implant for benign prostatic hyperplasia. This study examines the chronic response of prostate tissue to the Butterfly in histological specimens from patients in the Butterfly pilot clinical study.
Methods
Retrospective qualitative and semi-quantitative review of histological specimens of seven (7) patients who participated in the Butterfly pilot clinical study. Patients had at least 1-month implantation with the Butterfly prior to implant removal and TURP. Tissue samples were graded by two pathologists.
Results
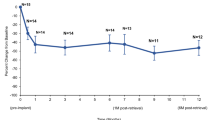
Four out of six patients had IPSS decreased from baseline. All seven patients’ samples had signs of chronic inflammation; one demonstrated acute inflammation and one demonstrated fibrosis. In three cases, intraglandular calcification was identified. There was no ischemic necrosis induced by the implant, and no encrustation, urethral edema, or cellular atypia was noted.
Conclusion
The Butterfly demonstrated an overall favorable safety profile in terms of tissue response. This study demonstrates that there is no significant tissue reaction in the prostatic urethra due to presence of Butterfly device.


Similar content being viewed by others
Data, material, and/or code availability
All relevant raw data are stored in a password protected drive. It is freely available to any scientist wishing to use them for non-commercial purposes. This data can be provided with request.
References
Fourcade RO, Théret N, Taïeb C (2008) Profile and management of patients treated for the first time for lower urinary tract symptoms/benign prostatic hyperplasia in four European countries. BJU Int 101:1111–1118. https://doi.org/10.1111/j.1464-410X.2008.07498.x
Gupta NK, Gange SN, McVary KT (2019) New and emerging technologies in treatment of lower urinary tract symptoms from benign prostatic hyperplasia. Sex Med Rev 7:491–498. https://doi.org/10.1016/j.sxmr.2018.02.003
Yang Q, Peters TJ, Donovan JL, Wilt TJ, Abrams P (2001) Transurethral incision compared with transurethral resection of the prostate for bladder outlet obstruction: a systematic review and meta-analysis of randomized controlled trials. J Urol 165:1526–1532. https://doi.org/10.1016/s0022-5347(05)66342-2
Sountoulides P, Karatzas A, Gravas S (2019) Current and emerging mechanical minimally invasive therapies for benign prostatic obstruction. Ther Adv Urol 11:1756287219828971. https://doi.org/10.1177/1756287219828971
Srinivasan A, Wang R (2020) An update on minimally invasive surgery for benign prostatic hyperplasia: techniques, risks, and efficacy. World J Mens Health 38:402–411. https://doi.org/10.5534/wjmh.190076
US Food and Drug Administration (2021) Non-clinical and clinical investigation of devices used for the treatment of benign prostatic hyperplasia (BPH). https://www.fda.gov/regulatory-information/search-fda-guidance-documents/non-clinical-and-clinical-investigation-devices-used-treatment-benign-prostatic-hyperplasia-bph
Armitage JN et al (2007) Epithelializing stent for benign prostatic hyperplasia: a systematic review of the literature. J Urol 177:1619–1624. https://doi.org/10.1016/j.juro.2007.01.005
Peyton CC, Badlani GH (2015) The management of prostatic obstruction with urethral stents. Can J Urol 22(Suppl 1):75–81
Dunnick NR, Sandler CM and Newhouse JH (1991) Textbook of uroradiology. Radiology vol. 181 (Lippincott Williams & Wilkins, 2012, 1991). https://doi.org/10.1148/radiology.181.2.438.
Roehrborn CG, Chin PT, Woo HH (2022) The UroLift implant: mechanism behind rapid and durable relief from prostatic obstruction. Prostate Cancer Prostatic Dis 25:79–85. https://doi.org/10.1038/s41391-021-00434-0
Katz R et al (2021) PD18–11 The ‘Butterfly’, a novel minimally invasive transurethral retraction device for BPH. J Urol. https://doi.org/10.1097/ju.0000000000002007.11
Balakrishnan N, Uvelius B, Zaszczurynski P, Lin DL, Damaser MS (2005) Biocompatibility of nitinol and stainless steel in the bladder: an experimental study. J Urol 173:647–650. https://doi.org/10.1097/01.ju.0000143197.93944.14
Wang D, Huang H, Law YM, Foo KT (2015) Relationships between prostatic volume and intravesical prostatic protrusion on transabdominal ultrasound and benign prostatic obstruction in patients with lower urinary tract symptoms. Ann Acad Med Singapore 44:60. https://doi.org/10.47102/annals-acadmedsg.v44n2p60
Mariappan P, Brown DJG, McNeill AS (2007) Intravesical prostatic protrusion is better than prostate volume in predicting the outcome of trial without catheter in white men presenting with acute urinary retention: a prospective clinical study. J Urol 178:573–577. https://doi.org/10.1016/j.juro.2007.03.116
Park YJ, Bae KH, Jin BS, Jung HJ, Park JS (2012) Is increased prostatic urethral angle related to lower urinary tract symptoms in males with benign prostatic hyperplasia/lower urinary tract symptoms? Korean J Urol 53:410–413. https://doi.org/10.4111/kju.2012.53.6.410
Nickel JC et al (2008) The relationship between prostate inflammation and lower urinary tract symptoms: examination of baseline data from the REDUCE trial. Eur Urol 54:1379–1384. https://doi.org/10.1016/j.eururo.2007.11.026
Delongchamps NB et al (2008) Evaluation of prostatitis in autopsied prostates-is chronic inflammation more associated with benign prostatic hyperplasia or cancer? J Urol 179:1736–1740. https://doi.org/10.1016/j.juro.2008.01.034
Morrison C, Thornhill J, Gaffney E (2000) The connective tissue framework in the normal prostate, B.P.H and prostate cancer: analysis by scanning electron microscopy after cellular digestion. Urol Res 28:304–307. https://doi.org/10.1007/s002400000123
Bushman WA, Jerde TJ (2016) The role of prostate inflammation and fibrosis in lower urinary tract symptoms. Am J Physiol 311:F817–F821. https://doi.org/10.1152/ajprenal.00602.2015
Cantiello F et al (2013) Periurethral fibrosis secondary to prostatic inflammation causing lower urinary tract symptoms: a prospective cohort study. Urology 81:1018–1024. https://doi.org/10.1016/j.urology.2013.01.053
Maglio M et al (2020) Histological, histomorphometrical, and biomechanical studies of bone-implanted medical devices: hard resin embedding. Biomed Res Int 2020:1804630. https://doi.org/10.1155/2020/1804630
Feld Y, Golan S, Leibovitch I (2018) Assessing the safety and efficacy of the ClearRing™ implant for the treatment of benign prostatic hyperplasia in a canine model. Curr Urol 11:206–211. https://doi.org/10.1159/000447220
Vjaters E et al (2020) First-in-man safety and efficacy of the ClearRing implant for the treatment of benign prostatic hyperplasia. Eur Urol Focus 6:131–136. https://doi.org/10.1016/j.euf.2018.09.013
Acknowledgements
We would like to thank Patho-Logica Ltd, Israel for supporting the initial analysis of the tissue specimen and Prof Rumelia Koren for help with providing images of the pathology and official pathology report.
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
RK: protocol/project development, data analysis, manuscript writing/editing; CS: data analysis, manuscript writing/editing; AP: data analysis, manuscript writing/editing; MAA: data collection or management, critical revision of manuscript; AS: data collection or management, critical revision of manuscript; SR: data collection or management, critical revision of manuscript; AZ: data collection or management, critical revision of manuscript; SA: data collection or management, critical revision of manuscript; JB: data collection or management, critical revision of manuscript; BC: protocol/project development, data collection or management, critical revision of manuscript, supervision.
Corresponding author
Ethics declarations
Conflict of interest
BC is a consultant for Boston Scientific and Olympus.
Ethical approval
The procedures used in this study adhere to the tenets of the Declaration of Helsinki. This study is a retrospective review of histological data of patients participating in the Butterfly pilot clinical study (NCT03912558), first posted on April 15, 2022 (ClinicalTrials.gov).
Consent to participate and publish
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Katz, R., Sze, C., Punyala, A. et al. Characterization of the histological response to the Butterfly Prostatic Retraction Device in patients with benign prostatic hyperplasia. World J Urol 41, 1141–1146 (2023). https://doi.org/10.1007/s00345-023-04319-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-023-04319-w