Abstract
Purpose
To evaluate the prognostic impact of the histological expression of CD133 in renal cell carcinoma (RCC).
Methods
From 1992 to 2009, 142 consecutive patients underwent radical nephrectomy or partial nephrectomy for RCC. All cases were reviewed by a single pathologist and then subjected to analysis of the immunohistochemical expression of CD133 using tissue microarray. Several clinical and pathological variables were also evaluated.
Results
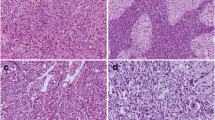
The median postoperative follow-up was 44 months. Of the 142 immunostained RCC specimens, 77 (54%) showed low and 65 (46%) high expression of CD133. Expression of CD133 was associated with clinical stage (P = 0.05), lymph node involvement (P = 0.03), metastatic disease (P = 0.02) and MVI (P = 0.03). Among other variables, clinical stage, necrosis and metastasis were associated with disease-specific survival (DSS) and progression-free survival (PFS) on univariate analysis. The 5-year PFS rates in patients who provided specimens with high and low expression of CD133 were 83 and 66%, respectively (P = 0.01). It was observed that the 5-year DSS for patients who provided specimens with high and low expression of CD133 was 90 and 71%, respectively (P = 0.003). Multivariate survival analysis showed that patients in the CD133 low-expression group had a higher probability of disease progression (HR 3.4, P = 0.02) and a higher probability of death from cancer (HR 2.4, P = 0.01).
Conclusions
Immunohistochemical expression of CD133 had an impact on survival in patients with RCC, which shows that CD133 might be a useful tool for risk stratification. Low expression of this marker remained as an independent predictor of poor DSS and PFS.


Similar content being viewed by others
References
American Cancer Society (2010) Cancer facts & figures 2010. American Cancer Society, Atlanta
Gupta K, Miller JD, Li JZ, Russell MW, Charbonneau C (2008) Epidemiologic and socioeconomic burden of metastatic renal cell carcinoma (mRCC): a literature review. Cancer Treat Rev 34:193–205
Kanao K, Mizuno R, Kikuchi E et al (2009) Preoperative prognostic nomogram (probability table) for renal cell carcinoma based on TNM classification. J Urol 181:480–485
Eichelberg C, Junker K, Ljungberg B, Moch H (2009) Diagnostic and prognostic molecular markers for renal cell carcinoma: a critical appraisal of the current state of research and clinical. Eur Urol 55:851–863
Park CH, Bergsagel DE, McCulloch EA (1971) Mouse myeloma tumor stem cells: a primary cell culture assay. J Natl Cancer Inst 46:411–422
Hombach-Klonisch S, Paranjothy T, Wiechec E et al (2008) Cancer stem cells as targets for cancer therapy: selected cancers as examples. Arch ImmunolTher Exp 56:165–180
Bussolati B, Bruno S, Grange C et al (2005) Isolation of renal progenitor cells from adult human kidney. Am J Pathol 166:545–555
Röper K, Corbeil D, Huttner WB (2000) Retention of prominin in microvilli reveals distinct cholesterol-based lipid micro-domains in the apical plasma membrane. Nat Cell Biol 2:582–592
Corbeil D, Röper K, Fargeas CA, Joester A, Huttner WB (2001) Prominin: a story of cholesterol, plasma membrane protrusions and human pathology. Traffic 2:82–91
Weigmann A, Corbeil D, Hellwig A, Huttner WB (1997) Prominin, a novel microvilli-specific polytopic membrane protein of the apical surface of epithelial cells, is targeted to plasmalemmal protrusions of non-epithelial cells. Proc NatlAcadSci 94:12425–12430
D’Alterio C et al (2010) Differential role of CD133 and CXCR4 in renal cell carcinoma. Cell Cycle 9:4491–4492
Budwit-Novotny DA, McCarty KS, Cox EB et al (1986) Immunohistochemical analyses of estrogen receptor in endometrial adenocarcinoma using a monoclonal antibody. Cancer Res 46(10):5419–5425
Frank I, Blute ML, Leibovich BC, Cheville JC, Lohse CM, Zincke H (2005) Independent validation of the 2002 American Joint Committee on cancer primary tumor classification for renal cell carcinoma using a large, single institution cohort. J Urol 173:1889–1892
Bedke J, Pritsch M, Buse S et al (2008) Prognostic stratification of localized renal cell carcinoma by tumor size. J Urol 180:62–67
Hollingsworth JM, Miller DC, Daignault S, Hollenbeck BK (2006) Rising incidence of small renal masses: a need to reassess treatment effect. J Natl Cancer Inst 98:1331–1334
Lane BR, Samplaski MK, Herts BR, Zhou M, Novick AC, Campbell SC (2008) Renal mass biopsy-a renaissance? J Urol 179:20–27
Klatte T, Seligson DB, LaRochelle J et al (2009) Molecular signatures of localized clear cell renal cell carcinoma to predict disease-free survival after nephrectomy. Cancer Epidemiol Biomarkers Prev 18:894–900
Mizrak D, Brittan M, Alison MR (2008) CD133: molecule of the moment. J Pathol 214:3–9
Sagrinati C, Netti GS, Mazzinghi B et al (2006) Isolation and characterization of multipotent progenitor cells from the Bowman’s capsule of adult human kidneys. J Am Soc Nephrol 17:2443–2456
Bruno S, Bussolati B, Grange C, Collino F, Graziano ME, Ferrando U, Camussi G (2006) CD133+ renal progenitor cells contribute to tumor angiogenesis. Am J Pathol 169(6):2223–2235
Dall’Oglio MF, Antunes AA, Sarkis AS et al (2007) Microvascular tumour invasion in renal cell carcinoma: the most important prognostic factor. BJU Int 100:552–555
Dall’Oglio MF, Ribeiro-Filho LA, Antunes AA et al (2007) Microvascular tumor invasion, tumor size and Fuhrman grade: a pathological triad for prognostic evaluation of renal cell carcinoma. J Urol 178:425–428
May M, Brookman-Amissah S, Kendel F et al (2009) Validation of a postoperative prognostic model consisting of tumor microvascular invasion, size, and grade to predict disease-free and cancer-specific survival of patients with surgically resected renal cell carcinoma. Int J Urol 16:616–621
Kume H, Suzuki M, Fujimura T et al (2010) Distant metastasis of renal cell carcinoma with a diameter of 3 cm or less-which is aggressive cancer? J Urol 184:64–68
Jeon Y et al (2010) Promoter hypermethylation and loss of CD133 gene expression in colorectal cancers. World J Gastroenterol 16(25):3153
Shimizu K, Itoh T, Shimizu M, Ku Y, Hori Y (2009) CD133 expression pattern distinguishes intraductal papillary mucinous neoplasms from ductal adenocarcinomas of the pancreas. Pancreas 38(8):e207–e214
Kemper K, Sprick MR, de Bree M, Scopelliti A, Vermeulen L, Hoek M, Zeilstra J, Pals ST, Mehmet H, Stassi G, Medema JP (2010) The AC133 epitope, but not the CD133 protein, is lost upon cancer stem cell differentiation. Cancer Res 70(2):719–729
Balch C, Nephew KP, Huang TH, Bapat SA (2007) Epigenetic “bi-valently marked” process of cancer stem cell-driven tumorigenesis. Bioessays 29:842–845
Ohm JE, McGarvey KM, Yu X, Cheng L, Schuebel KE, Cope L, Mohammad HP, Chen W, Daniel VC, Yu W, Berman DM, Jenuwein T, Pruitt K, Sharkis SJ, Watkins DN, Herman JG, Baylin SB (2007) A stem cell-like chromatin pattern may predispose tumor suppressor genes to DNA hypermethylation and heritable silencing. Nat Genet 39:237–242
Mathieu J et al (2011) HIF induces human embryonic stem cell markers in cancer cells. Cancer Res 71:4640–4652
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
da Costa, W.H., Rocha, R.M., da Cunha, I.W. et al. CD133 immunohistochemical expression predicts progression and cancer-related death in renal cell carcinoma. World J Urol 30, 553–558 (2012). https://doi.org/10.1007/s00345-011-0769-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-011-0769-x