Abstract
Manganese (Mn) is an essential micronutrient with positive effects on enhancing plant tolerance against fungal diseases. On the other hand, it has been suggested that metal–amino acid complexes are more effective than mineral sources in supplying Mn and improving the translocation and distribution of these metal nutrients in plants. Meanwhile, certain amino acids, that is, lysine and methionine, play an important role in crop resistance to biotic stresses. Little information is, however, available regarding the effect of foliar-applied Mn in mineral and complex forms with amino acids on crop tolerance to powdery mildew caused by Podosphaera fuliginea. In this nutrient solution experiment, the effect of foliar-applied Mn, in the form of MnSO4, Mn-methionine (Mn-Met) and Mn-lysine (Mn-Lys), on certain defense mechanisms of cucumber (Cucumis sativus L. cv. Espadana RZ) against powdery mildew was investigated. Different Mn sources were applied as foliar spray 4 days before and 6 days after inoculation with P. fuliginea. The results revealed that Mn application prior to pathogen inoculation decreased the disease in the treated and newly growing leaves; however, the efficiency of Mn–amino acid complexes in disease suppression was greater than that of MnSO4. Post-inoculation spray of Mn had no effect on the disease reduction of the newly growing leaves, but all Mn sources diminished disease severity equally in the treated leaves. In such leaves, regardless of the application time, the higher content of cell wall lignin was found in the plants treated with MnSO4, in comparison with those treated with Mn–amino acid complexes. In the newly growing leaves, the foliar application of Mn–amino acid complexes before inoculation resulted in a significant increase of leaf cell wall lignin content, whereas MnSO4 was ineffective. Pre-inoculation spray of MnSO4 led to increasing PO and PPO activities in the treated leaves, whereas Mn–amino acid complexes had no significant effect on the activity of these enzymes in the same leaves. Application of Mn-Met and Mn-Lys after pathogen inoculation led to a significant decrease of PPO activity in the treated leaves. In general, the results of the present study revealed that the time of Mn foliar application could have a considerable effect on controlling cucumber powdery mildew. Foliar application of Mn before infection with P. fuliginea could strengthen the defensive structures of the plant and help to prevent the disease incidence.
Similar content being viewed by others
Introduction
Powdery mildew is known as one of the most important diseases of vegetable crops, particularly cucumber (Cucumis sativus L.), under field and greenhouse conditions worldwide. Powdery mildew in cucumber is commonly caused by Podosphaera fuliginea (Schltdl.) and Golovinomyces cichoracearum (DC.); however, P. fuliginea is found more commonly on cucurbits than E. cichoracearum in greenhouse conditions. The development of P. fuliginea seems to be faster under greenhouse conditions (Zitter and others 1996).
The destructive effects of powdery mildews have been reported in greenhouse cucumbers in many parts of the world, including Iran. This fungal disease affects plant leaves, stems, and fruits, resulting in the considerable reduction of the quantitative and qualitative yield of cucumber (Dik and others 2004). Powdery mildew leads to the whitish, talcum-like powdery growth on leaf surfaces, petioles, and stems. Infected leaves usually wither and die, and the plants senesce prematurely (Zitter and others 1996). Severe infection by powdery mildew before the flowering stage can reduce the cucumber fruit yield by 20–40% (McGrath 1996). This disease can also disrupt photosynthesis and respiration processes, leading to the reduced fruit set, inadequate ripening, and poor flavor development (McGrath 1996).
Systemic and protectant fungicides are commonly used to control powdery mildew, but the continued application of fungicides has several negative consequences including the development of fungicide-resistant races of the pathogen (Reuveni and others 1996), the increase of residue levels (O’ Brien 1993), and serious risks for human health (Damalas and Eleftherohorinos 2011). Therefore, safe alternative methods for disease control are essential.
Among various environmental factors affecting crop tolerance to plant diseases, plant nutrition is of great importance, so proper plant nutrition management can significantly improve the resistance and tolerance of the plant to fungal diseases (Marschner 1995; Krauss 1999). Micronutrients can reduce the severity of diseases by affecting the physiology and biochemistry of the plant or by affecting the response of plants to pathogens (Marschner 1995). Nutrient deficiency causes the higher leakage of reducing sugars and amino acids outside the plant cell, thereby resulting in the higher susceptibility of the plant cell to pathogens (Dordas 2008).
Manganese (Mn) is an essential micronutrient for the growth and development of plants; it plays several biochemical and physiological roles in plants. A possible role for Mn is the enhancement of the plant’s defense system to diseases. Application of Mn leads to boosting the photosynthesis level and metabolic activities; it also promotes the plant’s competitive ability, thus enhancing plant tolerance to diseases (Graham and Webb 1991). Another role of Mn is the induction of protective mechanisms and the increase of the host’s resistance. The direct effect of Mn on increasing the host’s resistance is through the production of inhibitors or accumulation of the toxic levels of Mn around the site of infection and the subsequent absorption by the pathogen (Huber and William 1988). Mn nutrition can also affect plant’s resistance to diseases via strengthening lignification, increasing the concentration of soluble phenolic compounds, and inhibiting pectin methyl esterase and amino peptidase activities (Graham and Webb 1991). Mn regulates auxin levels in the plant tissues by activating photosynthesis, especially photosystem II (Marschner 1986). Auxin can induce the systemic resistance of the plant against diseases and increase cell division and cell development (Devlin and Witham 1983).
Several studies have shown that the external application of manganese induces systemic acquired resistance (SAR) and increases the defense capacity of plant tissues (Reuveni and others 1997; Yao and others 2012). It has been shown that excess concentrations of Mn in plant leaves significantly enhance salicylic acid concentration and increase the expression of proteins involved in ethylene and jasmonic acid synthesis. The proteins related to pathogen resistance could also be enhanced by the excess Mn, including a PR-like protein, and this could be accompanied by the increased activity of phenylalanine ammonia lyase (Yao and others 2012).
Mn is one of the active ingredients of some fungicides (Römheld and Marschner 1991). The role of optimum Mn nutrition in reducing some plant diseases such as the root rot of the cluster bean, the take-all patch of the bent grass, and the tan spot of the winter durum wheat has been proved in numerous studies (Heckman and others 2003; Simoglou and Dordas 2006; Wadhwa and others 2012).
There are also reports indicating the nutritional effect of Mn in the powdery mildew control. For example, the development of the powdery mildew of swede was greatly declined with increasing the soil application of Mn (Brain and Whlttington 1981). In another study, the foliar spray of Mn or Mn-containing fungicides decreased the powdery mildew of wheat (Huber and Keeler 1977). Furthermore, the fertilization of Mn to prevent Mn deficiency decreased the severity of the powdery mildew of barley (Vlamis and Yarwood 1962), wheat (Colquhoun 1940), and pumpkin (Abia and others 1977). Mn also controls biosynthesis of lignin via the activation of certain enzymes of the shikimic acid and phenylpropanoid pathways (Römheld and Marschner 1991; Marschner 1995). Yao and others (2012) indicated that high leaf Mn concentrations (less than 2500 µg g−1) enhanced the resistance of grapevine to powdery mildew by causing some delay in pathogen spreading after fungal inoculation, but Mn did not directly prevent pathogen growth on a long-term basis.
Mineral sources (that is, Mn sulfate) are common sources of Mn used in agricultural soils. Due to the low mobility of Mn within phloem in most plant species (Rengel 2001), the efficiency of these minerals Mn fertilizer sources is low. Recently, metal–amino acid complexes have been synthesized and introduced as effective fertilizer sources of micronutrients, for example, Fe and Zn (Ghasemi and others 2012). Amino acids have the ability to coordinate metal ions (such as Mn) via their carboxyl groups (Aravind and Prasad 2005) and can form stable complexes with micronutrients and improve the translocation and distribution of these metal nutrients in plants (Ducic and Polle 2005). In addition, application of amino acids has been reported to be effective in enhancing crop resistance to abiotic and biotic stresses (Kauffman and others 2007; Parađiković and others 2011; Cerdán and others 2013). For example, aspartate-derived amino acids, that is, lysine and methionine, were found to be elevated during pathogen infection in Arabidopsis (Yang and Ludewig 2014).
Although the beneficial effect of Mn nutrition on promoting host resistance and tolerance to fungal diseases has been proved in numerous studies, inadequate information is available regarding the mechanism of Mn impact on the reduction of plant disease. Furthermore, the effectiveness of Mn foliar spray at different times (before and after pathogen inoculation) in preventing and controlling disease has not been evaluated yet. In this study, the effectiveness of foliar-applied Mn in the mineral complex forms was studied with amino acids in two application time points (before and after pathogen inoculation) to examine the possible effects on inducing the local and systemic resistance of cucumber against P. fuliginea. To better understand the possible mechanisms of Mn impact, peroxidase and polyphenol oxidase activities and the lignin content of cell wall were evaluated in treated and newly growing leaves of cucumber individually.
Materials and methods
Plant Growth, Treatment Application, and Experimental Design
The experiment was carried out in the research greenhouse of Isfahan University of Technology, Isfahan, Iran. Seeds of cucumber (Cucumis sativus L. cv. Espadana RZ) were thoroughly rinsed with distilled water and germinated on moist filter paper in an incubator at 28 °C. Espadana RZ is one of the most commonly grown cultivars of cucumber in greenhouses of Iran. After germination, the seeds were sown in sterilized quartz sand and grown up to the cotyledon stage. The germinated seeds were first irrigated with distilled water and then with Johnson nutrient solution (1:10). After 1 week, cucumber seedlings were transplanted to the pots (2 L) containing the Johnson nutrient solution. The composition of the nutrient solution was 1.0 mM KNO3, 1.0 mM Ca(NO3)2, 1.0 mM NH4H2PO4, 1.0 mM MgSO4, 50 μM FeEDTA, 50 μM KCl, 25 μM H3BO3, 2.0 μM MnSO4, 2.0 μM ZnSO4, 0.5 μM CuSO4, and 0.5 μM H2Mo7O4. The pH of the nutrient solution was adjusted in the range of 5.8–6 using 0.1 N HCl or KOH when needed. The nutrient solution in the pots was continuously aerated with pumps and renewed weekly. The daily photoperiod in the greenhouse was 14 h, with the average daily maximum and minimum temperatures of 30 and 18 °C, respectively.
Three Mn sources including MnSO4 and synthesized Mn-lysine (Lys), and Mn-methionine (Mn-Met) were applied as the foliar spray. The concentration of Mn in all foliar-applied treatments was 0.5% (w/v). To distinguish Mn and amino acid effects, a group of plants were supplied with only Lys and Met. Control plants received no Mn and amino acids via foliar application. The control plants were sprayed with distilled water. At the 6-leaf stage, the upper surface of the cucumber leaves was sprayed with fresh aqueous solutions of fertilizers. In half of the plants, foliar spray treatments were applied 4 days before inoculation and for the other half, 6 days after inoculation with P. fuliginea. These times of Mn foliar application were selected based on the results obtained from a preliminary test (data not shown).
Pathogen Inoculation
The source of powdery mildew fungus (P. fuliginea) was obtained from infected cucumber plants in a local greenhouse at Isfahan province and maintained on cucumber plants by periodic transfer to the new plants when necessary. Inoculum was obtained from freshly sporulating colonies on the infected leaves of cucumber 9–12 days after inoculation. Conidia were gently brushed into 100 mL distilled water containing 20 µL Tween-20 and counted with the aid of a hemocytometer to give a concentration of 106 conidia/mL. For inoculation, the upper surfaces of all leaves were sprayed with a conidial suspension supplied by a hand sprayer. After inoculation, plants were incubated in a dew chamber at 20 °C for 24 h in darkness. Plants were then returned to the greenhouse bench (16–20 °C at night and 22–32 °C during the day, 14 h of light per day) for disease development.
Sample Collection
Fourteen days after inoculation, three fully recently expanded leaves of plants at the same position of each leaf group (the treated and newly growing leaves) were collected, frozen in liquid nitrogen, and kept in a −80 °C freezer for biochemical analysis.
Assay of Disease Severity
Disease severity was evaluated when disease symptoms were fully observed (14 days after inoculation with P. fuliginea). The percentage of the infected leaf area to the healthy leaf area (in the newly growing leaves and the treated leaves) of each tested plant was determined using the leaf area meter (Winarea-UT-11) device.
Enzyme Assay
The plant leaf samples were homogenized in a pestle and a mortar with a 100 mM TRIS–HCl buffer (pH 8) containing 2 mM EDTA, 5 mM DLdithiothreitol, 10% glycerol, 100 mM sodium borate, 4% (wt/vol) insoluble polyvinylpyrrolidone (PVP), and 1 mM phenylmethylsulphonylfluoride (PMSF). The homogenate was filtered through four layers of muslin cloth and centrifuged at 12,000×g for 40 min. The supernatant was used for enzyme analyses immediately. Total protein was determined using the Bradford method (1976).
Peroxidase (PO) Assay
Leaf activity of PO was assayed using the modified method proposed by Hammerschmidt and others (1982). The reaction mixture consisted of 1.5 mL of 0.01 M sodium phosphate buffer (pH 6) and 50 μL of the enzyme extract. To start the reaction, equal concentrations of guaiacol and 1% H2O2 (0.5 mL each) were added. The change in absorbance at 470 nm was recorded at 30-s intervals for 2 min, whereas the boiled enzyme preparation served as the blank. The enzyme activity was expressed as the changes in the absorbance min− 1 g FW− 1.
Polyphenol Oxidase (PPO) Assay
Leaf activity of PPO was determined according to the procedure proposed by Mayer and others (1965). The reaction mixture consisted of 1.5 mL of 0.1 M sodium phosphate buffer (pH 6.5) and 50 μL of the enzyme extract. To start the reaction, 50 μL of 0.01 M catechol was added. The change in the absorbance was recorded at 30-s intervals for 3 min and the PPO activity was expressed as the change in absorbance at 495 nm min− 1 g FW− 1.
Isolation of the Leaf Cell Wall (CW)
The fresh leaves were frozen and ground to powder using liquid nitrogen. A sample (2 g) of the powdered leaf material was homogenized in distilled water. The suspension was then centrifuged for 10 min at 10,000×g. The supernatant discarded and the precipitated materials were resuspended in distilled water and centrifuged as done in the previous step. The precipitation was washed two times with 10 volumes of absolute ethanol, rinsed in chloroform:methanol (1:2, v/v) for 12 h, and washed by acetone. The CW pellet was resuspended and filtered after each wash. The samples were finally dried overnight with air stream at 35 °C. The residue, considered as CW material, was weighed and kept in a desiccator at room temperature until use.
Determination of Leaf Cell Wall Lignin Content
Isolated leaf cell walls were used for the determination of lignin content. Lignin content was measured via a modified acetyl bromide (AcBr) procedure (Iiyama and Wallis 1990). To put it briefly, 6 mg of fine-powdered wall preparation was treated with a mixture (total of 2.5 mL) of 25% (w /w) AcBr in AcHO and 0.1 mL of 70% HClO ~ at 70 °C for 30 min, by shaking at 10-min intervals. After cooling with ice, the digestion mixture was transferred to a 50-mL volumetric flask containing 10 mL of 2 M NaOH and 12 mL AcHO and made up to 50 mL. The lignin content was determined by measuring the absorbance at 280 nm using a specific absorption coefficient of 20.0 g− 1 L cm− 1.
Elemental Analysis
At the end of the experiment, the cucumber seedlings were harvested by cutting with a sterilized razor blade at the stem point leveled to the upper surface of plant supporting plates and separated into shoot and root. Treated and newly growing leaves were also separated. Plant materials were first rinsed in tap water and then washed with distilled water. The plant tissues were oven dried at 70 °C for 48 h, weighed, and ground. The ground samples of plant tissues were dry-ashed at 500 °C, dissolved in 2 N HCl, and made to volume with hot distilled water. Root and shoot Mn concentration was determined by atomic absorption spectrophotometer (Perkin Elmer, Model 3030, US). All parameters were measured in treated and newly growing leaves separately.
Statistical Analysis
Treatments were arranged in a factorial test in a completely randomized design with three replications. The significance of the effects of the treatments and their interactions was evaluated by analysis of variance (ANOVA) with the multiple comparison post hoc test, using SAS software (version 9.1), and significant differences between treatment means were determined by comparison of means test (LSD test), at the 5% significance level. Correlation coefficients between the measured parameters were obtained by SPSS software (version 17.0).
Results
The results of the analysis of variance for the fertilizer and the application time in relation to the measured parameters are presented in Table 1.
Disease Severity in Treated and Newly Growing Leaves
The effect of foliar-applied Mn on the severity of powdery mildew was dependent on the time of application and Mn source (Fig. 1a, b). Application of Mn before inoculation with pathogen resulted in disease reduction in the treated and newly growing leaves, although this reduction was greater at the Mn–amino acid complexes treatments, in comparison with the MnSO4 treatment.
The effect of foliar-applied Mn in different forms including MnSO4 and the one complexed with lysine (Lys) and methionine (Met), at two application times (4 days before and 6 days after pathogen inoculation), on the infected leaf area of the treated (a) and newly growing (b) leaves of cucumber. Columns with similar letters are not significant at P < 0.05. Error bars represent the standard errors
Foliar application of Mn-Lys and Mn-Met after pathogen inoculation significantly reduced the disease severity on the treated leaves of cucumber, but the foliar spray of MnSO4 was ineffective in this regard (Fig. 1a). Severity of disease on the newly growing leaves was not affected by foliar-applied Mn treatments.
Foliar application of Mn after pathogen inoculation was ineffective in reducing the severity of disease on the newly growing leaves (Fig. 1b). The post-inoculation sprays of free amino acids significantly decreased disease severity on the treated leaves, while the pre-inoculation of Lys resulted in the increase of disease severity (Fig. 1a).
Mn Concentrations in Treated and Newly Growing Leaves
Foliar application of Mn resulted in the significant enhancement of Mn concentration in the treated and newly growing leaves of cucumber (Fig. 2a, b). For the treated leaves, the lower concentration of Mn was found at the Mn-Met treatment, while for the newly growing leaves, Mn-Met and Mn-Lys were more effective than MnSO4 in increasing the leaf Mn concentration (Fig. 2a, b). The range of Mn concentration was sufficient (120–150 mg kg−1) in the treated leaves and low (30–50 mg per kg) in the newly growing leaves.
The effect of foliar-applied Mn in different forms including MnSO4 and the one complexed with lysine (Lys) and methionine (Met), at two application times (4 days before and 6 days after pathogen inoculation), on Mn concentration of the treated (a) and newly growing (b) leaves of cucumber. Columns with similar letters are not significant at P < 0.05. Error bars represent the standard errors
Lignin Content of Leaf Cell Wall
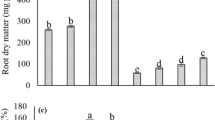
The effect of Mn foliar spray on the leaf lignin content varied depending on the source and time of application. In the treated leaves, regardless of the application time (4 days before inoculation or 6 days after inoculation), the higher content of lignin was found in the plants treated with MnSO4, in comparison with those treated with Mn–amino acid complexes (Fig. 3a). The use of Mn-Met after pathogen inoculation had no effect on the lignin content of the treated leaves.
The effect of foliar-applied Mn in different forms including MnSO4 and the one complexed with lysine (Lys) and methionine (Met), at two application times (4 days before and 6 days after pathogen inoculation), on the lignin content of the treated (a) and newly growing (b) leaves of cucumber. Columns with similar letters are not significant at P < 0.05. Error bars represent the standard errors
For the newly growing leaves, foliar application of MnSO4 prior to pathogen inoculation had no significant effect on the leaf cell wall lignin content, while application of Mn-Lys and Mn-Met resulted in the significant increase of the lignin content (Fig. 3b).
Activity of PO
In the treated leaves, pre-inoculation spray of MnSO4 led to the increase of PO activity, whereas Mn–amino acid complexes had no significant effect on the activity of this enzyme (Fig. 4a). Post-inoculation application of Mn-Met and MnSO4 resulted in the significant decrease of PO activity in the treated leaves, while Mn-Lys had no effect on the PO activity (Fig. 4a).
The effect of foliar-applied Mn in different forms including MnSO4 and the one complexed with lysine (Lys) and methionine (Met), at two application times (4 days before and 6 days after pathogen inoculation), on PO activity of the treated (a) and newly growing (b) leaves of cucumber. Columns with similar letters are not significant at P < 0.05. Error bars represent the standard errors
Foliar application of Mn and amino acids before inoculation with pathogen resulted in the higher activity of PO in the newly growing leaves, although this increase was the smallest at the MnSO4 treatment (Fig. 4b). Foliar application of MnSO4 and Mn-Met after pathogen inoculation resulted in the higher activity of PO in the newly growing leaves, while Mn-Lys was ineffective.
Activity of PPO
In the treated leaves, pre-inoculation spray of MnSO4 led to the increase of PPO activity, whereas Mn–amino acid complexes had no significant effect on the activity of this enzyme (Fig. 5a). Post-inoculation application of Mn-Met and Mn-Lys resulted in the significant decrease of PPO activity in the treated leaves, while MnSO4 had no effect on the PPO activity (Fig. 5a).
The effect of foliar-applied Mn in different forms including MnSO4 and the one complexed with lysine (Lys) and methionine (Met), at two application times (4 days before and 6 days after pathogen inoculation), on PPO activity of the treated (a) and newly growing (b) leaves of cucumber. Columns with similar letters are not significant at P < 0.05. Error bars represent the standard errors
Foliar application of Mn and amino acids before inoculation with pathogen resulted in the higher activity of PPO in the newly growing leaves, although this increase was the smallest at the Mn-Met treatment (Fig. 5b). Foliar application of amino acids as free or complexed forms with Mn after pathogen inoculation resulted in the lower activity of PPO in the newly growing leaves, while MnSO4 had no effect on the activity of the enzyme.
Discussion
Based on the results obtained from the present study, the effects of foliar-applied Mn on cucumber tolerance to powdery mildew caused by P. fuliginea were dependent on (1) the timing of application, before and after pathogen inoculation; (2) the age of leaves, treated and newly growing leaves; and (3) Mn fertilizer source, mineral form and the one complexed with amino acids.
Comparison of the treatment effects based on the two applications times indicated that pre-inoculation Mn spray, regardless of the source used, was more effective than post-inoculation Mn application in disease reduction on both treated and newly growing leaves. In general, it could be argued that Mn applied before inoculation with pathogen had a controlling effect on the treated leaves and a protective effect on the newly growing leaves against powdery mildew. Amino acid–Mn complexes, that is, Mn-Lys and Mn-Met, had a larger effect on pathogen suppression than MnSO4 did.
This confirmed that disease suppression on the plants supplied with Mn–amino acid complexes before inoculation with pathogen could also be associated with the effect of accompanying ligands as well as the Mn function. As shown in Fig. 1, amino acids were more effective than sulfate in disease suppression.
In contrast to Mn–amino acid complexes, free amino acids applied before pathogen inoculation had no effect on cucumber tolerance to powdery mildew or increased disease severity, as compared to the control.
It was likely that at this application time (before inoculation), amino acids were accumulated in apoplast spaces of the leaves, providing the required conditions for fungus growth and development. Amino acids are nutritional sources providing carbon and nitrogen for pathogens and their high concentrations in the leaf apoplast spaces of host plants could stimulate the growth of penetrating pathogens in the intercellular space and the cell wall (Hancock and Huisman 1981). High levels of nitrogen supplied via amino acids could also help to soften the leaf tissue and lead to the vulnerability of the cell wall. In the case of powdery mildew, pathogen that could have access only to the epidermal cells, physical and chemical properties of these cells, particularly chemical composition and cell wall properties, could be much more important than those of the leaf tissue in determining host susceptibility or resistance to disease (Hwang and others 1983).
The reason that pre-inoculation spray of Mn–amino acid complexes, unlike the free amino acids, decreased the disease severity could be related to the less biodegradability of amino acids in these complexes. In the previous studies, it has been shown that the stability constant of metal–amino acid complexes is higher than that of free amino acids; as a result, the availability of amino acids complexed with metal is less than that of free amino acids for microorganisms (Ghasemi and others 2013a).
It was interesting that in contrast to the pre-inoculation treatment, foliar application of free amino acids after pathogen inoculation diminished disease severity. This reduction might be associated with the role of Lys and Met in inducing plant resistance against pathogen. The effect of certain amino acids on the improvement of plant resistance to biotic and abiotic stresses has been reported (Zeier 2013). Distinct amino acid metabolic pathways constitute the integral parts of the plant immune system. Mutations in several genes involved in Asp-derived amino acid biosynthetic pathways play an important role in the plant resistance to specific pathogens. The catabolism of Lys produces the immune signal pipecolic acid (Pip), a cyclic, non-protein amino acid. Moreover, the acylation of amino acids can control plant resistance to pathogens and pests by the formation of protective plant metabolites, or by the modulation of the plant hormone activity (Zeier 2013). It has been recently found that the aspartate-derived amino acids, such as lysine and methionine, are increased during pathogen infection in Arabidopsis. Pipecolic acid, a lysine catabolite, has been identified as a critical regulator of the systemic acquired resistance. It is mobile in plants, functioning as an intensifier of defense responses and mediating systemic acquired resistance establishment via signal amplification (Yang and Ludewig 2014). Furthermore, a gene encoding a diaminopimelate aminotransferase could be involved in Lys biosynthesis, resulting in the elevated disease resistance via an elevated salicylic acid (SA) pathway in Arabidopsis (Hudson and others 2006).
Methionine is an essential sulfur-containing amino acid and an intermediate in the biosynthesis of the polyamines and the plant-ripening hormone ethylene (Amir 2008). Apart from its role as a protein constituent and its central role in the initiation of mRNA translation, methionine is a fundamental metabolite in plant cells; this is since, through its first metabolite S-adenosylmethionine (SAM), it controls the levels of several key metabolites such as ethylene, polyamines, and biotin. SAM is also the primary methyl group donor regulating different processes in plants (Amir 2008). It has been shown that Met serves as a donor for various secondary metabolites involved in pathogen and insect defense (Gigolashvili and others 2007). Methionine is the precursor for the biosynthesis of S-adenosylmethionine (SAM), which is a methyl donor for several reactions in lignin biosynthesis (Campbell and Sederoff 1996).
Based on the results, it could be concluded that Mn foliar nutrition is effective in improving the plant tolerance to powdery mildew if Mn supplementation occurs several days before pathogen inoculation. To the best of our knowledge, this is the first report indicating the effect of foliar application time of Mn fertilizer sources on fungal disease.
Mobility of Mn in the phloem of most plant species is low (Rengel 2001). Therefore, it seems that at post-inoculation foliar spray treatments, applied Mn could not be sufficiently transferred to the newly growing leaves, whereas the supplied Mn in pre-inoculation time tends to have more distribution in the newly growing leaves.
Our findings regarding leaf Mn concentration also showed that the pre-inoculation of Mn foliar application was more effective than post-inoculation foliar application in increasing the Mn concentration in the newly growing leaves. The highest concentration of Mn in the newly growing leaves was obtained by Mn–amino acid complexes; this was accompanied with the maximum disease depression in these leaves. These results suggested that in addition to Mn, the accompanying amino acids affected the disease control. After inoculation, the applied Mn in the form of complexes with amino acids resulted in the higher translocation of Mn to the newly growing leaves. Accordingly, disease reduction in foresaid leaves of plants treated with Mn–amino acid complexes was higher than that of plants treated by MnSO4.
Zhang and others (2015) showed that the uptake of Mn from Mn-Lys complex was higher than that of MnSO4 in the primary rat intestinal epithelial cells. They indicated that the uptake of Mn from Mn-Lys complex might be transported by cationic amino acid transporter (CAT) 1 and system bo,+; this was different from the ionized Mn2+ uptake pathway.
The elevated activity of PPO and PO in the treated leaves of plants sprayed with MnSO4 before inoculation could be related to the higher concentration of Mn in these leaves. The higher activity of PO and PPO in the newly growing leaves of plants treated with Mn–amino acid complexes, as compared with those treated with MnSO4 before pathogen inoculation, could be due to the higher translocation of Mn from old leaves to the young ones in the presence of amino acids. In fact, Mn was probably more accessible for plants treated with Mn–amino acid complexes than those treated with MnSO4. Several studies have shown that amino acids can increase the symplastic uptake and improve the translocation and distribution of metallic elements in plants (Ducic and Polle 2005; Na¨sholm and others 2009; Ghasemi and others 2013b). The mechanism of resistance induction in the plant supplied with Mn might be associated with the increase of PO and PPO activity in the leaves. Similarly, Reuveni and others (1997) reported the increase of the soluble fraction of PO in the upper leaves of plants treated with Mn.
The significant increase of the lignin-synthesizing enzymes PO and PPO in the plant supplied with Mn was accompanied with the higher lignin content of the leaf cell wall. In the newly growing leaves, in terms of spraying before inoculation, the anion associated with Mn was more important than Mn in enhancing the lignin content; this was such that Mn–amino acid complexes increased the lignin content to a higher level than that of MnSO4. These observations are in agreement with translocation and accumulation of Mn, as well as disease depression, especially in the newly growing leaves.
The higher lignin content of the plants supplied with Mn–amino acid complexes, as compared with those supplied with MnSO4, indicated that other factors such as accompanied amino acids might also have been involved. Evidence for the claim is that the free Met led to increasing the lignin content in the treated and newly growing leaves.
Conclusion
Foliar application of Mn was effective in inducing the systemic resistance of cucumber against powdery mildew caused by P. fuliginea only before pathogen inoculation.
According to the results, it was revealed that Mn–amino acid complexes including Mn-Lys and Mn-Met were more effective than MnSO4 in the suppression of the disease on the treated and newly growing leaves.
This might be due to the higher translocation of Mn from the treated leaves to the newly growing leaves and the direct protective effect of Lys and Met in the plants supplied with the Mn–amino acid complexes. The higher activity of lignin-synthesizing enzymes and, as a result, the higher lignification of the leaf cell wall led to the induction of systemic and local resistance to powdery mildew in the Mn-supplied cucumber plants. Further studies need to be carried out to identify the changes in the composition and properties of the leaf cell wall under Mn treatment. We did not measure other enzymes involved in lignin synthesis, but, certainly, it might also be useful to measure the activity of other enzymes in further experiments. According to the results obtained, foliar application of Mn before infection with Podosphaera fuliginea could be useful for enhancing cucumber tolerance to the disease, while Mn supplementation after the emergence of the disease symptoms was almost ineffective.
References
Abia JA, Hess WM, Smith BN (1977) Mn reduces susceptibility of Pumpkin seedlings to Sclerotinia sclerotiorum. Naturwissenschaften 64:437–438
Amir R (2008) Towards improving methionine content in plants for enhanced nutritional quality. Funct Plant Sci Biotechnol 2(1):36–46
Aravind P, Prasad MNV (2005) Cadmium-induced toxicity reversal by zinc in Ceratophyllum demersum L. (a free floating aquatic macrophyte) together with exogenous supplements of amino and organic acids. Chemosphere 61(11):1720–1733
Bradford M (1976) A rapid and sensitive method for the quantitation of protein utilizing the principle of protein-dye binding. Annu Rev Biochem 72:248–254
Brain PJ, Whhttington WJ (1981) The influence of soil pH on the severity of swede powdery mildew Erysiphe cruciferarum infection. Plant Phathol 30:105–110
Campbell MM, Sederoff RR (1996) Variation in lignin content and composition (mechanisms of control and implications for the genetic improvement of plants). Plant Physiol 110(1):3–13
Cerdán M, Sánchez-Sánchez A, Jordá JD, Juárez M, Sánchez-Andreu J (2013) Effect of commercial amino acids on iron nutrition of tomato plants grown under lime-induced iron deficiency. J Plant Nutr Soil Sci 176:859–866
Colquhoun TT (1940) Effect of Mn on powdery mildew of wheat. J Aust Inst Agric Sci 6:54
Damalas CA, Eleftherohorinos IG (2011) Pesticide exposure, safety issues, and risk assessment indicators. Int J Environ Res Publ Health 8(5):1402–1419
Devlin RM, Witham FH (1983) Plant Physiology, 4th edn. A division of wads worth, Inc., Wadads Worth Publishing Co., Belmont
Dik AJ, Gaag DJVD, Pijnakker J, Wubben JP (2004) Integrated control strategies for all pests and diseases in several glasshouse crops and implementation in practice. Bulletin OILB/SROP 27(8):35–39
Dordas Ch (2008) Role of nutrients in controlling plant diseases in sustainable agriculture: a review. Agron Sustain Dev 28:33–46
Ducic T, Polle A (2005) Transport and detoxification of manganese and copper in plants. Braz J Plant Physiol 17(1):103–112
Ghasemi S, Khoshgoftarmanesh AH, Hadadzadeh H, Jafari M (2012) Synthesis of iron-amino acid chelates and evaluation of their efficacy as iron source and growth stimulator for tomato in nutrient solution culture. J Plant Growth Regul 31:498–508
Ghasemi S, Khoshgoftarmanesh AH, Afyuni M, Hadadzadeh H, Schulin R (2013a) Zinc-amino acid complexes are more stable than free amino acids in saline and washed soils. Soil Biol Biochem 63:73–79
Ghasemi S, Khoshgoftarmanesh AH, Hadadzadeh H, Afyuni M (2013b) Synthesis, characterization, and theoretical and experimental investigations of zinc (II)–amino acid complexes as ecofriendly plant growth promoters and highly bioavailable sources of Zinc. J Plant Growth Regul 32:315–323
Gigolashvili T, Yatusevich R, Berger B, Müller C, Flügge UI (2007) The R2R3-MYB transcription factor HAG1/MYB28 is a regulator of methionine-derived glucosinolate biosynthesis in Arabidopsis thaliana. Plant J 51:247–261
Graham DR, Webb MJ (1991) Micronutrients and disease resistance and tolerance in plants. In: Mortvedt JJ, Cox FR, Shuman LM, Welch RM (eds) Micronutrients in agriculture, 2nd edn. Soil Sci. Soc. Amer. Inc., Madison, pp 329–370
Hammerschmidt R, Nuckles EM, Kuc J (1982) Association of enhanced peroxidase activity with induced systemic resistance of cucumber to Colletotrichum lagenarium. Physiol Plant Pathol 20:73–82
Hancock GC, Huisman OC (1981) Nutrient movement in host-pathogen systems. Ann Rev Phytopathol 19:309–311
Heckman JR, Clarke BB, Murphy JA (2003) Optimizing manganese fertilization for the suppression of take-all patch disease on creeping bent grass. Crop Sci 43:1395–1398
Huber DM, Keeler RR (1977) Alteration of wheat peptidase activity after infection with powdery mildew. P Am Phytophatol Soc 4:163
Huber DM, Wilhelm NS (1988) The role of manganese in resistance to plant diseases. In: Graham RD, Hannam RJ, Uren NC (eds) Manganese in soils and plants. Kluwer Academic Publishers, Dordrecht, pp 155–173
Hudson AO, Singh BK, Leustek T, Gilvarg T (2006) An LL-diaminopimelate aminotransferase defines a novel variant of the lysine biosynthesis pathway in plants. Plant Physiol 140:292–301
Hwang BK, Ibenthal WD, Heitefuss R (1983) Age, rate of growth, carbohydrate and amino acid contents of spring barley plants in relation to their resistance to powdery mildew, (Erysiphe graminis f.sp.hordei). Physiol Plant Pathol 22:1–14
Iiyama K, Wallis AFA (1990) Determination of lignin in herbaceous plants by an improved acetyl bromide procedure. J Sci Food Agric 51:145–161
Kauffman GL, Kneivel DP, Watschke TL (2007) Effects of a bio stimulant on the heat tolerance associated with photosynthetic capacity, membrane thermo stability, and polyphenol production of perennial ryegrass. Crop Sci Soc Am 47(1):261–267
Krauss A (1999) Balanced Nutrition and Biotic Stress, IFA Agricultural Conference on Managing Plant Nutrition, 29 June-2 July 1999. Barcelona, Spain
Marschner H (1986) Mineral nutrition of higher plants. Academic Press, London
Marschner H (1995) Mineral nutrition of higher plants, 2nd edn. Academic Press Limited, London
Mayer AM, Harel E, Shaul RB (1965) Assay of catechol oxidase a critical comparison of methods. Phytochemistry 5:738–789
McGrath MT (1996) Successful management of powdery mildew in pumpkin with disease threshold-based fungicide programs. Plant Dis 80:910–916
Na¨sholm T, Kielland K, Ganeteg U (2009) Uptake of organic nitrogen by plants. New Phytol 182:31–48
O’Brien RG (1993) Fungicide resistance in populations of cucurbit powdery mildew (Sphaerotheca fuliginea). N Z J Crop Hortic Sci 22:145–149
Parađiković N, Vinković T, Vinković Vrček I, Žuntar I, Bojić M, Medić-Šarić M (2011) Effect of natural biostimulants on yield and nutritional quality: an example of sweet yellow pepper (Capsicum annuum L.) plants. J Sci Food Agric 91(12):2146–2152
Rengel Z (2001) Xylem and phloem transport of micronutrients. In: Horst WJ et al (ed) Plant nutrition—food security and sustainability of agro-ecosystems. Kluwer Academic Publishers, Dordrecht, pp 628–629
Reuveni M, Agapov V, Reuveni FL (1996) Controlling powdery mildew caused by Sphaerotheca fuliginea in cucumber by foliar sprays of phosphate and potassium salts. Crop Prot 15:49–53
Reuveni M, Agapov V, Reuveni R (1997) A foliar spray of micronutrient solutions induces local and systemic protection against powdery mildew (Sphaerotheca fuliginia) in cucumber plants. Eur J Plant Pathol 103:581–588
Römheld V, Marschner H (1991) Function of micronutrients in plants. In: Mortvedt JJ, Cox FR, Shuman LM, Welch RM (eds) Micronutrients in agriculture. SSSA Inc., Madison, WI, pp 297–328
Simoglou KB, Dordas Ch (2006) Effect of foliar applied boron, manganese and zinc on tan spot in winter durum wheat. Crop Prot 25:657–663
Vlamis J, Yarwood CE (1962) Effect of liming of soil on barley powdery mildew. Plant Dis Rep 46:886–887
Wadhwa N, Joshi UN, Gandhi SK (2012) Copper and manganese increase resistance of cluster bean to root rot caused by Rhizoctonia. J Phytopathol 161:172–179
Yang H, Ludewig U (2014) Lysine catabolism, amino acid transport, and systemic acquired resistance; hat is the link? Plant Signal Behav 9:e289331-e289334
Yao YA, Wang J, Ma XM, Lutts S, Sun Ch, Ma J, Yang Y, Achal V, Xu G (2012) Proteomic analysis of Mn-induced resistance to powdery mildew in grapevine. J Exp Bot 63(14):5155–5170
Zeier J (2013) New insights into the regulation of plant immunity by amino acid metabolic pathways. Plant Cell Environ 36:2085–2103
Zhang H, Gilbert ER, Zhang K, Ding X, Luo Y, Wang J, Zeng Q, Bai S (2015) Uptake of manganese from manganese–lysine complex in the primary rat intestinal epithelial cells. J Anim Physiol Anim Nutr (Berl) 101(1):147–158
Zitter TA, Hopkins DL, Thomas CE (1996) Compendium of cucurbit diseases. American Phytopathological Society (APS) Press, Saint Paul
Acknowledgements
This study was supported by Isfahan University of Technology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eskandari, S., Khoshgoftarmanesh, A.H. & Sharifnabi, B. The Effect of Foliar-Applied Manganese in Mineral and Complex Forms with Amino Acids on Certain Defense Mechanisms of Cucumber (Cucumis sativus L.) Against Powdery Mildew. J Plant Growth Regul 37, 481–490 (2018). https://doi.org/10.1007/s00344-017-9747-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-017-9747-x