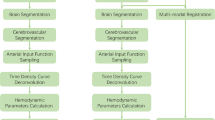
Abstract
Objective
To determine the reproducibility of quantitative computed tomography perfusion (CTP) parameters generated using different post-processing methods and identify the relative impact of subjective factors on the robustness of CTP parameters in acute ischemic stroke (AIS).
Materials and methods
A total of 80 CTP datasets from patients with AIS or transient ischemic attack (TIA) were retrospectively post-processed by two observers using different regions of interest (ROI) types, input models, and software. The CTP parameters were derived for 10 parenchymal ROIs. The intra-class correlation coefficients (ICCs) were used to assess the reproducibility of the CTP parameters for various post-processing methods. The Spearman correlation test was used to detect potential relationships between software and input models.
Results
The ICCs with 95% confidence intervals (CIs) were 0.94 (0.93–0.96), 0.94 (0.92–0.96), 0.82 (0.79–0.86), and 0.87 (0.85–0.90) for inter-reader agreement by using elliptic ROI, irregular ROI, single-input model, and dual-input model, respectively. The ICCs with 95% CI were 0.98 (0.98–0.98), 0.46 (0.43–0.50), and 0.25 (0.20–0.30) for inter-ROI type, inter-input model, and inter-software agreement, respectively.
Conclusions
Although the CTP parameters were stable when measured using different readers with different ROI types, they varied for different input models and software. The standardization of CTP post-processing is essential to reduce variability of CTP values.
Key Points
• The CTP parameters derived by different readers with different ROI types have agreements that range from good to excellent.
• The CTP parameters derived from different input models and software programs have poor agreement but significant correlations.





Similar content being viewed by others
Abbreviations
- AIF:
-
Arterial input function
- AIS:
-
Acute ischemic stroke
- CBF:
-
Cerebral blood flow
- CBV:
-
Cerebral blood volume
- CTP:
-
Computed tomography perfusion
- CTT:
-
Computed tomography technologist
- ICC:
-
Intra-class correlation coefficient
- MCA:
-
Middle cerebral artery
- MTT:
-
Mean transit time
- ROI:
-
Region of interest
- SVD:
-
Singular value decomposition
- TIA:
-
Transient ischemic attack
- TTP:
-
Time to peak
- VOF:
-
Venous output function
References
Powers WJ, Rabinstein AA, Ackerson T et al (2018) 2018 Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 49:e46–e110
Nogueira RG, Jadhav AP, Haussen DC et al (2018) Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. N Engl J Med 378:11–21
Albers GW, Marks MP, Kemp S et al (2018) Thrombectomy for stroke at 6 to 16 hours with selection by perfusion imaging. N Engl J Med 378:708–718
Broocks G, Hanning U, Faizy TD et al (2019) Ischemic lesion growth in acute stroke: water uptake quantification distinguishes between edema and tissue infarct. J Cereb Blood Flow Metab. https://doi.org/10.1177/0271678X19848505
Arenillas JF, Cortijo E, Garcia-Bermejo P et al (2018) Relative cerebral blood volume is associated with collateral status and infarct growth in stroke patients in SWIFT PRIME. J Cereb Blood Flow Metab 38:1839–1847
Furlanis G, Ajcevic M, Stragapede L et al (2018) Ischemic volume and neurological deficit: correlation of computed tomography perfusion with the National Institutes of Health Stroke Scale score in acute ischemic stroke. J Stroke Cerebrovasc Dis 27:2200–2207
Biggs D, Silverman ME, Chen F, Walsh B, Wynne P (2019) How should we treat patients who wake up with a stroke? A review of recent advances in management of acute ischemic stroke. Am J Emerg Med 37:954–959
Giles MF, Albers GW, Amarenco P et al (2011) Early stroke risk and ABCD2 score performance in tissue- vs time-defined TIA: a multicenter study. Neurology 77:1222–1228
Easton JD, Saver JL, Albers GW et al (2009) Definition and evaluation of transient ischemic attack: a scientific statement for healthcare professionals from the American Heart Association/American Stroke Association Stroke Council; Council on Cardiovascular Surgery and Anesthesia; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular Nursing; and the Interdisciplinary Council on Peripheral Vascular Disease. The American Academy of Neurology affirms the value of this statement as an educational tool for neurologists. Stroke 40:2276–2293
Tung CE, Olivot JM, Albers GW (2014) Radiological examinations of transient ischemic attack. Front Neurol Neurosci 33:115–122
Kamalian S, Kamalian S, Konstas AA et al (2012) CT perfusion mean transit time maps optimally distinguish benign oligemia from true “at-risk” ischemic penumbra, but thresholds vary by postprocessing technique. AJNR Am J Neuroradiol 33:545–549
Kolossvary M, De Cecco CN, Feuchtner G, Maurovich-Horvat P (2019) Advanced atherosclerosis imaging by CT: radiomics, machine learning and deep learning. J Cardiovasc Comput Tomogr 13:274–280
Goyal M, Jadhav AP, Bonafe A et al (2016) Analysis of workflow and time to treatment and the effects on outcome in endovascular treatment of acute ischemic stroke: results from the SWIFT PRIME randomized controlled trial. Radiology 279:888–897
Murase K, Nanjo T, Ii S et al (2005) Effect of x-ray tube current on the accuracy of cerebral perfusion parameters obtained by CT perfusion studies. Phys Med Biol 50:5019–5029
Wintermark M, Smith WS, Ko NU, Quist M, Schnyder P, Dillon WP (2004) Dynamic perfusion CT: optimizing the temporal resolution and contrast volume for calculation of perfusion CT parameters in stroke patients. AJNR Am J Neuroradiol 25:720–729
Van der Schaaf I, Vonken EJ, Waaijer A, Velthuis B, Quist M, van Osch T (2006) Influence of partial volume on venous output and arterial input function. AJNR Am J Neuroradiol 27:46–50
Zussman BM, Boghosian G, Gorniak RJ et al (2011) The relative effect of vendor variability in CT perfusion results: a method comparison study. AJR Am J Roentgenol 197:468–473
Koopman MS, Berkhemer OA, Geuskens R et al (2019) Comparison of three commonly used CT perfusion software packages in patients with acute ischemic stroke. J Neurointerv Surg 11:1249–1256
Sakai Y, Delman BN, Fifi JT et al (2018) Estimation of ischemic core volume using computed tomographic perfusion. Stroke 49:2345–2352
Kao YH, Mu Huo Teng M, Kao YT et al (2014) Automatic measurements of arterial input and venous output functions on cerebral computed tomography perfusion images: a preliminary study. Comput Biol Med 51:51–60
Soares BP, Dankbaar JW, Bredno J et al (2009) Automated versus manual post-processing of perfusion-CT data in patients with acute cerebral ischemia: influence on interobserver variability. Neuroradiology 51:445–451
Calamante F (2013) Arterial input function in perfusion MRI: a comprehensive review. Prog Nucl Magn Reson Spectrosc 74:1–32
Kudo K, Sasaki M, Yamada K et al (2010) Differences in CT perfusion maps generated by different commercial software: quantitative analysis by using identical source data of acute stroke patients. Radiology 254:200–209
Koo TK, Li MY (2016) A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med 15:155–163
Waaijer A, van der Schaaf IC, Velthuis BK et al (2007) Reproducibility of quantitative CT brain perfusion measurements in patients with symptomatic unilateral carotid artery stenosis. AJNR Am J Neuroradiol 28:927–932
Riordan AJ, Bennink E, Viergever MA, Velthuis BK, Dankbaar JW, de Jong HW (2013) CT brain perfusion protocol to eliminate the need for selecting a venous output function. AJNR Am J Neuroradiol 34:1353–1358
Bennink E, Oosterbroek J, Horsch AD et al (2015) Influence of thin slice reconstruction on CT brain perfusion analysis. PLoS One 10:e0137766
Fieselmann A, Kowarschik M, Ganguly A, Hornegger J, Fahrig R (2011) Deconvolution-based CT and MR brain perfusion measurement: theoretical model revisited and practical implementation details. Int J Biomed Imaging 2011:467563
Campbell BC, Christensen S, Levi CR et al (2011) Cerebral blood flow is the optimal CT perfusion parameter for assessing infarct core. Stroke 42:3435–3440
Kasasbeh AS, Christensen S, Parsons MW, Campbell B, Albers GW, Lansberg MG (2019) Artificial neural network computer tomography perfusion prediction of ischemic core. Stroke 50:1578–1581
Acknowledgments
We are grateful to Miss Guo of CT Kinetics, GE Healthcare, China, for providing the CK software.
Funding
This study has received funding from the Jilin Province Special Scientific Research Project of Health, with additional funding support from the Health Special Project of Finance Department of Jilin Province and funding support from National Key Research and Development Program of China. (No.2018YFC0116400)]. The first author of this study (Zhong-ping Chen) is the key coinvestigator of this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Dan Tong.
Conflict of interest
One of the authors of this manuscript (Yan Guo) is an employee of GE Healthcare. The remaining authors declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• cross-sectional study
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOC 93 kb)
Rights and permissions
About this article
Cite this article
Chen, ZP., Shi, ZZ., Li, YG. et al. Post-processing of computed tomography perfusion in patients with acute cerebral ischemia: variability of inter-reader, inter-region of interest, inter-input model, and inter-software. Eur Radiol 30, 6561–6569 (2020). https://doi.org/10.1007/s00330-020-07000-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-07000-7