Abstract
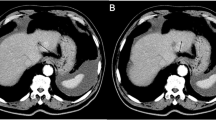
The purpose of this study was to evaluate the ability of MRI to detect magnetic particle uptake into advanced solid malignant tumors and to document the extension of these tumors, carried out in the context of magnetic drug targeting. In a prospective phase I trial, 11 patients were examined with MRI before and after magnetic drug targeting. The sequence protocol included T1-WI and T2-WI in several planes, followed by quantitative and qualitative evaluation of the signal intensities and tumor extensions. In nine patients, a signal decrease was observed in the early follow-up (2–7 days after therapy) on the T2-weighted images; two patients did not show a signal change. The signal changes in T1-WI were less distinct. In late follow-up (4–6 weeks after therapy), signal within nine tumors reached their initially normal level on both T1-WI and T2-WI; two tumors showed a slight signal decrease on T2-WI and a slight signal increase on T1-WI. Within the surveillance period, tumor remission in 3 out of 11 patients was observed, and in 5 patients tumor growth had stopped. The remaining three patients showed significant tumor growth. There was no statistically significant correlation between signal change and response. MRI is a suitable method to detect magnetite particles, deposited at the tumor site via magnetic drug targeting. MRI is therefore eligible to control the success of MDT and to assess the tumor size after the end of therapy.



Similar content being viewed by others
References
Alexiou C, Jurgons R, Schmid RJ, Bergemann C, Henke J, Erhardt W, Huenges E, Parak F (2003) Magnetic drug targeting—biodistribution of the magnetic carrier and the chemotherapeutic agent mitoxantrone after locoregional cancer treatment. J Drug Target 11:139–149
Mamot C, Drummond DC, Greiser U, Hong K, Kirpotin DB, Marks JD, Park JW (2003) Epidermal growth factor receptor (EGFR)-targeted immunoliposomes mediate specific and efficient drug delivery to EGFR- and EGFRvIII-overexpressing tumor cells. Cancer Res 63:3154–3161
Chen Y, Mcculloch RK, Gray BN (1994) Synthesis of albumin-Dextran sulfate microspheres possessing favourable loading and release characteristics for the anticancer drug Doxorubicin. J Control Release 31:49–54
Lubbe AS, Bergemann C, Huhnt W, Fricke T, Riess H, Brock JW, Huhn D (1996) Preclinical experiences with magnetic drug targeting: tolerance and efficacy. Cancer Res 56:4694–4701
Bonnemain B (1998) Superparamagnetic agents in magnetic resonance imaging: physicochemical characteristics and clinical applications. A review. J Drug Target 6:167–174
Kumano S, Murakami T, Kim T, Hori M, Okada A, Sugiura T, Noguchi Y, Kawata S, Tomoda K, Nakamura H (2003) Using superparamagnetic iron oxide-enhanced MRI to differentiate metastatic hepatic tumors and nonsolid benign lesions. Am J Roentgenol 181:1335–1339
Reimer P, Balzer T (2003) Ferucarbotran (Resovist): a new clinically approved RES-specific contrast agent for contrast-enhanced MRI of the liver: properties, clinical development, and applications. Eur Radiol 13:1266–1276
Mack MG, Balzer JO, Straub R, Eichler K, Vogl TJ (2002) Superparamagnetic iron oxide-enhanced MR imaging of head and neck lymph nodes. Radiology 222:239–244
Sigal R, Vogl T, Casselman J, Moulin G, Veillon F, Hermans R, Dubrulle F, Viala J, Bosq J, Mack M, Depondt M, Mattelaer C, Petit P, Champsaur P, Riehm S, Dadashitazehozi Y, de Jaegere T, Marchal G, Chevalier D, Lemaitre L, Kubiak C, Helmberger R, Halimi P (2002) Lymph node metastases from head and neck squamous cell carcinoma: MR imaging with ultrasmall superparamagnetic iron oxide particles (Sinerem MR)—results of a phase-III multicenter clinical trial. Eur Radiol 12:1104–1113
Bluemke DA, Weber TM, Rubin D, de Lange EE, Semelka R, Redvanly RD, Chezmar J, Outwater E, Carlos R, Saini S, Holland GA, Mammone JF, Brown JJ, Milestone B, Javitt MC, Jacobs P (2003) Hepatic MR imaging with ferumoxides: multicenter study of safety and effectiveness of direct injection protocol. Radiology 228:457–464
Lee JM, Kim CS, Youk JH, Lee MS (2003) Characterization of focal liver lesions with superparamagnetic iron oxide-enhanced MR imaging: value of distributional phase T1-weighted imaging. Korean J Radiol 4:9–18
Elmi MM, Sarbolouki MN (2001) A simple method for preparation of immuno-magnetic liposomes. Int J Pharm 215:45–50
Alexiou C, Arnold W, Klein R, Parak F, Hulin P, Bergemann C, Erhardt W, Wagenpfeil S, Lubbe A (2002) Locoregional cancer treatment with magnetic drug targeting. Cancer Res 60:6641–6648
Lubbe AS, Alexiou C, Bergemann C (2001) Clinical applications of magnetic drug targeting. J Surg Res 95:200–206
Montet X, Lazeyras F, Howarth N, Mentha G, Rubbia-Brandt L, Becker CD, Vallee JP, Terrier F (2004) Specificity of SPIO particles for characterization of liver hemangiomas using MRI. Abdom Imaging 29:60–70
Couvreur P, Kante B, Lenaerts V, Scailteur V, Roland M, Speiser P (1980) Tissue distribution of antitumor drugs associated with polyalkylcyanoacrylate nanoparticles. J Pharm Sci 69:199–202
Weissleder R, Bogdanov A, Neuwelt EA, Papisov M (1995) Longcirculating iron oxides for MR imaging. Adv Drug Deliv Rev 16:321–334
Schutt W, Gruttner C, Hafeli U, Zborowski M, Teller J, Putzar H, Schumichen C (1997) Applications of magnetic targeting in diagnosis and therapy—possibilities and limitations: a mini-review. Hybridoma 16:109–117
Brigger I, Dubernet C, Couvreur P (2002) Nanoparticles in cancer therapy and diagnosis. Adv Drug Deliv Rev 54:631–651
Wang YX, Hussain SM, Krestin GP (2001) Superparamagnetic iron oxide contrast agents: physicochemical characteristics and applications in MR imaging. Eur Radiol 11:2319–2331
Reimer P, Tombach B (1998) Hepatic MRI with SPIO: detection and characterization of focal liver lesions. Eur Radiol 8:1198–1204
Schima W (2002) Organ specific MRI contrast media in general practice. Wien Med Wochenschr (Suppl) 113:8–11
Jordan A, Wust P, Scholz R, Tesche B, Fahling H, Mitrovics T, Vogl T, Cervos-Navarro J, Felix R (1996) Cellular uptake of magnetic fluid particles and their effects on human adenocarcinoma cells exposed to AC magnetic fields in vitro. Int J Hyperthermia 12:705–722
Jordan A, Scholz R, Wust P, Fahling H, Krause J, Wlodarczyk W, Sander B, Vogl T, Felix R (1997) Effects of magnetic fluid hyperthermia (MFH) on C3H mammary carcinoma in vivo. Int J Hyperthermia 13:587–605
Thomassen T, Wiggen UN, Gundersen HG, Fahlvik AK, Aune O, Klaveness J (1991) Structure activity relationship of magnetic particles as MR contrast agents. Magn Reson Imaging 9:255–258
Babincova M, Babinec P, Bergemann C (2001) High-gradient magnetic capture of ferrofluids: implications for drug targeting and tumor embolization. Z Naturforsch [C] 56:909–911
Acknowledgments
This study was supported by the Graduiertenkolleg GRK 331 “Temperaturabhängige Effekte in Therapie und Diagnostik” of the German Research Foundation [Deutsche Forschungsgemeinschaft (DFG)].
Author information
Authors and Affiliations
Corresponding author
Additional information
The used data are part of the thesis of M.-I. Senfft von Pilsach.
Rights and permissions
About this article
Cite this article
Lemke, AJ., Senfft von Pilsach, MI., Lübbe, A. et al. MRI after magnetic drug targeting in patients with advanced solid malignant tumors. Eur Radiol 14, 1949–1955 (2004). https://doi.org/10.1007/s00330-004-2445-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-004-2445-7